Titration of a Weak Base with a Strong

Titration of a Weak Base with a Strong Acid The same principles applied above are also applicable where we have: 1. Before addition of any acid, we have a solution of the weak base and calculation of the p. H of the weak base should be performed as in previous sections. 2. After starting addition of the strong acid to the weak base, the salt of the weak base is formed. Therefore, a buffer solution results and you should consult previous lectures to find out how the p. H of buffers is calculated. 1

3. At the equivalence point, the amount of strong acid is exactly equivalent to the weak base and thus there will be 100% conversion of the weak base to its salt. The problem now is to calculate the p. H of the salt solution. 4. After the equivalence point, we would have a solution of the salt with excess strong acid. The presence of the excess acid suppresses the dissociation of the salt in water and the p. H of the solution controlled by the excess acid only. Now, let us apply the abovementioned concepts on an actual titration of a weak base with a strong acid. 2

Example Find the p. H of a 50 m. L solution of 0. 10 M NH 3 (kb = 1. 75 x 10 -5) after addition of 0, 10, 25, 50, 60 and 100 m. L of 0. 10 M HCl. Solution 1. After addition of 0 m. L HCl The solution is only 0. 10 M in ammonia, therefore we have: NH 3 + H 2 O D NH 4+ + OH- 3
![Kb = [NH 4+][OH-]/[NH 3] 1. 75*10 -5 = x * x / (0. Kb = [NH 4+][OH-]/[NH 3] 1. 75*10 -5 = x * x / (0.](http://slidetodoc.com/presentation_image_h/36aee54ac2113670f99f09b6202ace0c/image-4.jpg)
Kb = [NH 4+][OH-]/[NH 3] 1. 75*10 -5 = x * x / (0. 10 – x) kb is very small that we can assume that 0. 10>>x. We then have: 1. 75*10 -5 = x 2 / 0. 10 x = 1. 3 x 10 -3 M Relative error = (1. 3 x 10 -3 /0. 10) x 100 = 1. 3 % The assumption is valid, therefore: [OH-] = 1. 3 x 10 -3 M p. OH = 2. 88 p. H = 11. 12 4

2. After addition of 10 m. L HCl A buffer will be formed from the base and its salt Initial mmol NH 3 = 0. 10 x 50 = 5. 0 mmol HCl added = 0. 10 x 10 = 1. 0 mmol NH 3 left = 5. 0 – 1. 0 = 4. 0 [NH 3] = 4. 0/60 M mmol NH 4+ formed = 1. 0 [NH 4+] = 1. 0/60 M NH 3 + H 2 O D NH 4+ + OH- 5
![Kb = [NH 4+][OH-]/[NH 3] 1. 75*10 -5 = (1. 0/60 + x) * Kb = [NH 4+][OH-]/[NH 3] 1. 75*10 -5 = (1. 0/60 + x) *](http://slidetodoc.com/presentation_image_h/36aee54ac2113670f99f09b6202ace0c/image-6.jpg)
Kb = [NH 4+][OH-]/[NH 3] 1. 75*10 -5 = (1. 0/60 + x) * x / (4. 0/60 – x) kb is very small that we can assume that 1. 0/60 >>x. We then have: 1. 75*10 -5 = 1. 0/60 x / 4. 0/60 x = 7. 0 x 10 -5 Relative error = (7. 0 x 10 -5 /1. 0/60) x 100 = 0. 42 % The assumption is valid, therefore: [OH-] = 7. 0 x 10 -5 M p. OH = 4. 15 p. H = 9. 85 6

