Titration Prelab What is titration u Method to

Titration Pre-lab

What is titration? u. Method to determine the concentration of one solution based on its rxn with a known solution (titration standard) u. It determines the unknown molarity of a

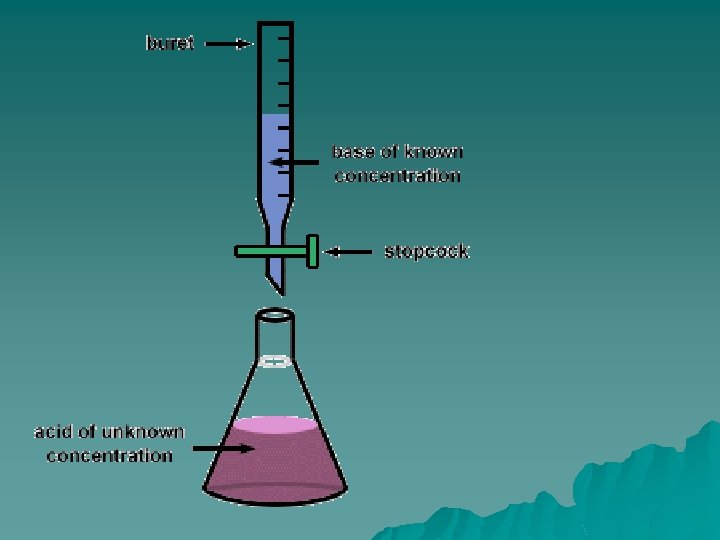
u. You The Process have the following – 50 ml Unknown solution (acid or base) –Known solution (acid or base) u. If you have an unknown acid you will need the known base and vice versa –An indicator (phenolphthalein or methyl orange) to show a

The process u. The lab process: –Pour 1/3 of the unknown (~16. 7 ml) into the Erlenmeyer flask. Be sure to record the exact amount in your data table –If you have an unknown acid put the flask underneath the base buret.

The Process –Add 3 drops of indicator u. Phenolphthalein is colorless in acids and pink in bases at about a p. H 8. 2 u. Methyl orange is red in acids and turns yellow at about 4. 4

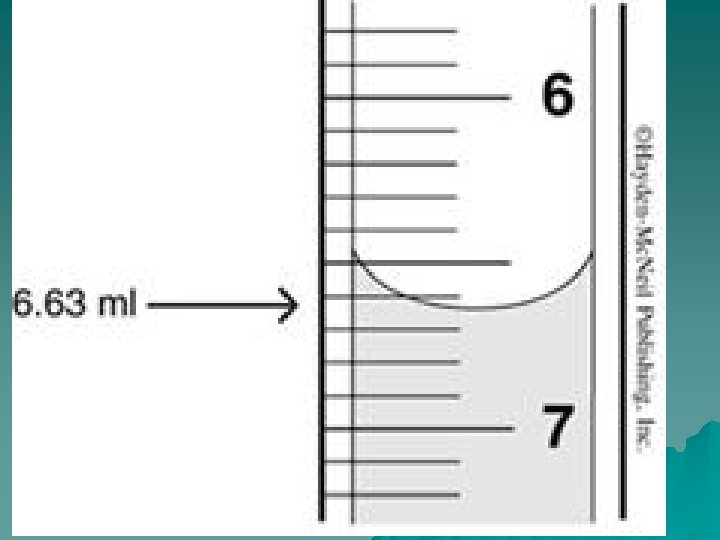
The Process u. Check your buret of the standard you are using. Be sure it is the opposite of the unknown, and that it is reasonably full. Be sure there are no air bubbles in the tip u. Take the initial reading of the buret and record it. Read to the nearest 100 th of a ml
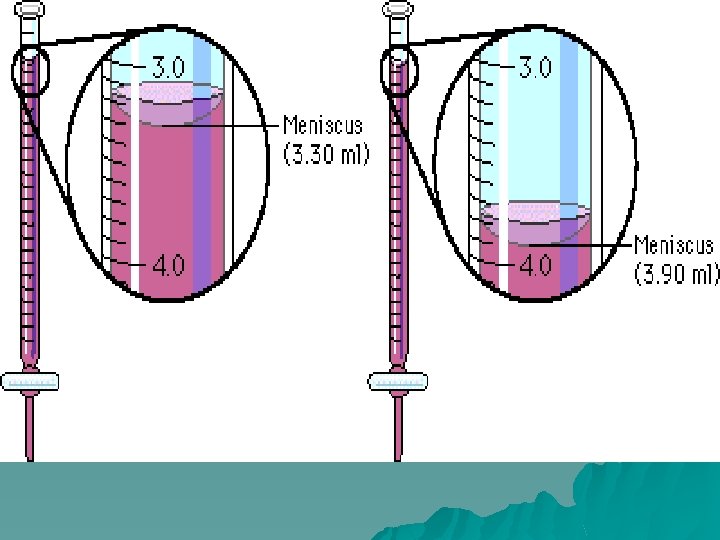

u. Have The Process 1 person constantly swirling the flask u. The other person should slowly add small amounts of standard solution u. When the unknown solution appears to be close to changing colors slow down and add 1 drop at a time until the color stays

The Process u. If the color of the end point is a deep color you added too much. The end point should be a light, barely a change in color. u. Record the final buret reading and the color in

The Process u. Rinse the flask with tap water thoroughly and repeat this 2 more times. u. Thoroughly clean and dry all equipment. Leave unused standard solution in the burets

The Calculations u. Calculate the molarity for each trial using M 1 V 1 = M 2 V 2 u. M 1 = molarity of acid u. V 1 = volume of acid u. M 2 = molarity of base u. V 2 = volume of base
- Slides: 13