Titrations AcidBase Part 2 AcidBase Titration u The

Titrations Acid-Base Part 2

Acid-Base Titration u. The process of adding a strong acid or a strong base to a titrate of acid or base with the purpose of neutralizing the substance in the beaker u Looking to reach equivalence point mol H+ = mol OH-

Acid-Base Titration done with a p. H probe, which is used to create a graph of p. H vs. volume added p. H u. Generally Volume added

Determination of the Equivalence Point u. Ways 1. to find the equivalence point Can find it stoichiometrically using stoichiometry for the neutralization reaction mol H+ = mol OH- stoichiometrically

Determination of the Equivalence Point u. Ways 2. to find the equivalence point Can find it graphically based on p. H vs. volume graph u u When the titrate is neutralized completely by the titrant, the p. H changes dramatically The midpoint of this dramatic change is the equivalence point – the middle of the area where neutralization is happening

Use of an Acid-Base Indicator • If the p. H at the equivalence point is known, an indicator can be used • • Want the p. Ka of the indicator to match the p. H at the equivalence point Indicator will then change color as p. H changes dramatically, giving visual end to the titration

Things to KNOW about Titrations v. Need to be able to EXPLAIN the shape of the curve at ALL points through the titration v What happens initially after a small amount of acid/base added v When excess of the titrant still remains v What happens near the equivalence point v What happens beyond the equivalence point – when excess of the strong acid or strong base used as a titrate is added

Things to CALCULATE about Titrations v Calculate v Normal v Calculate v Or the initial p. H of acid/base in the beaker acid/base calculation for strong or weak acid/base the p. H at the halfway point of the titration use p. H at halfway point to find Ka or Kb of substance titrated v Calculation of the p. H at the equivalence point v OR using the equivalence point to find the concentration of unknown acid/base you are titrating v Calculation of the p. H at a point beyond the equivalence point

Five Types of Titrations 1. Strong acid titrated with a strong base 2. Strong base titrated with a strong acid 3. Weak acid titrated with a strong base 4. Weak base titrated with a strong acid 5. Polyprotic acid titrated with a strong base

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find initial p. H

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find initial p. H = - log [H 1+] p. H = - log (0. 200 M) = 0. 699

Strong Acid titrated with Strong Base – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH p. H u. Example Starts at LOW p. H (strong acid in beaker) Volume added

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find p. H at halfway point

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find p. H at halfway point 0. 0500 L H 1+ x (0. 200 mol H 1+ / 1 L) x (1 mol OH 1 - / 1 mol H 1+) x (1 L / 0. 100 mol OH 1 -) = 0. 100 L OH 1 - (Needed to Reach Equivalence Point) = 0. 100 L OH 1 - / 2 = 0. 050 L OH 1 - (Needed for Halfway Point)

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find p. H at halfway point 0. 050 L OH 1 - x (0. 100 mol OH 1 - / 1 L) x (1 mol H 1+ / 1 mol OH 1 -) = 0. 00500 mol H 1+ 0. 010 mol H 1+ initially – 0. 00500 mol H 1+ consumed = 0. 00500 mol H 1+ remain 0. 00500 mol H 1+ / 0. 10 L total volume = 0. 050 M H 1+

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find p. H at halfway point 0. 050 M H 1+ p. H = - log [H 1+] = - log (0. 050 M H 1+) = 1. 3

Strong Acid titrated with Strong Base adding strong base, there is a very SMALL change as long as there is still excess acid. p. H u. Upon A very small change with excess strong acid still present Volume added

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find p. H at equivalence point

Strong Acid titrated with Strong Base the equivalence point is reached, there begins to be a dramatic jump in p. H as moles acid = moles base p. H u. As Dramatic change Volume added

Strong Acid titrated with Strong Base u. Even just beyond the equivalence point, the addition of excess base leads to a basic p. H Excess base now = jump to high p. H Midpoint = equivalence point = 7 Volume added

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find p. H after 150. 0 m. L of Na. OH has been added

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find p. H after 150. 0 m. L of Na. OH has been added 0. 150 L OH 1 - x (0. 100 mol OH 1 - / 1 L) x (1 mol H 1+ / 1 mol OH 1 -) = 0. 015 mol H 1+ Excess of 0. 00500 mol OH 1 - / 0. 2 L = 0. 025 M OH 1 -

Strong Acid titrated with Strong Base u. Example – you have 50. 0 m. L of 0. 200 M HNO 3 and look to titrate it with 0. 100 M Na. OH u Find p. H after 150. 0 m. L of Na. OH has been added 0. 025 M OH 1 p. OH = - log [OH 1 -] = - log (0. 025 M OH 1 -) = 1. 6 14 – p. OH = p. H = 14 – 1. 6 = 12. 4

Strong Acid titrated with Strong Base u. As more excess strong base is added, there is NOT a dramatic change in p. H Same excess base = no dramatic change in p. H Volume added

