Tissue Reservoirs Melissa Churchill HIV Neuropathogenesis Laboratory Centre

Tissue Reservoirs Melissa Churchill HIV Neuropathogenesis Laboratory, Centre for Biomedical Research, Burnet Institute. Departments of Medicine and Microbiology, Monash University

Outline § What is a tissue reservoir § Types of tissue reservoirs § The central nervous system as a tissue reservoir § Eradication and the CNS reservoir § Highlights from the Neuro. AIDS pre conference symposium: July 18 th AMREP.
Types of tissue reservoirs
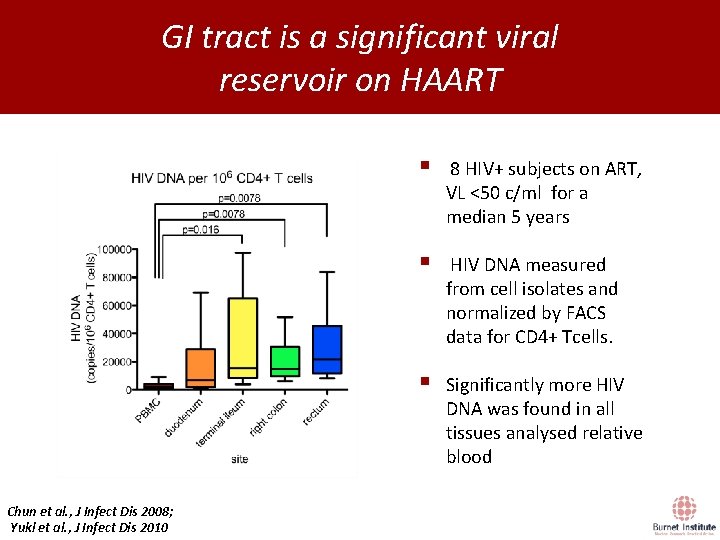
GI tract is a significant viral reservoir on HAART Chun et al. , J Infect Dis 2008; Yukl et al. , J Infect Dis 2010 § 8 HIV+ subjects on ART, VL <50 c/ml for a median 5 years § HIV DNA measured from cell isolates and normalized by FACS data for CD 4+ Tcells. § Significantly more HIV DNA was found in all tissues analysed relative blood
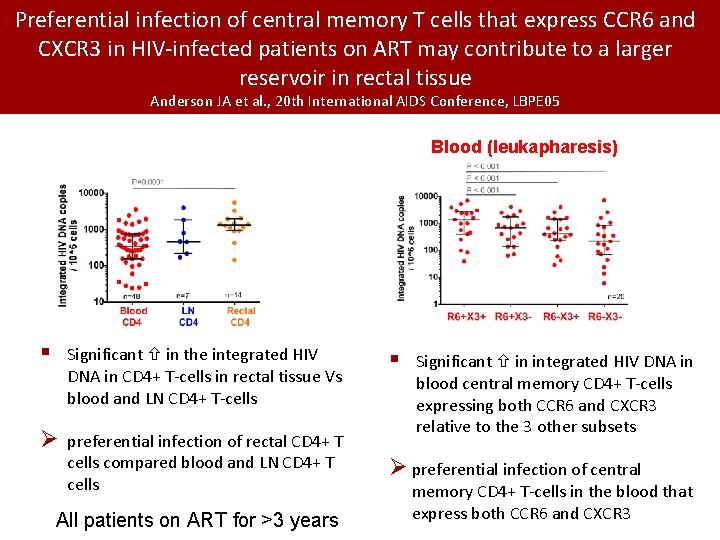
Preferential infection of central memory T cells that express CCR 6 and CXCR 3 in HIV-infected patients on ART may contribute to a larger reservoir in rectal tissue Anderson JA et al. , 20 th International AIDS Conference, LBPE 05 Blood (leukapharesis) § Significant in the integrated HIV DNA in CD 4+ T-cells in rectal tissue Vs blood and LN CD 4+ T-cells Ø preferential infection of rectal CD 4+ T cells compared blood and LN CD 4+ T cells All patients on ART for >3 years § Significant in integrated HIV DNA in blood central memory CD 4+ T-cells expressing both CCR 6 and CXCR 3 relative to the 3 other subsets Ø preferential infection of central memory CD 4+ T-cells in the blood that express both CCR 6 and CXCR 3
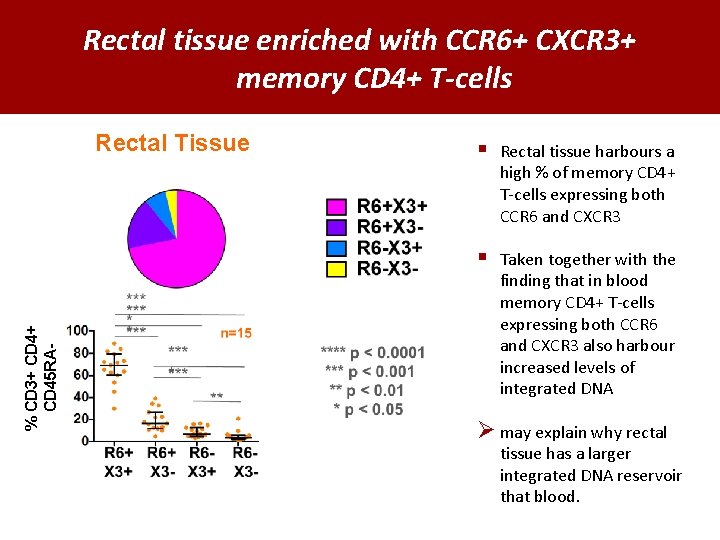
Rectal tissue enriched with CCR 6+ CXCR 3+ memory CD 4+ T-cells % CD 3+ CD 45 RA- Rectal Tissue § Rectal tissue harbours a high % of memory CD 4+ T-cells expressing both CCR 6 and CXCR 3 § Taken together with the finding that in blood memory CD 4+ T-cells expressing both CCR 6 and CXCR 3 also harbour increased levels of integrated DNA Ø may explain why rectal tissue has a larger integrated DNA reservoir that blood.
Types of tissue reservoirs

