Titrations 1 of 24 Boardworks Ltd 2012 2
Titrations 1 of 24 © Boardworks Ltd 2012

2 of 24 © Boardworks Ltd 2012

Titration is a quantitative technique that is used to accurately determine the concentration of a substance in solution. During a titration, a solution of known concentration, called a standard solution, is added to a solution of unknown concentration. The purpose of a titration is to determine the volume of solution required to reach an endpoint. An endpoint is an observable physical change, such as a colour change. Why is measuring volume useful? 3 of 24 © Boardworks Ltd 2012
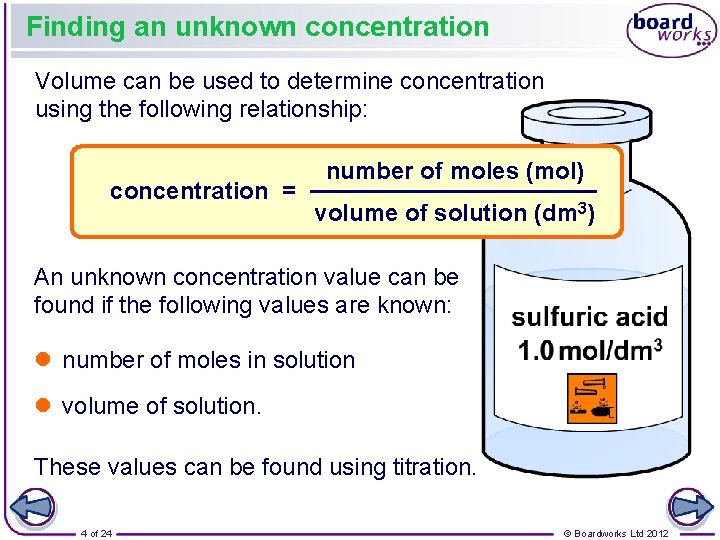
Finding an unknown concentration Volume can be used to determine concentration using the following relationship: concentration = number of moles (mol) volume of solution (dm 3) An unknown concentration value can be found if the following values are known: l number of moles in solution l volume of solution. These values can be found using titration. 4 of 24 © Boardworks Ltd 2012
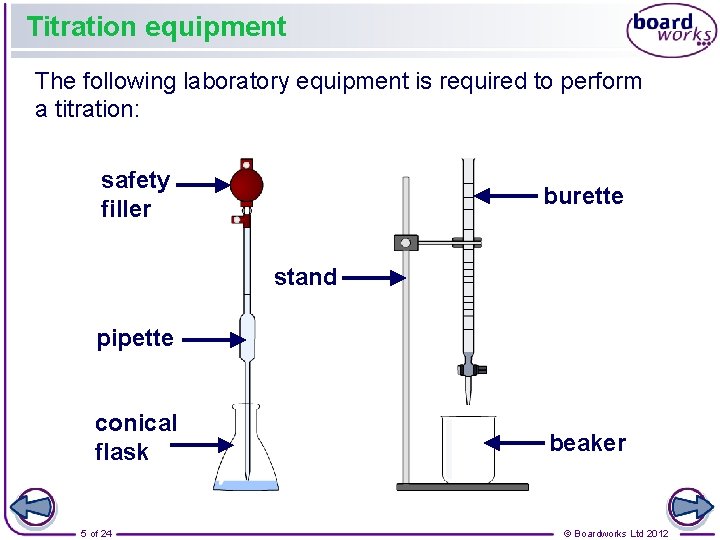
Titration equipment The following laboratory equipment is required to perform a titration: safety filler burette stand pipette conical flask 5 of 24 beaker © Boardworks Ltd 2012

Indicators The endpoint of a titration is often marked by a colour change. This is provided by an indicator solution. Indicators are substances which change colour according to the p. H of a solution. A small amount of indicator solution is added to one of the solutions during a titration. When the indicator changes colour, the endpoint of the titration has been reached. 6 of 24 © Boardworks Ltd 2012
Performing a titration 7 of 24 © Boardworks Ltd 2012
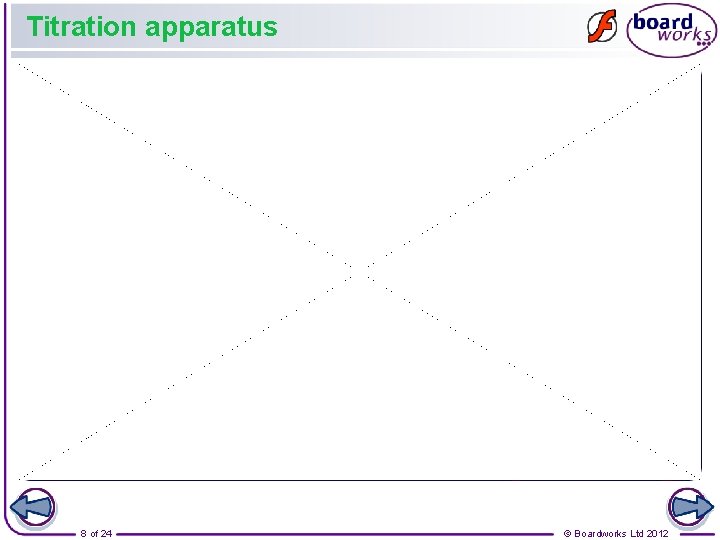
Titration apparatus 8 of 24 © Boardworks Ltd 2012

