Titrations of acids and bases Titrations of acids

Titrations of acids and bases
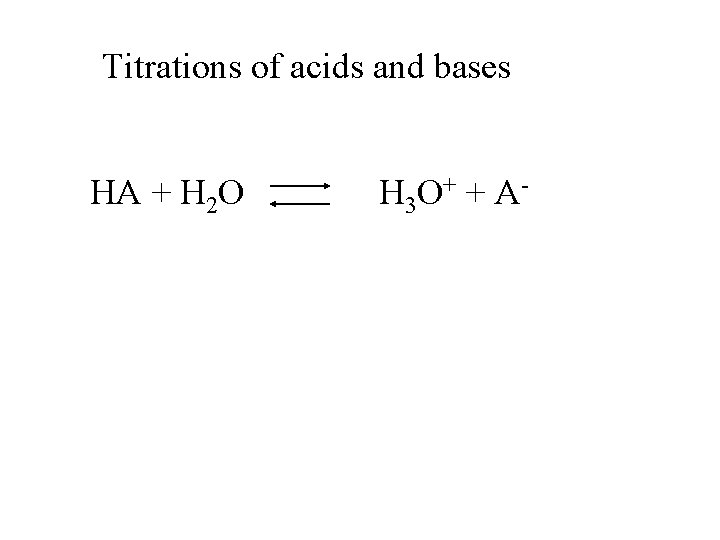
Titrations of acids and bases HA + H 2 O H 3 O + + A -
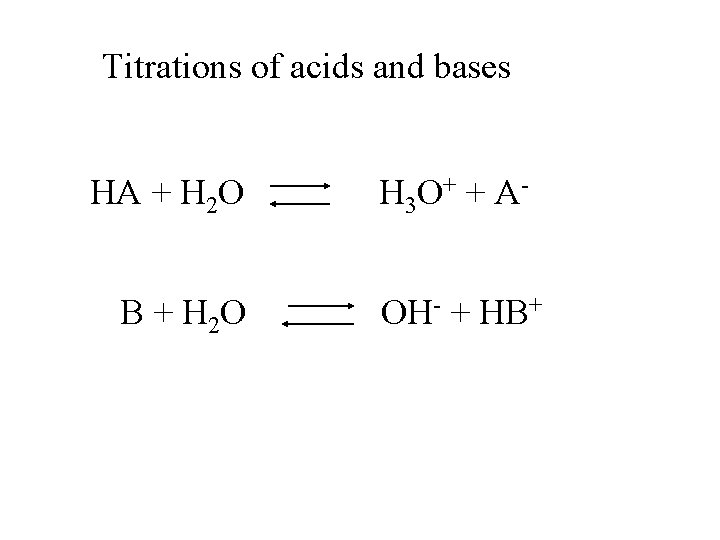
Titrations of acids and bases HA + H 2 O B + H 2 O H 3 O + + A OH- + HB+
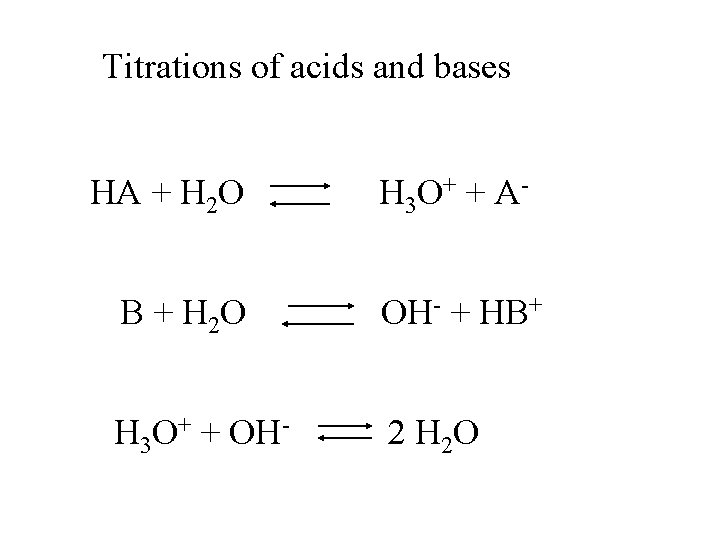