The CNS as viral reservoir § The CNS is a potential but controversial viral reservoir of HIV § Potential: during the course of disease HIV enters the CNS. § Controversial: no direct evidence that in virally suppressed patients the CNS contains HIV infected cells harboring competent pro-viral genomes. However, there is a significant amount of indirect evidence from numerous studies to suggest, that at least in some HIV infected virally suppressed patients, the CNS may be a viral reservoir

Key questions relevant to the CNS compartment as a tissue reservoir § Are cells of the CNS infected with HIV-1, in sufficient numbers, and contain integrated pro-viral genomes during suppressive c. ART? § Is there evidence of ongoing persistent replication and immune activation within the CNS in the presence of suppressive c. ART? § Do HIV-1 infected cells in the CNS have mechanisms in place to allow for the virus to escape from the biochemical decay processes or immune mechanisms and persist for long period of times, ie long lived cells (latency) § Do cells of the CNS have regulatory mechanisms that facilitate a latent infection?

Are cells of the CNS infected with HIV and contain an integrated pro-viral genome?

CNS targets of HIV infection § Macrophages: major sites of productive infection § resident microglia § perivascular microglia and macrophages, § meningeal macrophages, § choroid plexus macrophages (Hickey, 1999 a). § Astrocytes: infection non-productive and restricted (Takahashi et al, 1996; Sharer et al. , 1996; An et al, 1999)
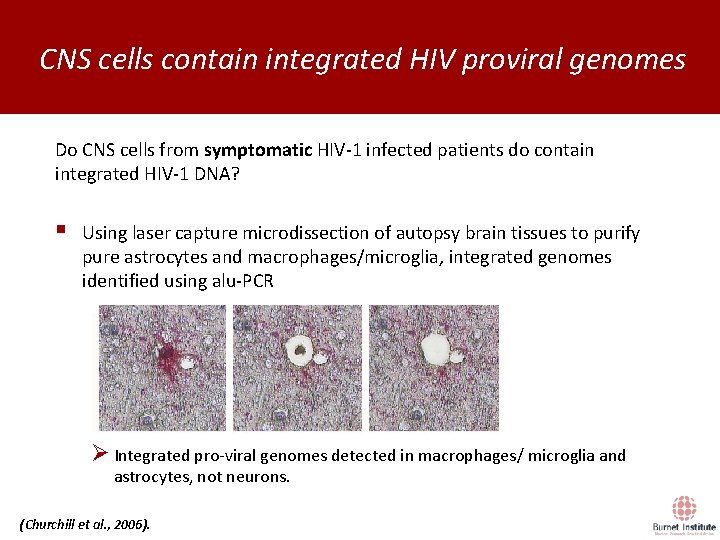
CNS cells contain integrated HIV proviral genomes Do CNS cells from symptomatic HIV-1 infected patients do contain integrated HIV-1 DNA? § Using laser capture microdissection of autopsy brain tissues to purify pure astrocytes and macrophages/microglia, integrated genomes identified using alu-PCR Ø Integrated pro-viral genomes detected in macrophages/ microglia and astrocytes, not neurons. (Churchill et al. , 2006).
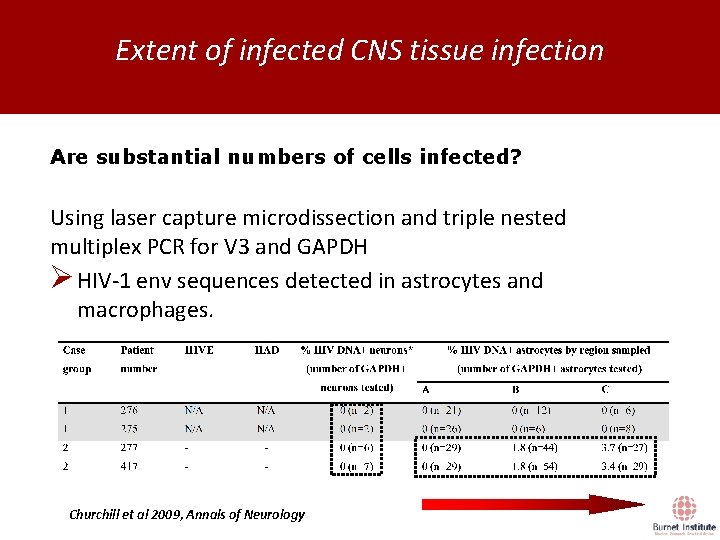
Extent of infected CNS tissue infection Are substantial numbers of cells infected? Using laser capture microdissection and triple nested multiplex PCR for V 3 and GAPDH Ø HIV-1 env sequences detected in astrocytes and macrophages. Churchill et al 2009, Annals of Neurology
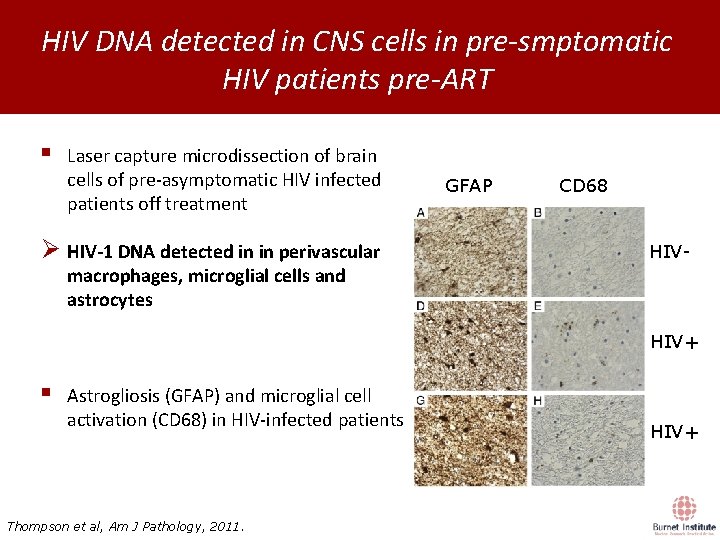
HIV DNA detected in CNS cells in pre-smptomatic HIV patients pre-ART § Laser capture microdissection of brain cells of pre-asymptomatic HIV infected patients off treatment Ø HIV-1 DNA detected in in perivascular GFAP CD 68 HIV- macrophages, microglial cells and astrocytes HIV+ § Astrogliosis (GFAP) and microglial cell activation (CD 68) in HIV-infected patients Thompson et al, Am J Pathology, 2011. HIV+

Independent and distinct evolution of HIV-1 occurs in the CNS compartment: evidence of infection and replication § HIV-1 sequence compartmentalization § CNS and non CNS § § different brain regions (Ohagen et al 2003, Thomas et al 2007, Harrington et al. , 2009, Schnell et al. , 2009, Sturdevant et al. , 2012) different cell types (Smit et al 2001) (Thompson, Churchill et al, Annals Neurol. 2004) at the level of Env, Nef, Tat and the LTR § Functional compartmentalization § CNS derived envelopes have an enhanced ability to utilise low levels of CD 4 permitting m-tropism. Ø more open conformation, exposing CD 4 binding domain § sensitivity to CD 4 binding site n-Abs than matched viruses from blood (Gorry et al. , 2002, Peters et al. , 2004 & 2008, Thomas et al. , 2007, Dunfee et al. , 2009)