9 of 24 © Boardworks Ltd 2012

Using titration results How are the results of a titration used to calculate the unknown concentration of a solution? Step 1: Write a balanced equation for the reaction. Step 2: Calculate the number of moles of standard solution by rearranging the concentration formula: moles = concentration × volume Step 3: Use the balanced equation to determine the number of moles for the solution under investigation. Step 4: Use the concentration formula to determine the unknown concentration value: concentration = moles ÷ volume 10 of 24 © Boardworks Ltd 2012

Titration calculations – worked example The endpoint of a titration was reached when 20 cm 3 of 0. 1 mol/dm 3 sodium hydroxide was added to 25 cm 3 of hydrochloric acid. What is the concentration of the acid? 1. Write a balanced equation for the reaction: HCl + Na. OH Na. Cl + H 2 O 2. Calculate the number of moles of sodium hydroxide: moles = concentration (mol/dm 3) × volume (dm 3) = 0. 1 × (20. 00 ÷ 1000) = 0. 002 mol Na. OH 11 of 24 © Boardworks Ltd 2012

Titration calculations – worked example 3. The balanced equation from step 1 shows that one mole of HCl reacts with one mole of Na. OH: HCl + Na. OH Na. Cl + H 2 O Therefore 0. 002 moles of Na. OH will react with: 0. 002 moles of HCl. 4. Use the number of moles to calculate the concentration of the hydrochloric acid: moles 0. 002 = concentration = volume (dm 3) (25 ÷ 1000) = 0. 08 mol/dm 3 12 of 24 © Boardworks Ltd 2012
Titration calculations questions 13 of 24 © Boardworks Ltd 2012

14 of 24 © Boardworks Ltd 2012

The p. H scale shows the acidity or alkalinity of a solution. 1 2 3 4 5 6 7 8 9 10 11 12 13 14 strong acid weak acid neutral weak alkali strong alkali p. H is a measure of the concentration of hydrogen ions, H+, in solution. l The greater the concentration of H+ ions, the lower the p. H value and the stronger the acid. l The smaller the concentration of H+ ions, the larger the p. H value and the weaker the acid. 15 of 24 © Boardworks Ltd 2012

Neutralization reactions Hydrogen ions are released by acids in solution: HCl H+ + Cl. Hydrogen ions can be neutralized by the addition of an alkali. Alkalis release hydroxide ions, OH-, in solution: Na. OH Na+ + OHThe OH- ions from an alkali react with H+ ions from an acid to form water, which is p. H neutral. H+ + OH- H 2 O This is called a neutralization reaction. Neutralization reactions often form the basis of titrations. 16 of 24 © Boardworks Ltd 2012
p. H change during a titration 17 of 24 © Boardworks Ltd 2012
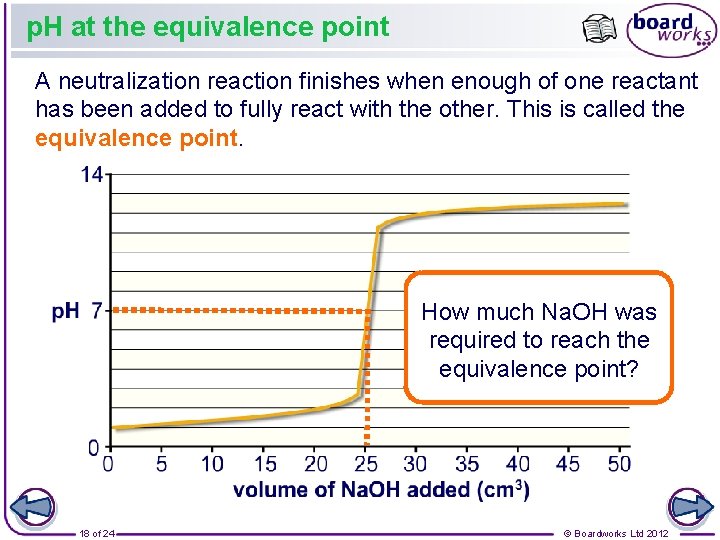
p. H at the equivalence point A neutralization reaction finishes when enough of one reactant has been added to fully react with the other. This is called the equivalence point. How much Na. OH was required to reach the equivalence point? 18 of 24 © Boardworks Ltd 2012
p. H ranges of indicators 19 of 24 © Boardworks Ltd 2012

Selecting an indicator When choosing an indicator for a titration, it is important to match its p. H range (the p. H values where it changes colour) to the equivalence point of the titration. l Phenolphthalein has a p. H range of 8– 9. l Methyl orange has a p. H range of 3– 4. l Litmus has a p. H range of 5– 8. l Bromothymol blue has a p. H range of 6– 7. Would litmus be a useful indicator for a titration? 20 of 24 © Boardworks Ltd 2012

21 of 24 © Boardworks Ltd 2012
Glossary 22 of 24 © Boardworks Ltd 2012
Anagrams 23 of 24 © Boardworks Ltd 2012
Multiple-choice quiz 24 of 24 © Boardworks Ltd 2012
- Slides: 24