Titrations of acids and bases HA + H 2 O H 3 O + + A - B + H 2 O OH- + HB+ H 3 O+ + OH- 2 H 2 O
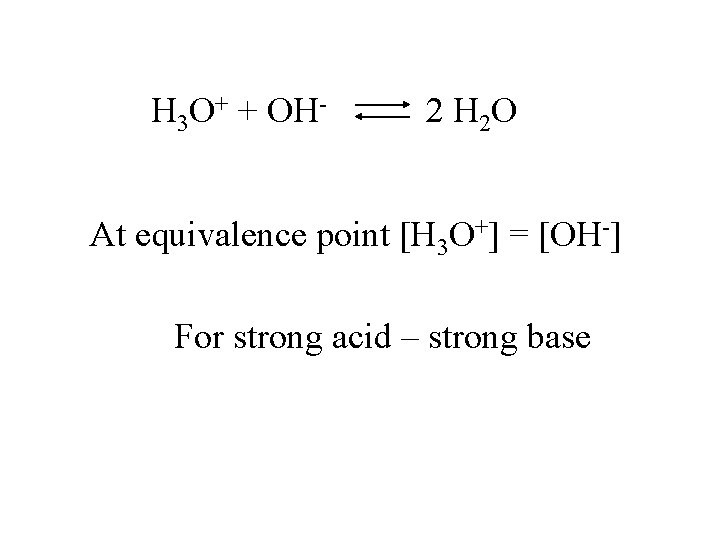
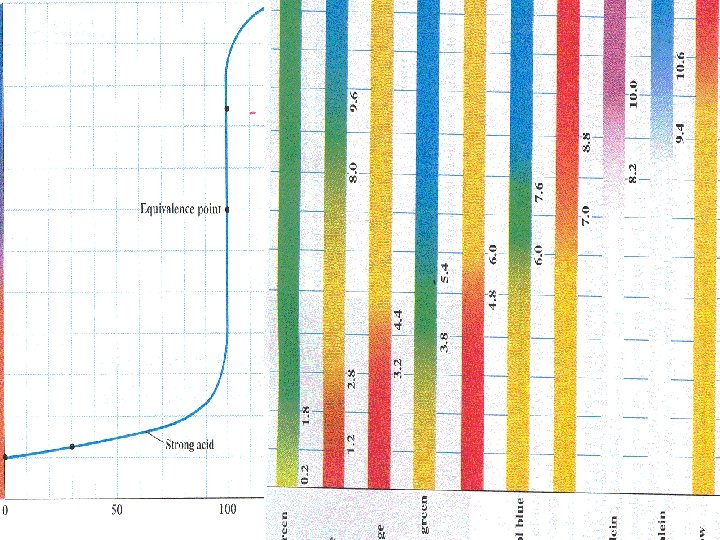
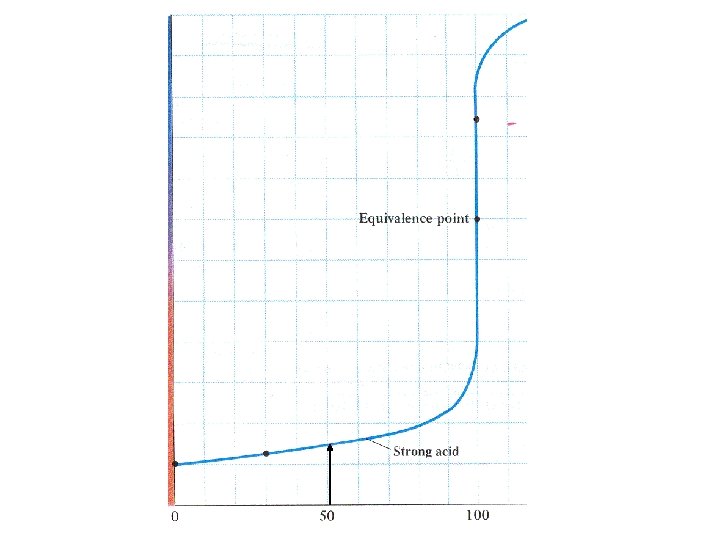
H 3 O+ + OH- 2 H 2 O At equivalence point [H 3 O+] = [OH-] For strong acid – strong base

