Titanium Group 4 B Elements Titanium was discovered

Titanium

Group 4 B Elements

Titanium was discovered combined in a mineral in Cornwall, England in 1791 by amateur geologist and pastor William Gregor. • Titanium when found in the nature is always bonded to other minerals. • It is the 9 th most abundant element in the earths crust at 0. 63% by mass. • It is the 7 th most abundant metal. The oxide was independently rediscovered in 1795 by German chemist Martin Heinrich Klaproth in rutile from Hungary. Klaproth found that it contained a new element and named it for the Titans of Greek mythology. 1825 Berzelius isolates metallic titanium • The highest concentrations of Titanium are located in Australia, Canada, New Zealand, Norway, and Ukraine. • Known reserves exceed 600 million tons and is currently being mined at 90, 000 tones a year.

History of Titanium § It is widely distributed and occurs primarily in the minerals anatase, brookite, ilmenite, perovskite, rutile, titanite (sphene), as well in many iron ores. (rutile, anatase and brookite as Ti. O 2) which are widely distributed in the Earth's crust. § Titanium is contained in meteorites and has been detected in the sun and in stars § The coolest type of star with a surface temperature of 3, 200 °C (5, 790 °F). § Rocks brought back from the moon during the Apollo 17 mission are composed of 12. 1% Ti. O 2. § It is also found in coal ash, plants, and even the human body. § The metal is extracted from its principal mineral ores via the Kroll process

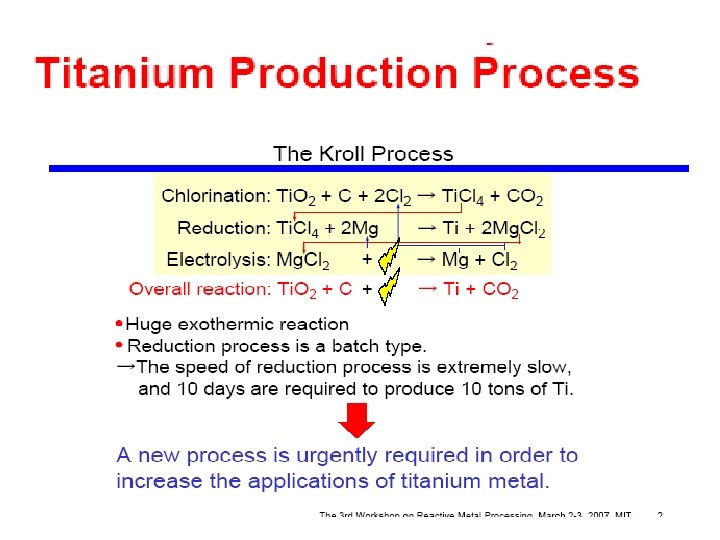
Extraction Unlike iron, Titanium cannot be extracted from their oxides by reduction with carbon because: Titanium forms titanium carbide with carbon, which makes the metal brittle. The processes required to extract titanium from its various ores known as the Kroll process Titanium sponge, made by the Kroll process

Titanium is isolated using the Kroll method. This involves the action of chlorine and carbon on the titanium ore followed by fractional distillation and then reduction with magnesium.

COST • Titanium of very high purity was made in small quantities when Anton Eduard van Arkel and Jan Hendrik de Boer discovered the iodide, • or crystal bar, process in 1925, by reacting with iodine and decomposing the formed vapors over a hot filament to pure metal. Titanium extraction is expensive because the process involves several stages and a lot of energy. This especially limits the uses of titanium.

Isotopes of Titanium § Naturally occurring titanium is composed of 5 stable isotopes: 46 Ti, 47 Ti, 48 Ti, 49 Ti, and 50 Ti, with 48 Ti being the most abundant (73. 8% natural abundance). § Eleven radioisotopes have been characterized, with the most stable being 44 Ti with a half-life of 63 years, 45 Ti with a half-life of 184. 8 minutes, 51 Ti with a half-life of 5. 76 minutes, and 52 Ti with a half-life of 1. 7 minutes. § All of the remaining radioactive isotopes have halflives that are less than 33 seconds and the majority of these have half-lives that are less than half a second.

Physical properties of Titanium 1. Titanium is metallic element, recognized for its high strength-to-weight ratio. 2. It is a strong metal with low density that is quite ductile (especially in an oxygen-free environment) 3. lustrous, and metallic-white in color 4. The relatively high melting point (more than 1650 °C or 3, 000 °F) makes it useful as a refractory metal. 5. It is paramagnetic and has fairly low electrical and thermal conductivity. 6. Titanium's properties are chemically and physically similar to zirconium, because both of them have the same number of valence electrons and are in the same group in the periodic table

Titanium, when pure, is a lustrous, white metal. The metal burns in air and is the only element that burns in nitrogen. It is marvellous in fireworks.

Alloys of Titanium • Commercial (99. 2% pure) grades of titanium have tensile strength equal to that of common, low-grade steel alloys, but are 45% lighter. • Titanium is 60% more dense than aluminum, but more than twice as strong as the most commonly used 6061 -T 6 aluminum alloy. • Certain titanium alloys (e. g. , Beta C) achieve tensile strengths of over 200, 000 psi (1, 400 MPa). • However, titanium loses strength when heated above 430 °C (806 °F).