Ongoing replication in the CNS in the presence of c. ART

Ongoing replication during c. ART: asymptomatic CSF viral persistence Eden et al. , JID 2010 § In patients on suppressive c. ART (Plasma VL<50 cps/ml) Drug EFV LPV/r ATV/r TDF ABC ZDC 3 TC FTC CSF viral escape, n(%) 4 (15 %) 1 (5 %) 2 (10 %) 3 (9 %) 4 (22 %) 0 (0 %) 4 (10 %) 3 (12 %) Ø 10% patients had detectable CROI 2014 of. UPDATESVL (>50 cps/ml) in CSF using a Rate of asymptomatic CSF escape: standard assay - 13% in UK (Abstract 442) - 10. 3%atinal. , Italy (Abstract Letendre (CROI 2009) 443) §- 300 with (Abstract suppressed 23%patients in Sweden 445) plasma VLrelates (<50 cps/ml) using CSF escape to depression incidence (Abstract 33) an ultra sensitive assay Ø 40% of patients has CSF viral escape with more than 2 cps/ml in the CSF. Slide courtesy of Steve Deeks
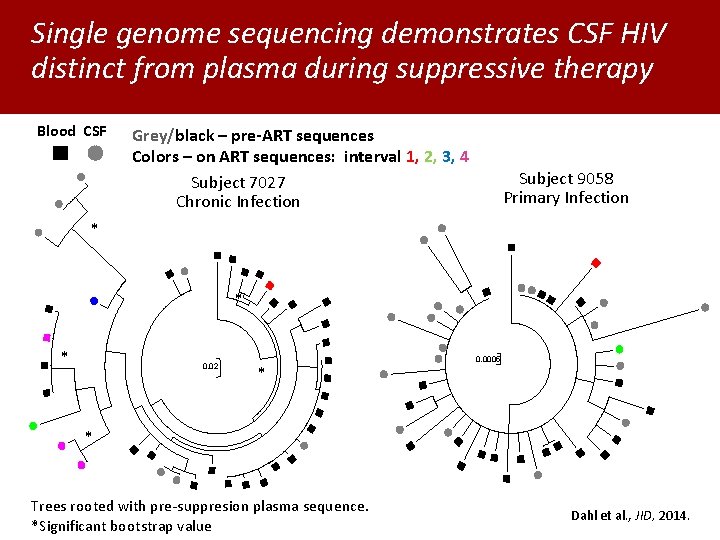
Single genome sequencing demonstrates CSF HIV distinct from plasma during suppressive therapy Blood CSF Grey/black – pre-ART sequences Colors – on ART sequences: interval 1, 2, 3, 4 Subject 7027 Chronic Infection Subject 9058 Primary Infection * * * 0. 02 * 0. 0005 * Trees rooted with pre-suppresion plasma sequence. *Significant bootstrap value Dahl et al. , JID, 2014.

Indirect evidence of ongoing replication during c. ART: symptomatic CSF ‘escape’ Lamivudine Abacavir Lopinavir/r hand tremor, CSF slurred WBC = ataxia, 26 cells/ul* speech, aphasia CSF HIV RNA Patient 2000 § Plasma VL <50 cps/ml for 8 yrs, § presents with onset of HAND Ø found elevated levels of virus in CSF Latest CD 4 308 cells/ul * Normal CSF WBC < 5 cells/ul Slide courtesy of A/Prof Serena Spudich Peluso et al. , AIDS, 2012. Also: Canestri et al. , Clin Infect Diseases, 2010.

Indirect evidence of ongoing replication during c. ART: symptomatic CSF ‘escape’ Genotyping of the CSF virus revealed resistance to almost all drugs in regime Ø Suggested ongoing replication and evolution in the CNS away from the virus detected in the plasma and fully suppressed Ø Adjustment of the drug regiment resulted in resolution of signs and symptoms Slide courtesy of A/Prof Serena Spudich

