Tissue Plasminogen Activator for Acute Ischemic Stroke National
















- Slides: 17

Tissue Plasminogen Activator for Acute Ischemic Stroke National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group

Summarise the paper in 200 words

Summarise the paper in 200 words • Aims – Compare intravenous tissue plasminogen activator (t-PA) with placebo in treatment of acute ischaemic stroke. • Method – 2 part double blinded multi-centre randomised controlled trial – Patients with ischaemic stroke (no intracranial haemorrhage on CT) within 3 hours of onset were included – Stratified block randomisation to treatment with placebo or t-PA

Summarise the paper in 200 words • Outcome measures: – Part 1: Complete resolution of neurological deficit or improvement in NIHSS score >4 within 24 hours of onset of stroke – Part 2: Recovery with minimal or no deficit 3 months after treatment, measured using a combination of 4 outcome measures (Barthel index, modified Rankin scale, Glasgow outcome scale, NIHSS)

Summarise the paper in 200 words • Results – Part 1 (n=291): no significant difference in percentage of patients with neurological improvement at 24 hours (relative risk =1. 2 (95% CI 0. 9 -1. 6)) – Part 2 (n=333): Odds ratio for favourable outcome in combined test statistic at 3 months in t-PA group =1. 7 (95% CI 1. 2 -2. 6, p=0. 008) – No significant difference in mortality, but higher rate of symptomatic intracerebral haemorrhage within 36 hrs seen in t-PA group (p<0. 001)

Summarise the paper in 200 words • Conclusion – Despite an increased incidence of symptomatic intracerebral hemorrhage, treatment with t-PA within 3 hours onset of ischemic stroke improved clinical outcome at 3 months.

Abstract

The Good. . . • • Multi-centre RCT Clear power calculation performed Few patients lost to follow up Intention to treat analysis

The Bad. . . • Patient selection – were all eligible patients randomised? • No CONSORT diagram • Differences between placebo and treatment groups – No mean given for NIHSS score - makes comparison difficult – Higher rate of “Large-vessel occlusive stroke” in placebo group – Higher rate of edema and mass effect in placebo group • Unconventional statistical tests applied – (Mantel-Haenszel test and Wald test) • No p-value given for differences in adverse events – (symptomatic intracerebral haemorrhage and serious systemic bleeding)

The Bad. . . • Dichotomous end-point in part 2 – No assessment of magnitude of effect of treatment can be made • Randomisation method – Permuted block design with blocks of various sizes. Stratified by clinical centre (8 centres) and time to treatment • Was part 2 only done because part 1 failed to deliver a significant outcome?

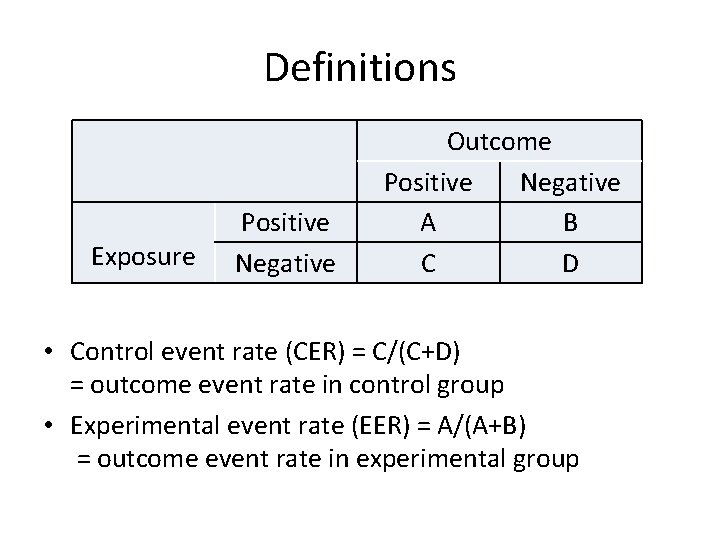
Definitions Exposure Positive Negative Outcome Positive Negative A B C D • Control event rate (CER) = C/(C+D) = outcome event rate in control group • Experimental event rate (EER) = A/(A+B) = outcome event rate in experimental group

Definitions Outcome Exposure Positive Negative Positive A B Negative C D • Control event rate (CER) = C/(C+D) • Experimental event rate (EER) = A/(A+B) • Absolute risk reduction (ARR) = CER-EER • Relative risk (RR) = EER/CER • Relative risk reduction (RRR) = (CER-EER)/CER

Definitions • Number needed to treat (NNT) – The number of subjects that must be treated with the intervention, compared with the control, for one extra subject to experience the beneficial effect. – NNT = 1/ARR

Definitions Outcome Exposure Positive Negative Positive A B Negative C D • Control event rate (CER) = C/(C+D) • Experimental event rate (EER) = A/(A+B) • • Absolute risk reduction (ARR) = CER-EER Relative risk (RR) = EER/CER Relative risk reduction (RRR) = (CER-EER)/CER Number needed to treat (NNT) = 1/ARR

Bonus Points • Using the numbers given for the Barthel index in part 2 of the study (Table 4), calculate the number needed to treat (NNT) in order to get a favourable functional outcome. • Absolute risk reduction = 50 -38 =12% • NNT= 1/ARR = 1/0. 12 = 8. 3

Bonus Points • Using the data given in Table 6, calculate the overall number needed to harm (NNH) for symptomatic intracranial haemorrhage (parts 1 and 2 of the study combined). • Risk of symptomatic intracranial haemorrhage – t-PA group = (8+12)/(144+168) = 0. 064 = 6. 4% – Placebo = (0+2)/(147+165) = 0. 006 = 0. 6% • Absolute risk reduction = 6. 4 -0. 6 = 5. 8% • NNH = 1/ARR = 1/0. 058 = 17. 2

• Further analysis of stroke thrombolysis: www. thennt. com/thrombolytics-for-stroke/