H 3 O+ + OH- 2 H 2 O At equivalence point [H 3 O+] = [OH-] Kw = [ H 3 O +] = [OH-] = 10 -14

H 3 O+ + OH- 2 H 2 O At equivalence point [H 3 O+] = [OH-] Kw = [ H 3 O +] = [OH-] = 10 -14 p. H = 7
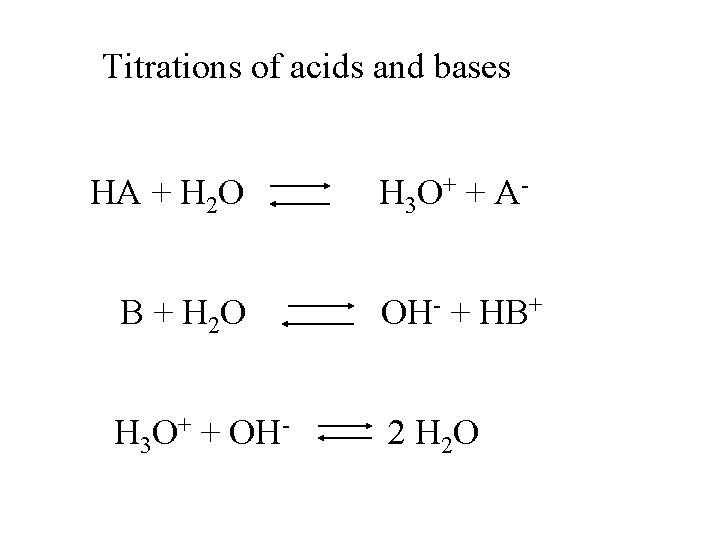
Titrations of acids and bases HA + H 2 O H 3 O + + A - B + H 2 O OH- + HB+ H 3 O+ + OH- 2 H 2 O