Ongoing replication in the CNS in the presence of c. ART: Immune activation and biomarkers
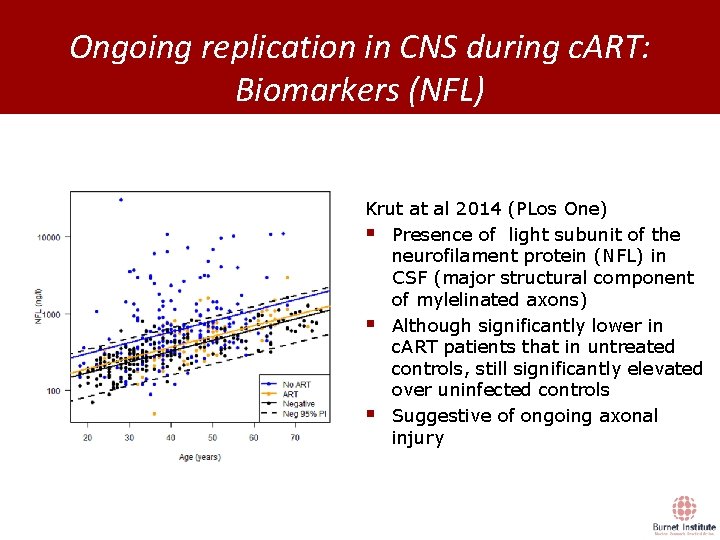
Ongoing replication in CNS during c. ART: Biomarkers (NFL) Krut at al 2014 (PLos One) § Presence of light subunit of the neurofilament protein (NFL) in CSF (major structural component of mylelinated axons) § Although significantly lower in c. ART patients that in untreated controls, still significantly elevated over uninfected controls § Suggestive of ongoing axonal injury
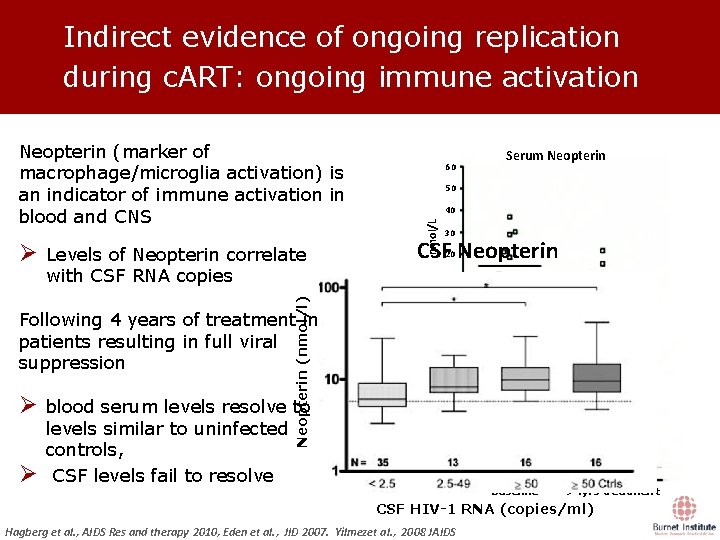
Indirect evidence of ongoing replication during c. ART: ongoing immune activation Levels of Neopterin correlate with CSF RNA copies Neopterin (nmol/l) Ø 60 40 30 CSF Neopterin 20 10 Baseline Following 4 years of treatment in patients resulting in full viral suppression Ø blood serum levels resolve to levels similar to uninfected controls, CSF levels fail to resolve 60 >4 yrs treatment CSF Neopterin 50 40 nmol/L Ø Serum Neopterin 50 nmol/L Neopterin (marker of macrophage/microglia activation) is an indicator of immune activation in blood and CNS 30 20 10 Baseline >4 yrs treatment CSF HIV-1 RNA (copies/ml) Hagberg et al. , AIDS Res and therapy 2010, Eden et al. , JID 2007. Yilmezet al. , 2008 JAIDS
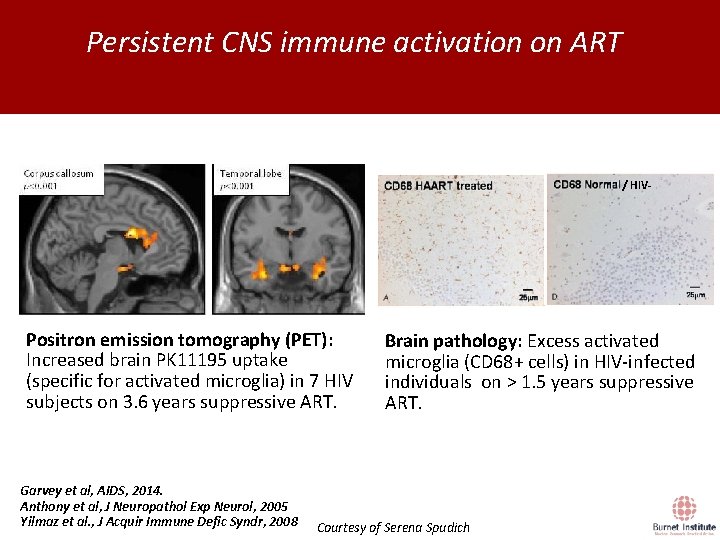
Persistent CNS immune activation on ART / HIV- Positron emission tomography (PET): Increased brain PK 11195 uptake (specific for activated microglia) in 7 HIV subjects on 3. 6 years suppressive ART. Garvey et al, AIDS, 2014. Anthony et al, J Neuropathol Exp Neurol, 2005 Yilmaz et al. , J Acquir Immune Defic Syndr, 2008 Brain pathology: Excess activated microglia (CD 68+ cells) in HIV-infected individuals on > 1. 5 years suppressive ART. Courtesy of Serena Spudich