3. After addition of 25 m. L HCl A buffer will be formed from the base and its salt Initial mmol NH 3 = 0. 10 x 50 = 5. 0 mmol HCl added = 0. 10 x 25 = 2. 5 mmol NH 3 left = 5. 0 – 2. 5 = 2. 5, [NH 3] = 2. 5/75 M mmol NH 4+ formed = 2. 5, [NH 4+] = 2. 5/75 M NH 3 + H 2 O D NH 4+ + OH- 7
![Kb = [NH 4+][OH-]/[NH 3] 1. 75*10 -5 = (2. 5/75 + x) * Kb = [NH 4+][OH-]/[NH 3] 1. 75*10 -5 = (2. 5/75 + x) *](http://slidetodoc.com/presentation_image_h/36aee54ac2113670f99f09b6202ace0c/image-8.jpg)
Kb = [NH 4+][OH-]/[NH 3] 1. 75*10 -5 = (2. 5/75 + x) * x / (2. 5/75 – x) kb is very small that we can assume that 2. 5/75 >>x. We then have: 1. 75*10 -5 = 2. 5/75 x / 2. 5/75 x = 1. 75 x 10 -5 Relative error = (1. 75 x 10 -5 /2. 5/75) x 100 = 0. 052 % The assumption is valid, therefore: [OH-] = 1. 75 x 10 -5 M p. OH = 4. 76 p. H = 9. 24 8
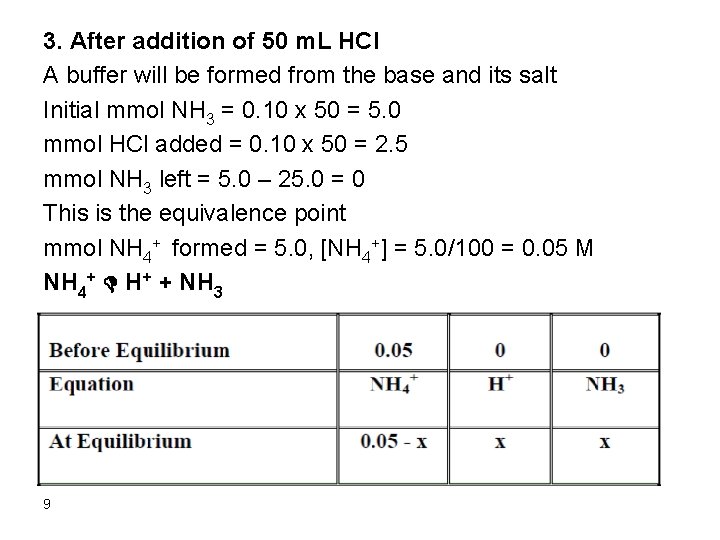
3. After addition of 50 m. L HCl A buffer will be formed from the base and its salt Initial mmol NH 3 = 0. 10 x 50 = 5. 0 mmol HCl added = 0. 10 x 50 = 2. 5 mmol NH 3 left = 5. 0 – 25. 0 = 0 This is the equivalence point mmol NH 4+ formed = 5. 0, [NH 4+] = 5. 0/100 = 0. 05 M NH 4+ D H+ + NH 3 9

Ka = 10 -14/1. 75 x 10 -5 = 5. 7 x 10 -10 Ka = [H+][NH 3]/[NH 4+] Ka = x * x / (0. 05 – x) Ka is very small. Assume 0. 05 >> x 5. 7*10 -10 = x 2/0. 05 x = 5. 33 x 10 -6 Relative error = (5. 33 x 10 -6/0. 05) x 100 = 0. 011 % The assumption is valid and the [H+] = 5. 33 x 10 -6 M p. H = 5. 27 10

5. After addition of 60 m. L HCl Initial mmol of NH 3 = 0. 10 x 50 = 5. 0 Mmol HCl added = 0. 10 x 60 = 6. 0 Mmol HCl excess = 6. 0 – 5. 0 = 1. 0 [H+] = 1. 0/110 M p. H = 2. 04 6. After addition of 100 m. L HCl Initial mmol of NH 3 = 0. 10 x 50 = 5. 0 Mmol HCl added = 0. 10 x 100 = 10. 0 Mmol HCl excess = 10. 0 – 5. 0 = 5. 0 [H+] = 5. 0/150 M p. H = 1. 48 11