Food additives'


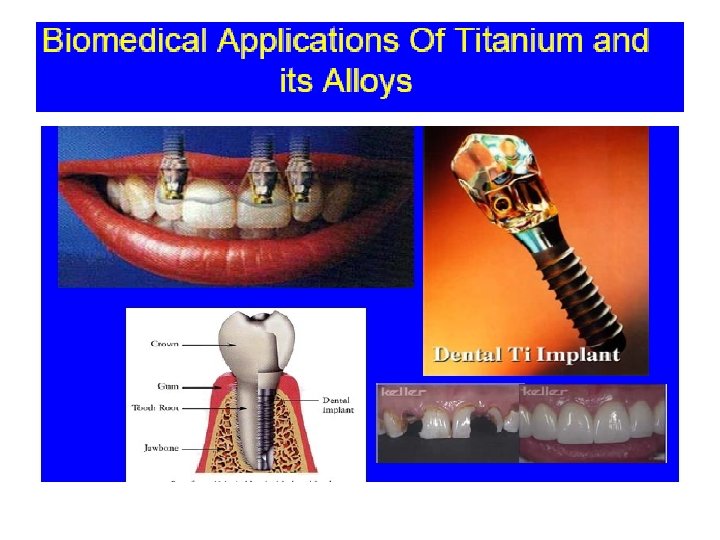
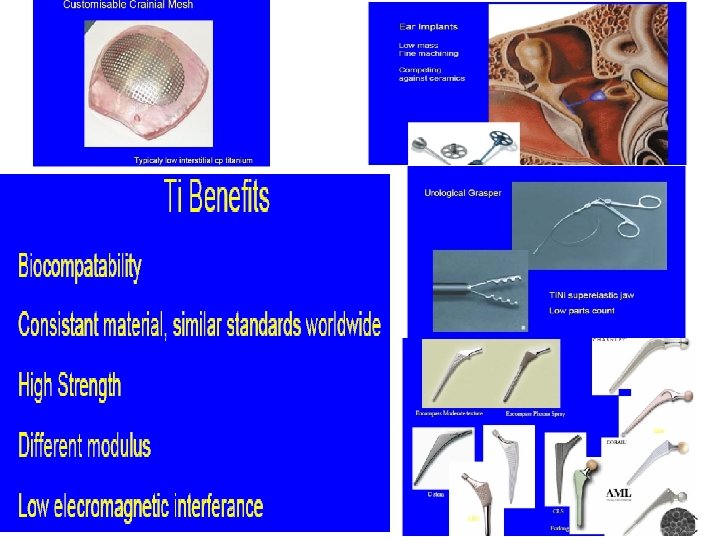

Uses of Titanium Properties Uses 1. Low density, Strong Can be alloyed with Iron, Aluminium, Vanadium, Molybdenum Can make strong lightweight alloys for aircraft and aerospace manufacture 2. Lustrous / high strength to weight ratio/Silver colour Jewellery, sporting goods , mobile phones 3. Corrosion-resistant Reaction Vessels and pipes 4. Biocompatable (non-toxic to or rejected by the body) Medical prostheses, orthopedic and dental implants and instruments

General Uses of Titanium > Titanium is used for alloys with aluminum, molybdenum, manganese, iron, and other metals. These alloys of titanium are used principally in the aerospace industry, for both airframes and engines, where lightweight strength and ability to withstand extremes of temperature are important. > Titanium is as strong as steel, but much lighter. It is twice as strong as aluminum. It is nearly as resistant to corrosion as platinum. > Titanium is a component of joint replacement parts, including hip ball and sockets.

JIt has excellent resistance to sea water and is used for propeller shafts, rigging, and other parts of ships exposed to salt water. J A titanium anode coated with platinum provides cathodic protection from corrosion by salt water. JTitanium paint is an excellent reflector of infrared radiation, and is extensively used in solar observatories where heat causes poor viewing conditions.

A Pure titanium dioxide is relatively clear and has an extremely high index of refraction with an optical dispersion higher than diamond. It is produced artificially for use as a gemstone, but it is relatively soft. A Star sapphires and rubies exhibit their asterism as a result of the presence of Ti. O 2. A The dioxide is used extensively for paint as it is permanent and has good covering power. Titanium oxide pigment accounts for the largest use of the element.

Chemical Reactions Titanium is one of the few elements that urns in pure nitrogen gas, reacting at 00 °C to form titanium nitride §it readily reacts with oxygen When exposed to elevated temperatures in air. This occurs at 1, 200 °C in air, .

Chemical Reactions

Aluminium and Titanium resist corrosion as they have a very thin layer of their oxides on the surface, which stops air and water getting to the metal. Ti is slow to react with water and air, because it forms a passive and protective oxide coating that protects it from further reaction.

More Reactions g. This picture shows the reaction between titanium metal and potassium perchlorate (KCl. O 4).

Health and safety • Titanium is non-toxic even in large doses and does not play any natural role inside the human body. • An estimated 0. 8 milligrams of titanium is ingested by humans each day but most passes through without being absorbed. • An unknown mechanism in plants may use titanium to stimulate the production of carbohydrates and encourage growth. This may explain why most plants contain about 1 part per million (ppm) of titanium, food plants have about 2 ppm, and horsetail and nettle contain up to 80 ppm. • As a powder or in the form of metal shavings, titanium metal poses a significant fire hazard and, when heated in air, an explosion hazard. • Water and carbon dioxide-based methods to extinguish fires are ineffective on burning titanium; Class D dry powder fire fighting agents must be used instead.


Hafnium is used in nuclear reactor control rods in nuclear submarines
- Slides: 29