Latency in the CNS

in vitro latency § in vitro studies have demonstrated that infection of astrocytes is non productive/latent (Brack-Werner et al, 1992; Neumann et al; 1995, Gorry et al, 1999) § one of the major blocks to viral production in astrocytes is at the level of transcription (Shahabuddin, Volsky et al. 1992, Swingler, Easton et al. 1992, Ludvigsen, Brack-Werner et al. 1996, Niikura, et al, . 1996, Ensoli, Wang et al. , 1997) § in vitro studies have demonstrated persistent HIV-1 can be reactivated in human fetal glial cells using TNF alpha and interlukin-1 beta (Tornatore et al. , 1991) § Elevated levels of COUP-TF biding protein CTIP 2(Bcl 11 B) in HIV+ ‘latent’ microglia involved in the recruitment of histone demethylases to the LTR (Le Douce et al, 2012; , Desplats et al. , 2013 Neurology)
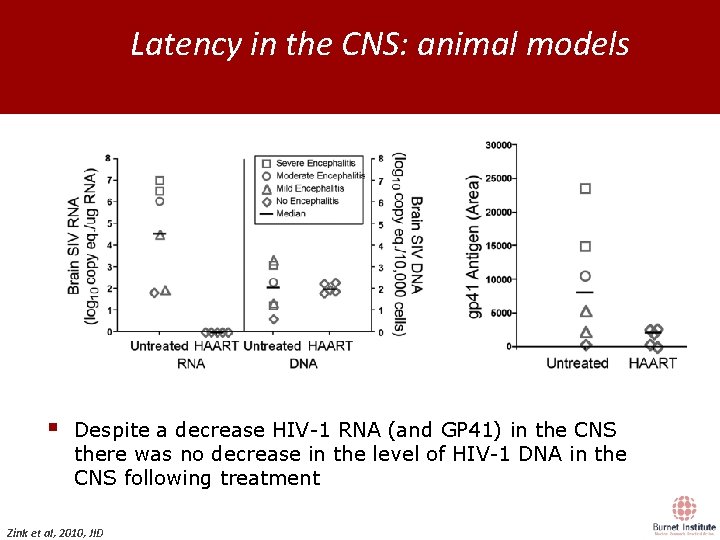
Latency in the CNS: animal models Day 0 12 IV SIV Innoculation 175 Examination Treatment • no ART • saquinavir/atazanavir, integrase inhibitor • atazanavir, integrase inhibitor § Despite a decrease HIV-1 RNA (and GP 41) in the CNS there was no decrease in the level of HIV-1 DNA in the CNS following treatment Zink et al, 2010, JID