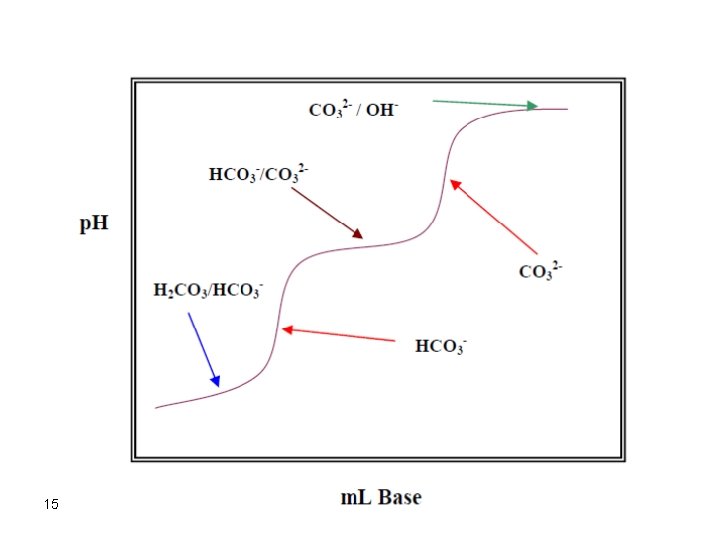
Titration of a Polyprotic Acid with a Strong Base Each proton in a polyprotic acid is supposed to titrate separately. However, only those protons which satisfy the empirical relation ka 1 > 104 ka 2 can result in an observable break at the point of equivalence. For example, carbonic acid shows two breaks in the titration curve. Each one corresponds to a specific proton of the acid. The method of calculation of the p. H is similar to that described above but initially for the first proton the second. Each equivalence point requires a separate indicator to visualize the end point. 12

There are few points to put in mind when dealing with problems of titration of polyprotic acids with strong bases: 1. Before addition of any base, you only have the polyprotic acid solution and thus calculation of the p. H is straightforward as previously described. 2. When we start addition of base, the first proton is titrated and bicarbonate will form. A buffer solution of carbonic acid and carbonate is formed and you should refer to the section on such calculations. 3. When all the first proton is titrated, all carbonic acid is now converted to bicarbonate (an amphoteric protonated salt) and calculation of the p. H is achieved using the appropriate root mean square equation. 13

4. Further addition of base starts titrating the second proton thus some bicarbonate is converted to carbonate and a buffer is formed. Calculate the p. H of the resulting buffer in the same way as in step 2. 5. When enough base is added so that the titration of the second proton is complete, all bicarbonate is converted to carbonate and this is the second equivalence point. The p. H is calculated for carbonate (unprotonated salt). 6. Addition of excess base will make the solution basic where this will suppress the dissociation of carbonate. The hydrogen ion concentration is calculated from the concentration of excess hydroxide. 14
15

Example Find the p. H of a 50 m. L solution of a 0. 10 M H 2 CO 3 after addition of 0, 25, 50, 75, 100, and 150 m. L of 0. 10 M Na. OH. Ka 1=4. 3 x 10 -7 and ka 2 = 4. 8 x 10 -11. Solution After addition of 0 m. L Na. OH We only have the carbonic acid solution and the p. H calculation for such types of solution was discussed earlier and can be worked as below: H 2 CO 3 D H+ + HCO 3 ka 1 = 4. 3 x 10 -7 HCO 3 - D H+ + CO 32 ka 2 = 4. 8 x 10 -11 16