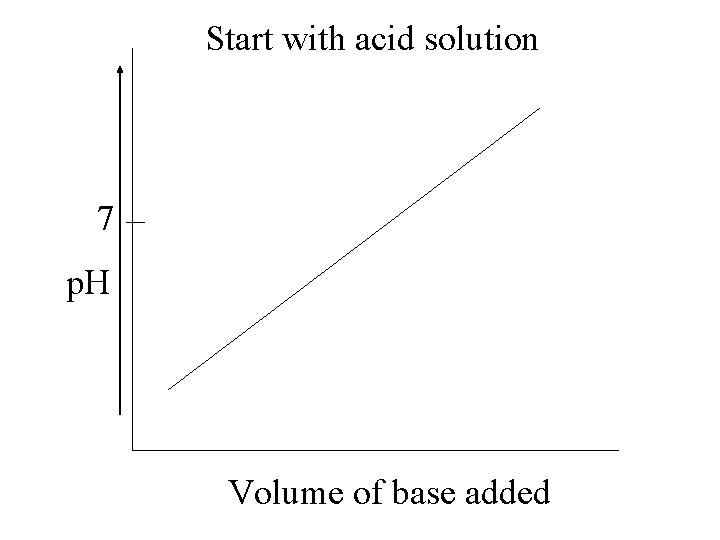
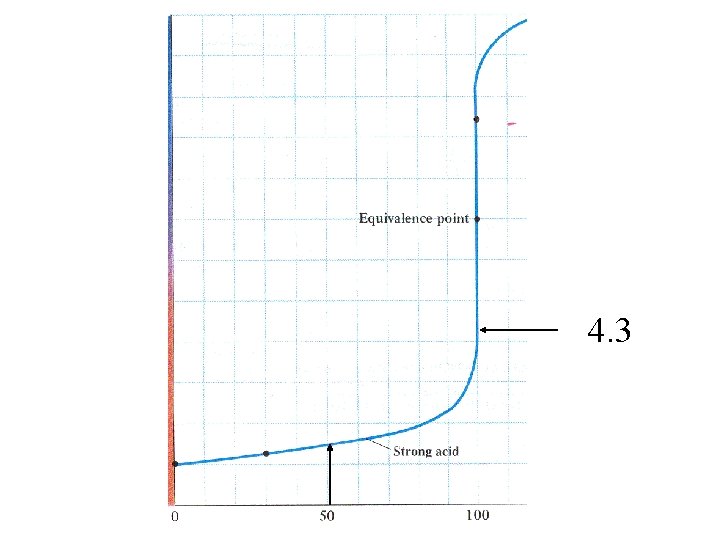
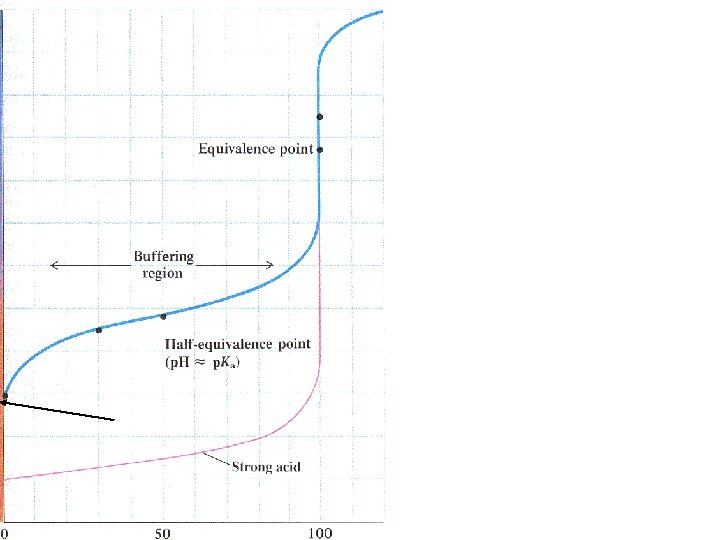
Start with acid solution 7 p. H Volume of base added
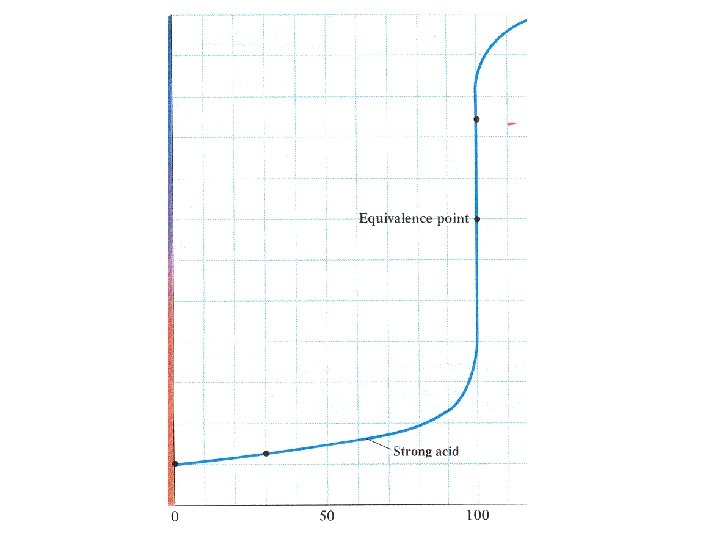
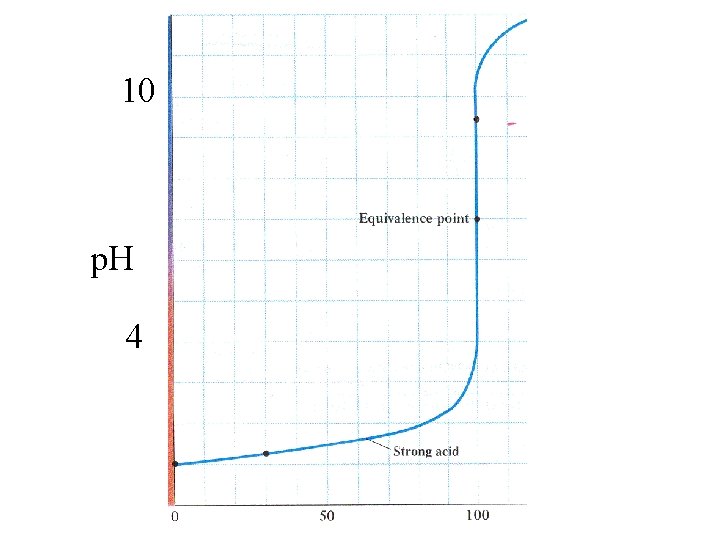
10 p. H 4