Latency in the CNS: responsiveness to transcriptional activators
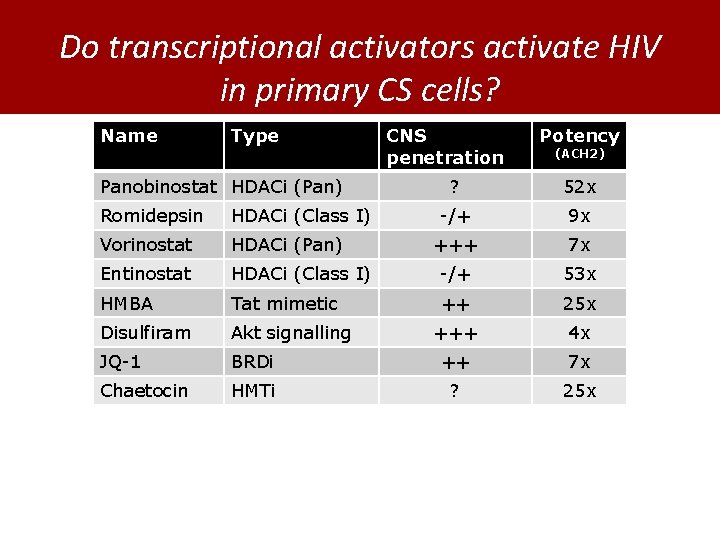
Do transcriptional activators activate HIV in primary CS cells? Name Type Panobinostat HDACi (Pan) CNS penetration Potency (ACH 2) ? 52 x -/+ 9 x +++ 7 x Romidepsin HDACi (Class I) Vorinostat HDACi (Pan) Entinostat HDACi (Class I) -/+ 53 x HMBA Tat mimetic ++ 25 x Disulfiram Akt signalling +++ 4 x JQ-1 BRDi ++ 7 x Chaetocin HMTi ? 25 x
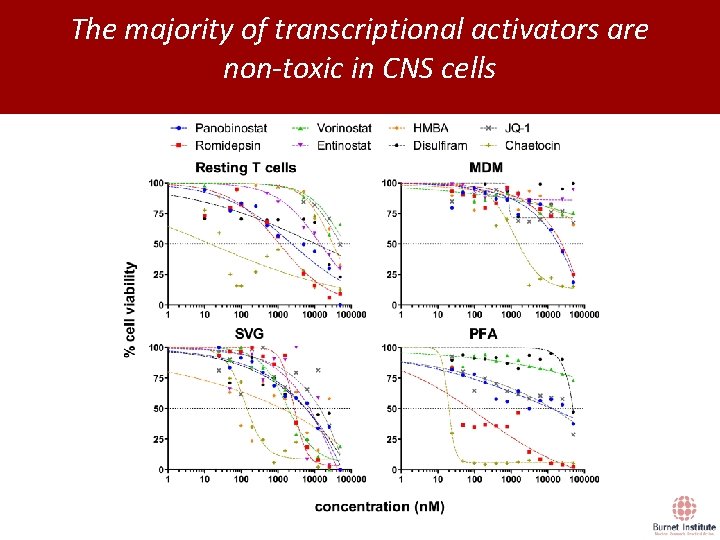
The majority of transcriptional activators are non-toxic in CNS cells
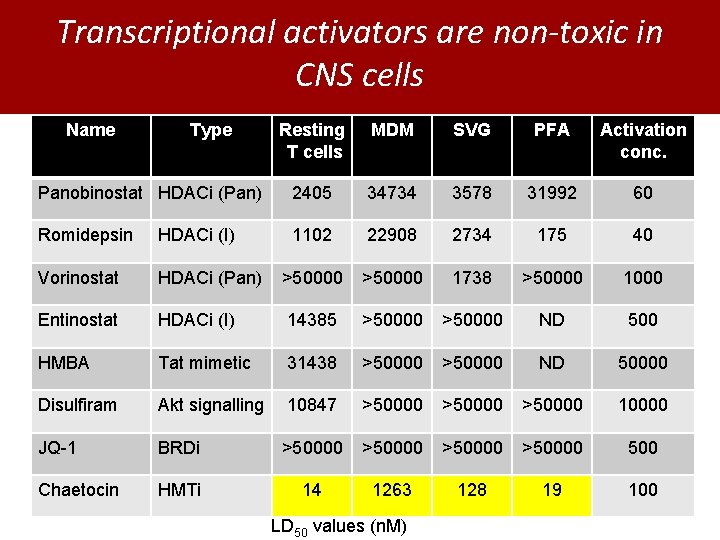
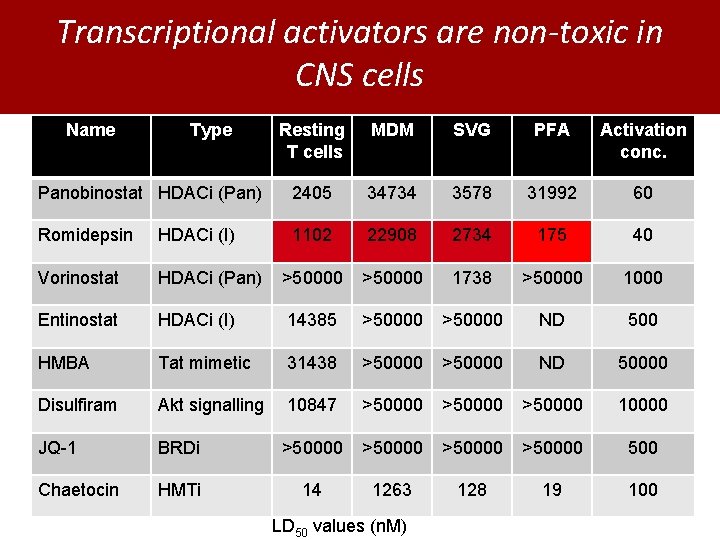
Transcriptional activators are non-toxic in CNS cells Name Type Resting T cells MDM SVG PFA Activation conc. Panobinostat HDACi (Pan) 2405 34734 3578 31992 60 Romidepsin HDACi (I) 1102 22908 2734 175 40 Vorinostat HDACi (Pan) >50000 1738 >50000 1000 Entinostat HDACi (I) 14385 >50000 ND 500 HMBA Tat mimetic 31438 >50000 ND 50000 Disulfiram Akt signalling 10847 >50000 10000 JQ-1 BRDi >50000 500 Chaetocin HMTi 14 1263 128 19 100 LD 50 values (n. M)