Since ka 1 is much greater than ka 2, we can neglect the H+ from the second step and therefore we have: H 2 CO 3 D H+ + HCO 3 ka 1 = 4. 3 x 10 -7 Ka 1 = x * x/(0. 10 – x) Assume 0. 10>>x since ka 1 is small 4. 3*10 -7= x 2/0. 10, x = 2. 1 x 10 -4 Relative error = (2. 1 x 10 -4/0. 10) x 100 = 0. 21% The assumption is valid and [H+] = 2. 1 x 10 -4 M, p. H = 3. 68 17
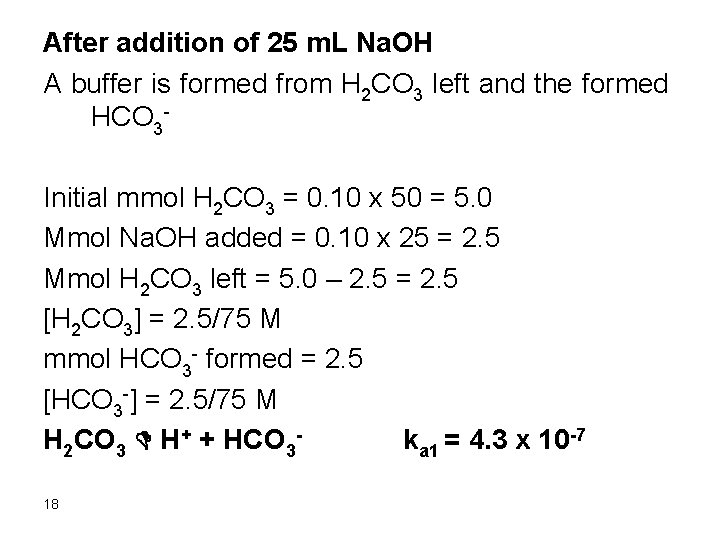
After addition of 25 m. L Na. OH A buffer is formed from H 2 CO 3 left and the formed HCO 3 Initial mmol H 2 CO 3 = 0. 10 x 50 = 5. 0 Mmol Na. OH added = 0. 10 x 25 = 2. 5 Mmol H 2 CO 3 left = 5. 0 – 2. 5 = 2. 5 [H 2 CO 3] = 2. 5/75 M mmol HCO 3 - formed = 2. 5 [HCO 3 -] = 2. 5/75 M H 2 CO 3 D H+ + HCO 3 ka 1 = 4. 3 x 10 -7 18

ka 1 = x(2. 5/75 + x)/(2. 5/75 – x) ka 1 is very small and in presence of the common ion the dissociation will be further suppressed. Therefore, assume 2. 5/75>>x. x = 4. 3 x 10 -7 M Relative error = {4. 3 x 10 -7/(2. 5/75)} x 100 = 0. 0013% The assumption is valid [H+] = 4. 3 x 10 -7 M p. H = 6. 37 19

After addition of 50 m. L Na. OH Initial mmol H 2 CO 3 = 0. 10 x 50 = 5. 0 mmol Na. OH added = 0. 10 x 50 = 5. 0 mmol H 2 CO 3 left = 5. 0 – 5. 0 = 0 This is the first equivalence point mmol HCO 3 - formed = 5. 0 [HCO 3 -] = 5. 0/100 = 0. 05 M Now the solution contains only the protonated salt. Calculation of the p. H can be done using the relation [H+] = {(ka 1 kw + ka 1 ka 2[HCO 3 -])/(ka 1 + [HCO 3‑]}1/2 [H+] = {(4. 3 x 10 -7 * 10 -14 + 4. 3 x 10 -7 * 4. 8 x 10 -11 * 0. 0. 05)/(4. 3 x 10 -7 + 0. 0. 05)}1/2 [H+] = 4. 5 x 10 -9 M p. H = 8. 34 20

After addition of 75 m. L Na. OH Here you should remember that 50 m. L of the Na. OH will be used in the titration of the first proton. Therefore, it is as if we add 25 m. L to the HCO 3 solution. We then have: Initial mmol HCO 3 - = 5. 0 Mmol Na. OH added = 0. 10 x 25 = 2. 5 Mmol HCO 3 - left = 5. 0 – 2. 5 = 2. 5 [HCO 3 -] = 2. 5/125 M mmol CO 32 - formed = 2. 5 [CO 32 -] = 2. 5/125 M Once again we have a buffer solution from HCO 3 - and CO 32 -. The p. H is calculated as follows: HCO 3 - D H+ + CO 32 ka 2 = 4. 8 x 10 -11 21

ka 1 = x(2. 5/125 + x)/(2. 5/125 – x) ka 1 is very small and in presence of the common ion the dissociation will be further suppressed. Therefore, assume 2. 5/125>>x. x = 4. 8 x 10 -11 M Relative error = {4. 8 x 10 -11/(2. 5/125)} x 100 = V. small The assumption is valid, [H+] = 4. 8 x 10 -11 M p. H = 10. 32 22