100 ml 0. 1 M HCl

100 ml 0. 1 M HCl Titrate with 0. 1 M Na. OH
100 ml 0. 1 M HCl Titrate with 0. 1 M Na. OH H 3 O+ + OH- 2 H 2 O
100 ml 0. 1 M HCl Titrate with 0. 1 M Na. OH H 3 O+ + OH- 2 H 2 O Every part of a mole of Na. OH added reduces the moles of H 3 O+ by an equal amount.

100 ml 0. 1 M HCl Titrate with 0. 1 M Na. OH H 3 O+ + OH- 2 H 2 O As Na. OH solution is added, the overall volume increases. This further decreases [H 3 O+].

100 ml 0. 1 M HCl Titrate with 0. 1 M Na. OH Neutralization and dilution; both increase the p. H of the solution.

100 ml 0. 1 M HCl Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution.

100 ml 0. 1 M HCl Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. Start with 0. 1 L x 0. 1 M H 3 O+

100 ml 0. 1 M HCl Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. Start with 0. 1 L x 0. 1 M H 3 O+ 0. 01 moles H 3 O+

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. Add 0. 05 L x 0. 1 M OH-

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. Add 0. 05 L x 0. 1 M OH 0. 005 moles OH-

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. 0. 005 moles OHH 3 O+ is neutralized by OH- 1: 1 ratio

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. 0. 005 moles OHRemaining H 3 O+ = 0. 01 moles – 0. 005 moles

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. 0. 005 moles OHRemaining H 3 O+ = 0. 005 moles Total volume = 150 ml = 0. 15 L

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. 0. 005 moles OH[H 3 O+]= 0. 005 moles 0. 15 L

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. 0. 005 moles OH[H 3 O+]= 0. 005 moles = 0. 033 M 0. 15 L

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 50 ml of Na. OH solution. 0. 005 moles OH[H 3 O+]= 0. 005 moles = 0. 033 M 0. 15 L p. H = 1. 48

100 ml 0. 1 M HCl Titrate with 0. 1 M Na. OH Add 99. 9 ml of Na. OH solution.