Transcriptional activators are non-toxic in CNS cells Name Type Resting T cells MDM SVG PFA Activation conc. Panobinostat HDACi (Pan) 2405 34734 3578 31992 60 Romidepsin HDACi (I) 1102 22908 2734 175 40 Vorinostat HDACi (Pan) >50000 1738 >50000 1000 Entinostat HDACi (I) 14385 >50000 ND 500 HMBA Tat mimetic 31438 >50000 ND 50000 Disulfiram Akt signalling 10847 >50000 10000 JQ-1 BRDi >50000 500 Chaetocin HMTi 14 1263 128 19 100 LD 50 values (n. M)
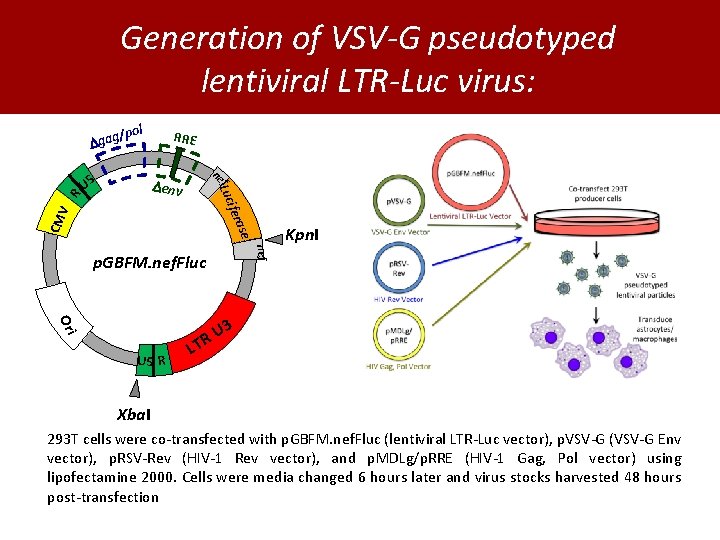
Generation of VSV-G pseudotyped lentiviral LTR-Luc virus: pol U 5 RRE ne f env ase CM V ifer Luc R / gag nef p. GBFM. nef. Fluc Kpn. I Ori 3 U R U 5 R LT Xba. I 293 T cells were co-transfected with p. GBFM. nef. Fluc (lentiviral LTR-Luc vector), p. VSV-G (VSV-G Env vector), p. RSV-Rev (HIV-1 Rev vector), and p. MDLg/p. RRE (HIV-1 Gag, Pol vector) using lipofectamine 2000. Cells were media changed 6 hours later and virus stocks harvested 48 hours post-transfection
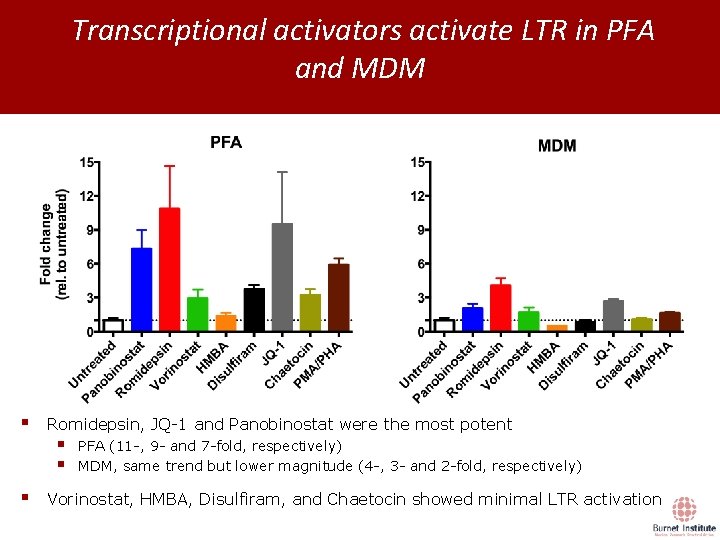
Transcriptional activators activate LTR in PFA and MDM § § Romidepsin, JQ-1 and Panobinostat were the most potent § § PFA (11 -, 9 - and 7 -fold, respectively) MDM, same trend but lower magnitude (4 -, 3 - and 2 -fold, respectively) Vorinostat, HMBA, Disulfiram, and Chaetocin showed minimal LTR activation