After addition of 100 m. L Na. OH At this point, all carbonic acid was converted into carbonate. The first 50 m. L of Na. OH were consumed in converting H 2 CO 3 to HCO 3 -. Therefore, as if we add 50 m. L to HCO 3 - solution and we have: Initial mmol HCO 3 - = 5. 0 mmol Na. OH added = 0. 10 x 50 = 5. 0 mmol HCO 3 - left = 5. 0 – 5. 0 = ? ? This is the second equivalence point mmol CO 32 - formed = 5. 0 [CO 32 -] = 5. 0/150 M 23

CO 32 - + H 2 O = HCO 3 - + OHKb = kw/ka 2 We used ka 2 since it is the equilibrium constant describing relation between CO 32 - and HCO 3 -. However, in any equilibrium involving salts look at the highest charge on any anion to find which ka to use. Kb = 10 -14/4. 8 x 10 -13 = 2. 1 x 10 -4 24

Kb = x * x/(5. 0/150 – x) Assume 5. 0/150 >> x 2. 1 x 10 -4 = x 2/(5. 0/150) x = 2. 6 x 10 -3 Relative error = (2. 6 x 10 -3 /(5. 0/150)) x 100 = 7. 9% Therefore, assumption is invalid and we have to use the quadratic equation. However, I’ll accept the answer this time. Therefore, [OH-] = 2. 6 x 10 -3 M p. OH = 2. 58 p. H = 14 – 2. 58 = 11. 42 25

After addition of 150 m. L Na. OH At this point, all carbonic acid was converted into carbonate requiring 100 m. L Na. OH. mmol Na. OH excess = 0. 1 x 50 = 5. 0 [OH-] = 5. 0/200 p. OH = 1. 60 p. H = 14. 00 – 1. 60 = 12. 40 26

Titration of a Polybasic base with a Strong Acid Example Find the p. H of a 50 m. L solution of a 0. 10 M Na 3 PO 4 (ka 1 = 1. 1 x 10 -2, ka 2 = 7. 5 x 10 -8, ka 3 = 4. 8 x 10 -13) after addition of 0, 25, 50, 75, 100, 125, 150, and 175 m. L of 0. 10 M HCl. Solution 1. After addition of 0 m. L HCl At this point, we only have the solution of PO 43 - (an unprotonated salt) and we can find the p. H as follows 27

PO 43 - + H 2 O D HPO 42 - + OH- kb= kw/ka 3 We used ka 3 since it is the equilibrium constant describing relation between PO 43 - and HPO 42 -. However, in any equilibrium involving salts look at the highest charge on any anion to find which ka to use. Kb = 10 -14/4. 8 x 10 -13 = 0. 020 28

Kb = x * x/0. 10 – x Assume 0. 10 >> x 0. 02 = x 2/0. 10 x = 0. 045 Relative error = (0. 045/0. 10) x 100 = 45% Therefore, assumption is invalid and we have to use the quadratic equation. If we solve the quadratic equation we get: X = 0. 036 Therefore, [OH-] = 0. 036 M p. OH = 1. 44 p. H = 14 – 1. 44 = 12. 56 29

2. After addition of 25 m. L HCl A buffer starts forming from phosphate remaining and the hydrogen phosphate produced from the reaction. PO 43 - + H+ D HPO 42 Initial mmol PO 43 - = 0. 10 x 50 = 5. 0 Mmol H+ added = 0. 10 x 25 = 2. 5 Mmol PO 43 - left = 5. 0 – 2. 5 = 2. 5 [PO 43 -] = 2. 5/75 M mmol HPO 42 - formed = 2. 5 [HPO 42 -] = 2. 5/75 M Now we look at any dissociation equilibrium equation containing both species. This can be obtained from the relation from ka 3, for example 30

HPO 42 - D PO 43 - + H+ Ka 3 = x(2. 5/75 + x)/(2. 5/75 – x) Since ka 3 is very small, assume 2. 5/75 >> x 4. 8 x 10 -13 = x(2. 5/75)/(2. 5/75) x = 4. 8 x 10 -13 It is clear that the relative error will be exceedingly small and the assumption is, for sure, valid. [H+] = 4. 8 x 10 -13 M p. H = 12. 32 31