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 99. 9 ml of Na. OH solution.

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 99. 9 ml of Na. OH solution. 0. 0999 L x 0. 1 M = 0. 00999 moles OH-

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 99. 9 ml of Na. OH solution. 0. 0999 L x 0. 1 M = 0. 00999 moles OHMoles H 3 O+ = 0. 01 – 0. 00999 = 1 x 10 -5

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 99. 9 ml of Na. OH solution. 0. 0999 L x 0. 1 M = 0. 00999 moles OHMoles H 3 O+ = 0. 01 – 0. 00999 = 1 x 10 -5 Total volume = 0. 1 L + 0. 0999 L = 0. 1999 L

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 99. 9 ml of Na. OH solution. 0. 0999 L x 0. 1 M = 0. 00999 moles OH[H 3 O+]= 1 x 10 -5 moles 0. 1999 L

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 99. 9 ml of Na. OH solution. 0. 0999 L x 0. 1 M = 0. 00999 moles OH[H 3 O+]= 1 x 10 -5 moles = 5 x 10 -5 0. 1999 L

100 ml 0. 1 M HCl 0. 01 moles H 3 O+ Titrate with 0. 1 M Na. OH Add 99. 9 ml of Na. OH solution. 0. 0999 L x 0. 1 M = 0. 00999 moles OH[H 3 O+]= 1 x 10 -5 moles = 5 x 10 -5 0. 1999 L p. H = 4. 3
4. 3
+ 0. 1 ml will give p. H = 7 4. 3

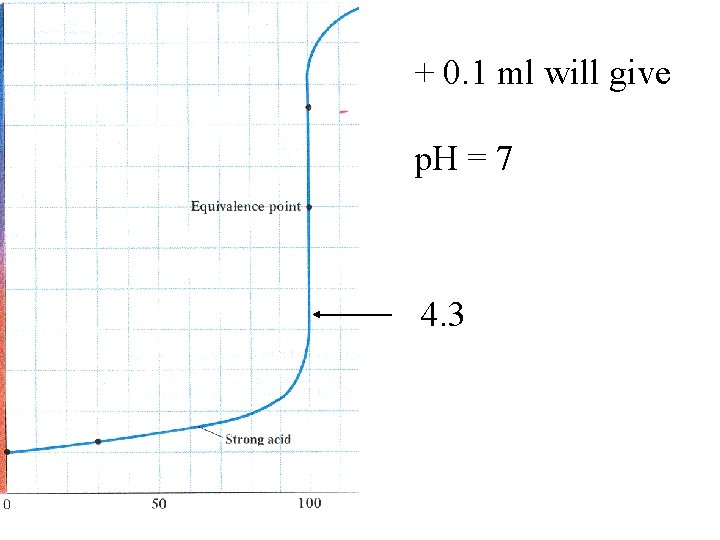
If more 0. 1 M Na. OH solution is added after the equivalence point, there is no H 3 O+ to neutralize it.

At p. H = 7 the volume is 0. 2 L

At p. H = 7 the volume is 0. 2 L Add 0. 1 m. L 0. 1 M Na. OH

At p. H = 7 the volume is 0. 2 L Add 0. 1 m. L 0. 1 M Na. OH Add 0. 0001 L x 0. 1 M = 10 -5 moles OH[OH-] = 1 x 10 -5 moles = 5 x 10 -5 M 0. 201 L

At p. H = 7 the volume is 0. 2 L Add 0. 1 m. L 0. 1 M Na. OH Add 0. 0001 L x 0. 1 M = 10 -5 moles OH[OH-] = 1 x 10 -5 moles = 5 x 10 -5 M 0. 2001 L p. OH = 4. 3 p. H = 9. 7
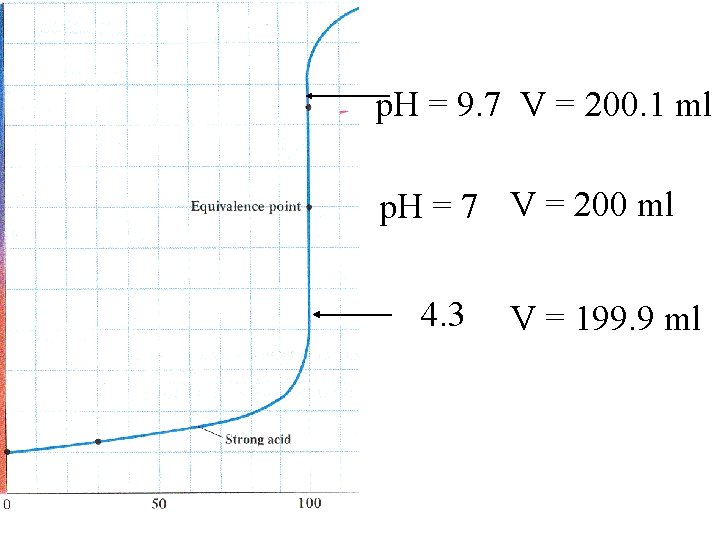
p. H = 9. 7 V = 200. 1 ml p. H = 7 V = 200 ml 4. 3 V = 199. 9 ml