Summary § There is strong data to suggest persistent HIV replication can occur in the CNS during suppressive c. ART § The CNS by nature presents unique and extensive challenges. How do we treat activation in the CNS? What are the targets? Do antiretroviral drugs penetrate the CNS? Do antiretroviral function in CNS? Immune control? Biomarkers? § Assuming a CNS reservoir exists current ‘cure’ strategies are likely to result in replication in the CNS

Summary § Tissue reservoirs are currently a major barrier to the eradication/cure of HIV-1 § § identification and characterisation of all viral reservoirs of HIV-1 including the CNS Importantly ‘Do cells within CNS cells, during suppressive c. ART, contain a competent integrated HIV-1 genome? ’

Acknowledgements Burnet Institute HIV Neuropathogenesis Lachlan Gray Daniel Cowley Wan Jun Cheng Hung On Anne Ellet Steve Wesselingh Kirby Institute Stuart Turville Peter Doherty Institute Damian Purcell Jonathan Jacobsen Johns Hopkins Justin Mc. Arthur HIV Molecular Pathogenesis Carlos Pardo Paul Gorry St Vincents Hospital Sydney Michael Roche Bruce Brew HIV Molecular Interactions Gilles Guillemin Gilda Tachedjian Alfred Hospital Sharon Lewin, Hao Lu Michael Moso Fiona Wightman APP 1051093 APP 1009533 Yale University Serena Spudich R 21 MH 100954 -02 NIHU 19 A 1096109
- Slides: 37