3. After addition of 50 m. L HCl At this point, all PO 43 - will be converted to HPO 42 Initial mmol PO 43 - = 0. 10 x 50 = 5. 0 mmol H+ added = 0. 10 x 50 = 5. 0 mmol PO 43 - left = 5. 0 – 5. 0 = ? ? This is the first equivalence point mmol HPO 42 - formed = 5. 0 [HPO 42 -] = 5. 0/100 = 0. 05 M This is a protonated salt with two charges where we should use ka 2 and ka 3, i. e. the relation [H+] = {(ka 2 kw + ka 2 ka 3[HPO 42 -])/(ka 2 + [HPO 42‑]}1/2 [H+] = 2. 3 x 10 -10 p. H = 9. 65 32

4. After addition of 75 m. L HCl A second buffer is formed where we have HPO 42 - + H+ D H 2 PO 4 You should understand that 50 m. L were consumed in the conversion of PO 43 - to HPO 42 -, thus 25 m. L only were added to HPO 42 Initial mmol HPO 4 - = 0. 10 x 50 = 5. 0 mmol H+ added = 0. 10 x 25 = 2. 5 mmol HPO 42 - left = 5. 0 – 2. 5 = 2. 5 [HPO 42 -] = 2. 5/125 mmol H 2 PO 42 - formed = 2. 5 [H 2 PO 4 -] = 2. 5/125 33

H 2 PO 4 - D H+ + HPO 42 Before Equilibrium 2. 5/125 0 Equation H 2 PO 42 - H+ 2. 5/125 – x 2. 5/125 + x x At Equilibrium Ka 2 = x(2. 5/125 + x)/(2. 5/125 – x) Since ka 3 is very small, assume 2. 5/125 >> x 7. 5 x 10 -8 = x(2. 5/125)/(2. 5/125) x = 7. 5 x 10 -8 It is clear that the relative error will be exceedingly small and the assumption is, for sure, valid [H+] = 7. 5 x 10 -8 M p. H = 7. 12 34

5. After addition of 100 m. L HCl 50 m. L of HCl were consumed in converting PO 43 - into HPO 42 Initial mmol HPO 42 - = 0. 10 x 50 = 5. 0 mmol H+ added = 0. 10 x 50 = 5. 0 mmol HPO 42 - left = 5. 0 – 5. 0 = 0 This is the second equivalence point [H 2 PO 4 -] = 5. 0/150 = 0. 033 M At this point, all HPO 42 - will be completely converted into H 2 PO 4 - which is a protonated salt where the p. H can be calculated from the relation [H+] = {(ka 1 kw + ka 1 ka 2[H 2 PO 4 -])/(ka 1 + [H 2 PO 4‑]}1/2 [H+] = 2. 5 x 10 -5 M p. H = 4. 6 35

6. After addition of 125 m. L HCl H 2 PO 4 - + H+ D H 3 PO 4 50 m. L were consumed in converting PO 43 - to HPO 42 and 50 m. L were consumed in converting HPO 42 - into H 2 PO 4 -, therefore as if we add 25 m. L to H 2 PO 4 Initial mmol H 2 PO 4 - = 0. 10 x 50 = 5. 0 Mmol H+ added = 0. 10 x 25 = 2. 5 Mmol HPO 42 - left = 5. 0 – 2. 5 = 2. 5 [HPO 42 -] = 2. 5/175 M mmol H 3 PO 4 formed = 2. 5 [H 3 PO 4] = 2. 5/175 M This is a buffer formed from the acid and its conjugate base. The best way to calculate the p. H is to use the ka 1 expression where: 36

H 3 PO 4 D H+ + H 2 PO 4 Before Equilibriu m 2. 5/175 0 Equation H 3 PO 4 H 2 PO 4 - H+ At 2. 5/175 – 2. 5/175 x Ka 1 =Equilibriu x(2. 5/175 + xx)/(2. 5/175 – x) +x Since m ka 1 is very small (!!!), assume 2. 5/175 >> x 1. 1 x 10 -2 = x(2. 5/175)/(2. 5/175) x = 1. 1 x 10 -2 M Relative error = {1. 1 x 10 -2/(2. 5/175)} x 100 = 77% It is clear that the relative error is very large and the assumption is, for sure, invalid and we should use the quadratic equation. [H+] = 5. 2 x 10 -3 M p. H = 2. 29 37