Strong Base titrated with Strong Acid u. OPPOSITE!!!!! u To find initial p. H – u Just u To based on [OH-] since it dissociates completely find p. H at halfway point – u Find moles of both, the acid added will be limiting u Moles of excess base / total Liters = M of [OH-] use to get p. H u To find p. H at equivalence point – u Since u To mol [OH-] = mol [H+] and no significant conjugates – p. H = 7 find p. H beyond equivalence point – u Find moles of excess acid added / total L = M of [H+] can get p. H
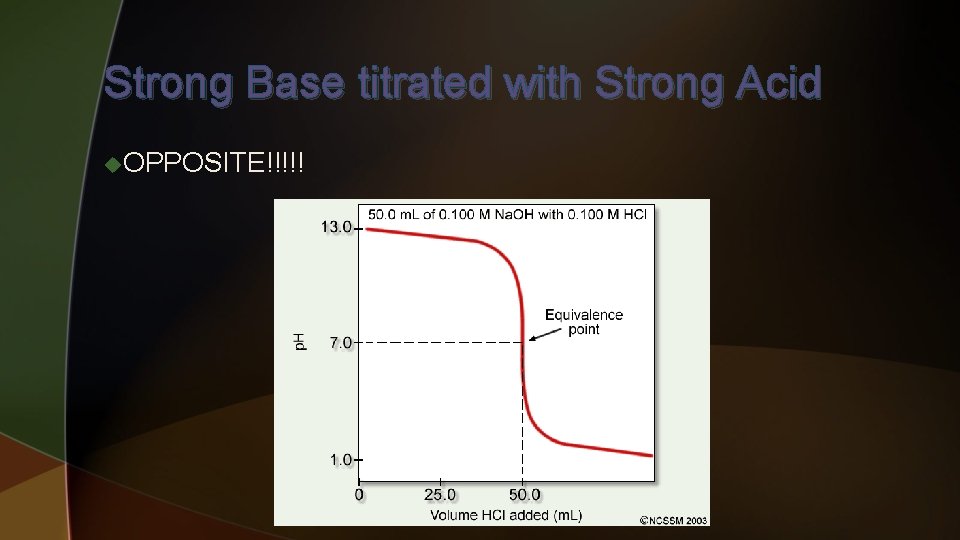
Strong Base titrated with Strong Acid u. OPPOSITE!!!!!

LAB u. Create u. If a titration curve molarity of original acid isn’t known, you can use the curve to find its molarity. HOW? ? ? That’s what you’re designing!!!

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) initial p. H

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) initial p. H HC 2 H 3 O 2 + H 2 O H 3 O 1+ + C 2 H 3 O 21 I 0. 1 M --0 0 C -x +x +x E 0. 1 M – x x x

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) initial p. H u u 1. 8 x 10 -5 / 0. 1 < 5% x 2 / 0. 1 = 1. 8 x 10 -5 X = [H 1+] = 0. 0013 M p. H = - log (0. 0013 M) = 2. 89

WEAK Acid titrated with Strong Base starts at a low p. H (though not quite as low) with weak acid in beaker p. H u. Still Starts at LOW p. H (weak acid in beaker) Volume added

WEAK Acid titrated with Strong Base u. The REACTION that occurs is now slightly different: HC 2 H 3 O 2 + OH- C 2 H 3 O 2 - + H 2 O Makes a GOOD conjugate base now that also influences the p. H and

WEAK Acid titrated with Strong Base u. The REACTION that occurs is now slightly different: HC 2 H 3 O 2 + OH- C 2 H 3 O 2 - + H 2 O With not all weak acid reacted and good conjugate base also formed, it creates a BUFFER

WEAK Acid titrated with Strong Base spike in p. H due to formation of the conjugate base. Then mostly levels off as with both weak acid and conjugate base you have a BUFFER. p. H u Initial Jump = conjugate base made, then buffer region Volume added

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) p. H at halfway point

HALFWAY POINT u. Ratio of weak acid to conjugate base is the same u Half up. H of weak acid used up, equal amount of conjugate base formed = p. Ka!!!

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) p. H at equivalence point

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) p. H at equivalence point C 2 H 3 O 21 - + H 2 O OH 1 - + HC 2 H 3 O 2 I 0. 050 M ---0 0 C -x +x +x E 0. 050 M – x x x

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) p. H at equivalence point kb = 5. 6 x 10 -10 / 0. 050 < 5%

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) p. H at equivalence point x 2 / 0. 050 = 5. 6 x 10 -10 x = 5. 3 x 10 -6 M = [OH 1 -] p. OH = - log (5. 3 x 10 -6) = 5. 3 p. H = 8. 7