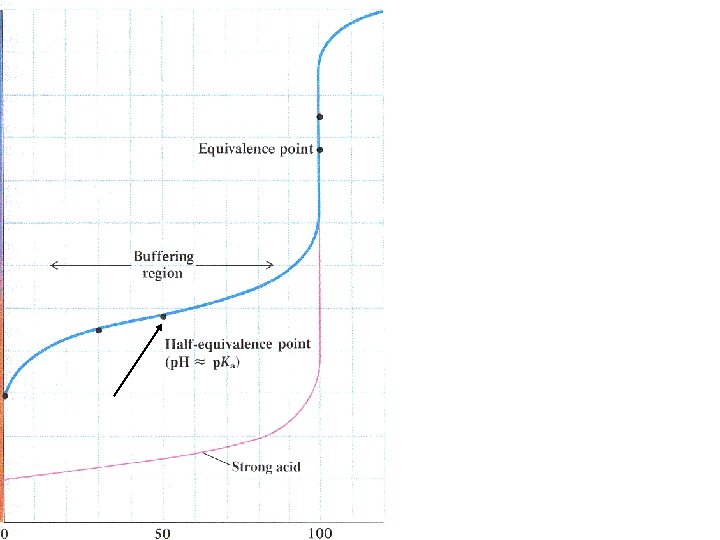
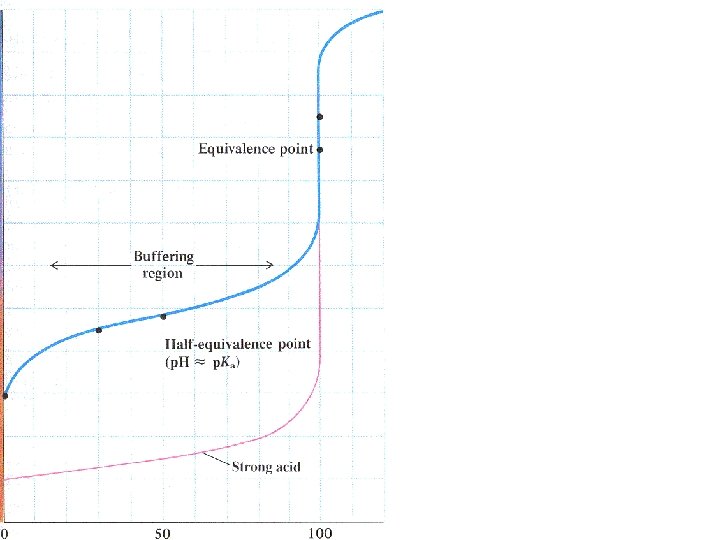
Titrating a weak acid with a strong base
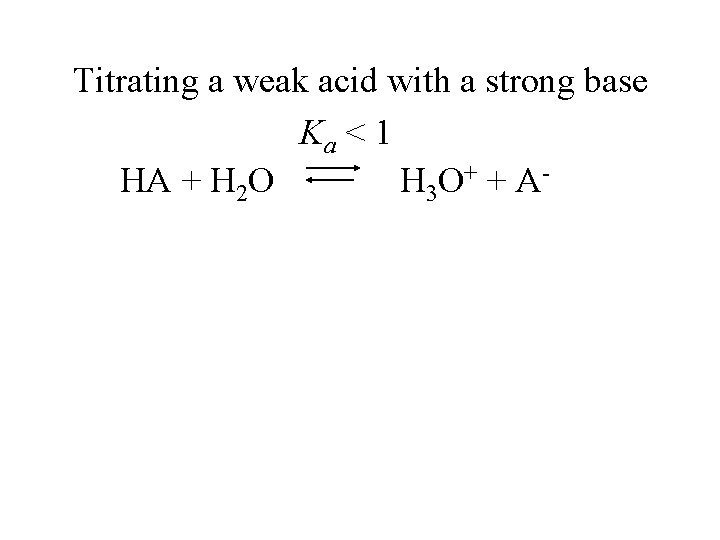
Titrating a weak acid with a strong base Ka < 1 HA + H 2 O H 3 O + + A -
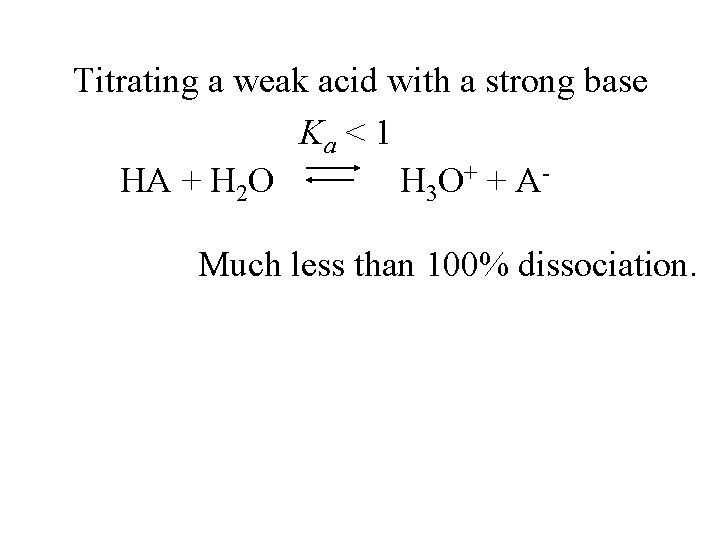
Titrating a weak acid with a strong base Ka < 1 HA + H 2 O H 3 O + + A Much less than 100% dissociation.