7. After addition of 150 m. L HCl At this point, all PO 43 - is converted into the acid Initial mmol H 2 PO 4 - = 0. 10 x 50 = 5. 0 Mmol H+ added = 0. 10 x 50 = 5. 0 Mmol HPO 42 - left = 5. 0 – 5. 0 = 0 This is the third equivalence point mmol H 3 PO 4 formed = 5. 0 [H 3 PO 4] = 5/200 = 0. 025 38

Therefore, we only have the acid in solution and calculation of the p. H is done as follows: H 3 PO 4 D H+ + H 2 PO 4 ka 1 = 1. 1 x 10 -2 H 2 PO 4 - D H+ + HPO 42 ka 2 = 7. 5 x 10 -8 HPO 42 - D H+ + PO 43 ka 3 = 4. 8 x 10 -13 Since ka 1 >> ka 2 (ka 1/ka 2 > 104) the amount of H+ from the second and consecutive equilibria is negligible if compared to that coming from the first equilibrium. Therefore, we can say that we only have: 39

H 3 PO 4 D H+ + H 2 PO 4 - ka 1 = 1. 1 x 10 -2 Ka 1 = x * x/(0. 025 – x) Assume 0. 025>>x since ka 1 is small (!!!) 1. 1*10 -2 = x 2/0. 025 x = 0. 017 Relative error = (0. 017/0. 025) x 100 = 68% The assumption is invalid according to the criteria we set at 5% and thus we have to use the quadratic equation. 40
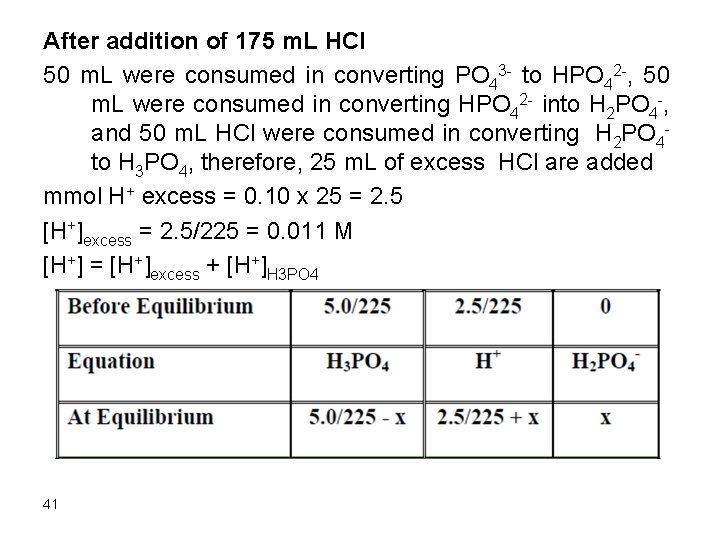
After addition of 175 m. L HCl 50 m. L were consumed in converting PO 43 - to HPO 42 -, 50 m. L were consumed in converting HPO 42 - into H 2 PO 4 -, and 50 m. L HCl were consumed in converting H 2 PO 4 to H 3 PO 4, therefore, 25 m. L of excess HCl are added mmol H+ excess = 0. 10 x 25 = 2. 5 [H+]excess = 2. 5/225 = 0. 011 M [H+] = [H+]excess + [H+]H 3 PO 4 41

Since at this point both H+ from excess HCl and phosphoric acid contribute to the overall [H+] We can calculate the [H+] from ka 1: Ka 1 = x(2. 5/225 + x)/(5. 0/225 – x) Since ka 1 is very small (!!!), assume 2. 5/225 >> x 1. 1 x 10 -2 = x(2. 5/225)/(5. 0/225) x = 2. 2 x 10 -2 M Relative error = {2. 2 x 10 -2/(2. 5/225)} x 100 = 198% It is clear that the relative error is very large and the assumption is, for sure, invalid and we should use the quadratic equation. 42
- Slides: 42