WEAK Acid titrated with Strong Base the equivalence point is reached, there again is the point where the base neutralizes all of the acid and there is a significant jump in p. H. The p. H at the equivalence point is GREATER than 7 since the GOOD conjugate base remains. p. H u As Halfway point: p. H = p. Ka Dramatic jump up as equivalence point is reached again Volume added
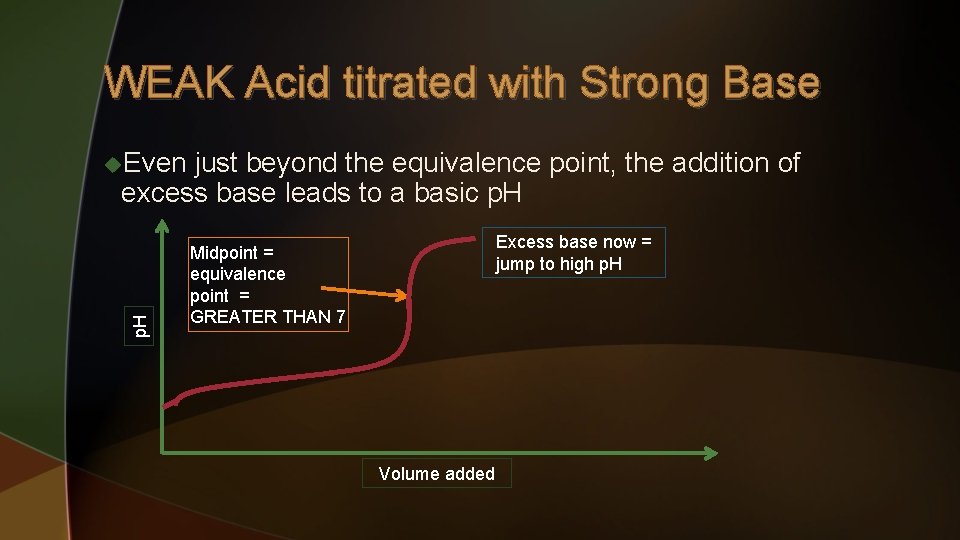
WEAK Acid titrated with Strong Base just beyond the equivalence point, the addition of excess base leads to a basic p. H u. Even Excess base now = jump to high p. H Midpoint = equivalence point = GREATER THAN 7 Volume added

WEAK Acid titrated with Strong Base u. As more excess strong base is added, there is NOT a dramatic change in p. H Same excess base = no dramatic change in p. H Volume added

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) p. H after 100. 0 m. L Na. OH added

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) p. H after 100. 0 m. L Na. OH added 50. 0 m. L of Na. OH is excess As Na. OH is a stronger base than acetate and is a strong base, the p. H will be solely based on the Na. OH concentration.

WEAK Acid titrated with Strong Base u. Example u Find – you have 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH (Ka of acetic acid = 1. 8 x 10 -5) p. H after 100. 0 m. L Na. OH added 0. 0500 L OH 1 - excess x 0. 100 M OH 1 - = 0. 00500 mol OH 10. 0050 mol OH 1 - / 0. 150 L total volume = 0. 033 M OH 1 p. OH = - log (0. 033 M) = 1. 48 p. H = 12. 52

WEAK Base titrated with Strong Acid u. OPPOSITE!!!!! u To find initial p. H – u To find p. H at halfway point – u To find p. H at equivalence point – u To find p. H after equivalence point -
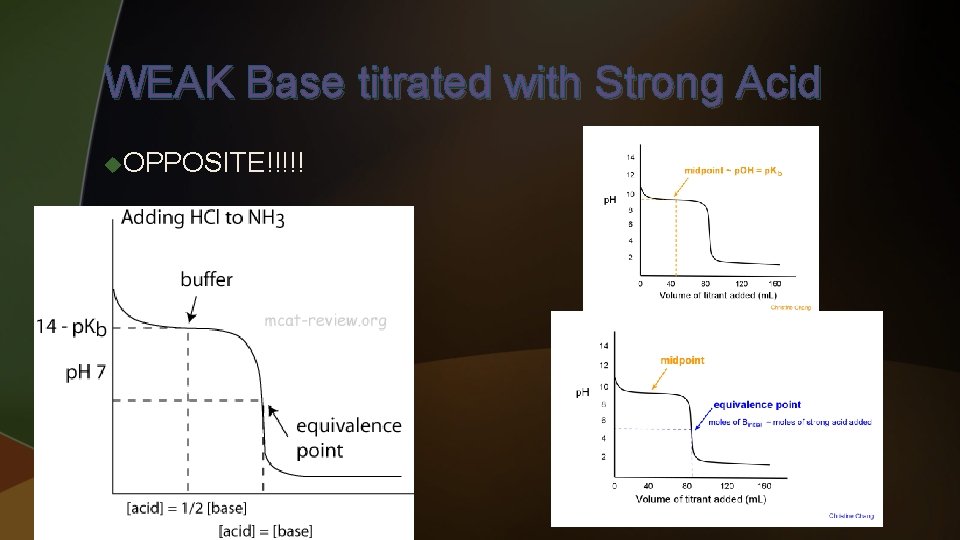
WEAK Base titrated with Strong Acid u. OPPOSITE!!!!!

LAB u. Create u. If a titration curve the identity and the Ka / Kb of a weak acid or a weak base are not known, you can determine it. HOW? ? ? That’s what you’re designing!!!

Polyprotic Acids u. Acids that have more than one H+ u That means there will be more than one equivalence point!! u Each H+ reacted must be neutralized with a stoichiometric amount of base u Example – H 2 CO 3 with Na. OH

LAB u. Titration u What of a polyprotic acid with a strong base. would this look like? u Why would it look that way?
- Slides: 51