Titrating a weak acid with a strong base Ka < 1 HA + H 2 O H 3 O + + A Much less than 100% dissociation. Every OH- added neutralizes an H 3 O+ and shifts the equilibrium to the right.

Titrate 100 ml 0. 10 M CH 3 COOH With 0. 10 M Na. OH

Titrate 100 ml 0. 10 M CH 3 COOH Ka = 1. 8 x 10 -5 With 0. 10 M Na. OH
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-54.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] With 0. 10 M Na. OH 0 ml Na. OH
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-55.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] p. H = 2. 87 With 0. 10 M Na. OH 0 ml Na. OH
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-57.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH]. 1 L x 0. 1 M = 0. 01 moles CH 3 COOH With 0. 10 M Na. OH 50 ml Na. OH 0. 05 L x 0. 1 M = 0. 005 moles Na. OH
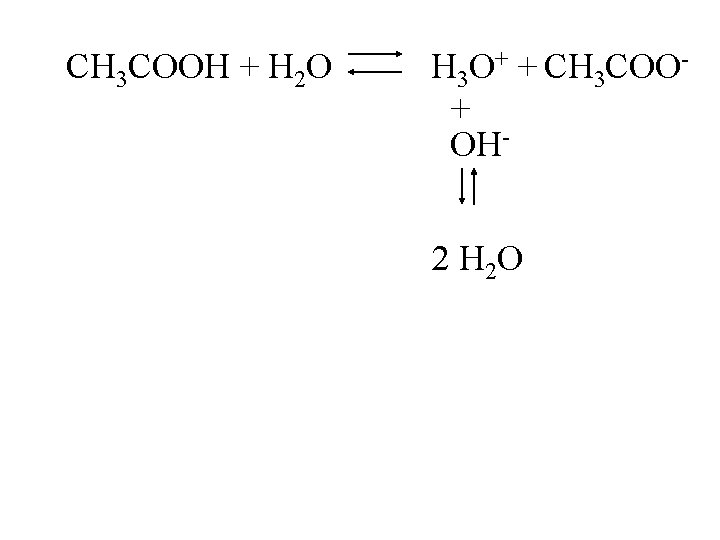
CH 3 COOH + H 2 O H 3 O+ + CH 3 COO+ OH 2 H 2 O
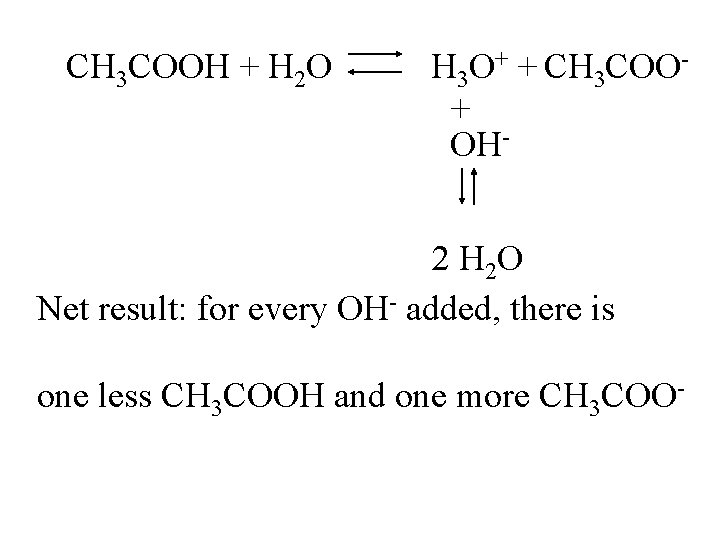
CH 3 COOH + H 2 O H 3 O+ + CH 3 COO+ OH- 2 H 2 O Net result: for every OH- added, there is one less CH 3 COOH and one more CH 3 COO-
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-60.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH]. 1 L x 0. 1 M = 0. 01 moles CH 3 COOH 0. 01 - 0. 005 moles CH 3 COOH = 0. 005 moles With 0. 10 M Na. OH 50 ml Na. OH 0. 05 L x 0. 1 M = 0. 005 moles Na. OH
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-61.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH]. 1 L x 0. 1 M = 0. 01 moles CH 3 COOH 0. 01 - 0. 005 moles CH 3 COOH = 0. 005 moles CH 3 COOWith 0. 10 M Na. OH 50 ml Na. OH 0. 05 L x 0. 1 M = 0. 005 moles Na. OH
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-62.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] 0. 01 - 0. 005 moles CH 3 COOH = 0. 005 moles CH 3 COO 0. 15 L solution With 0. 10 M Na. OH 50 ml Na. OH
![[CH 3 COOH] = 0. 005 moles = 0. 033 M 0. 15 L [CH 3 COOH] = 0. 005 moles = 0. 033 M 0. 15 L](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-63.jpg)
[CH 3 COOH] = 0. 005 moles = 0. 033 M 0. 15 L 0. 005 moles CH 3 COO- = 0. 033 M
![[CH 3 COOH] = 0. 005 moles = 0. 033 M 0. 15 L [CH 3 COOH] = 0. 005 moles = 0. 033 M 0. 15 L](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-64.jpg)
[CH 3 COOH] = 0. 005 moles = 0. 033 M 0. 15 L 0. 005 moles CH 3 COO- = 0. 033 M Ka = 1. 8 x 10 -5 = [H 3 O+][CH 3 COO-] [CH 3 COOH] Buffer solution: p. Ka = p. H = 4. 74
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-66.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] 0. 01 moles CH 3 COO 0. 20 L solution With 0. 10 M Na. OH 100 ml Na. OH
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-67.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] 0. 01 moles CH 3 COO[CH 3 COO-] = 0. 05 M 0. 20 L solution With 0. 10 M Na. OH 100 ml Na. OH
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-68.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] [CH 3 COO-] = 0. 05 M Kb = Kw Ka = 10 -14 = 5. 6 x 10 -10 1. 8 x 10 -5 [CH 3 COOH][OH-] Kb = [CH 3 COO-]
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-69.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] [CH 3 COO-] = 0. 05 M Kb = Kw Ka = 10 -14 = 5. 6 x 10 -10 1. 8 x 10 -5 [CH 3 COOH][OH-] [CH 3 COO-] y 2 = 0. 05
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-70.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] y 2 = (5. 6 x 10 -10)(0. 05) = 2. 8 x 10 -11 Kb = Kw Ka = 10 -14 = 5. 6 x 10 -10 1. 8 x 10 -5 [CH 3 COOH][OH-] [CH 3 COO-] y 2 = 0. 05
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-71.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] y 2 = (5. 6 x 10 -10)(0. 05) = 2. 8 x 10 -11 y = 5. 3 x 10 -6 Kw 10 -14 Kb = = = 5. 6 x 10 -10 Ka 1. 8 x 10 -5 2 y [CH 3 COOH][OH ] = Kb = 0. 05 [CH COO-] 3
![Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H](http://slidetodoc.com/presentation_image/d995c9651bb4e4cae44d273c05339619/image-72.jpg)
Titrate 100 ml 0. 10 M CH 3 COOH +][CH COO-] -5 = [H O Ka = 1. 8 x 10 3 3 [CH 3 COOH] y 2 = (5. 6 x 10 -10)(0. 05) = 2. 8 x 10 -11 y = 5. 3 x 10 -6 p. OH =5. 3; p. H = 8. 7 Kw 10 -14 Kb = = = 5. 6 x 10 -10 Ka 1. 8 x 10 -5 2 y [CH 3 COOH][OH ] = Kb = 0. 05 [CH COO-] 3

After the equivalence point, OH- is being added to a saturated buffer system.

After the equivalence point, OH- is being added to a saturated buffer system. p. H increases rapidly
- Slides: 75