Tipping the balance between electrostatics and steric effects

Tipping the balance between electrostatics and steric effects: the microwave spectra and molecular structures of 2 -chloro-1, 1 -difluoroethylene-acetylene and cis-1, 2 -difluoroethylene-acetylene Helen O. Leung & Mark D. Marshall Department of Chemistry Amherst College Supported by the National Science Foundation

Some haloethylene-HCCH complexes Cole & Legon CPL 369, 31 (2003) Leung, Marshall, Cashion, & Chen JCP 128, 064315 (2008) less nucleophilic less electropositive more nucleophilic Leung, Marshall, & Feng JPCA 117, 13419 (2013) • Steric factors favor top binding mode. • Electrostatic factors lead to side binding mode. • Cl relaxes steric requirements, allowing HCCH to adopt a side binding mode. • When forced to adopt a top binding mode, HCCH prefers F to Cl. • When forced to adopt a side binding mode, HCCH prefers Cl to F. Leung, Marshall, & Grimes JCP 134, 034303 (2011) Leung, Marshall, & Khan JPCA 121, 5651 (2017)

Exploring other haloethylene-HCCH complexes • Does HCCH bind to an F, H pair in cis-1, 2 -difluoroethylene? • How does HCCH bind to 2 -chloro-1, 1 -difluoroethylene? • • Top binding with F, H pair? Side binding with Cl, H pair?
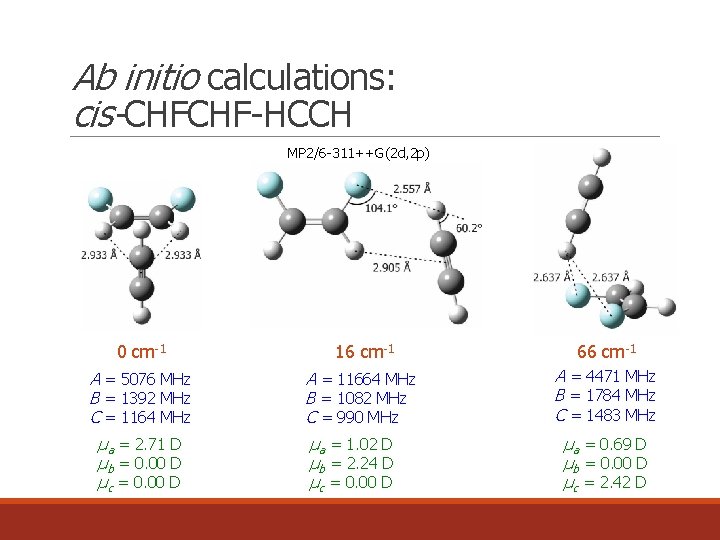
Ab initio calculations: cis-CHFCHF-HCCH MP 2/6 -311++G(2 d, 2 p) 0 cm-1 16 cm-1 66 cm-1 A = 5076 MHz B = 1392 MHz C = 1164 MHz A = 11664 MHz B = 1082 MHz C = 990 MHz A = 4471 MHz B = 1784 MHz C = 1483 MHz µa = 2. 71 D µb = 0. 00 D µc = 0. 00 D µa = 1. 02 D µb = 2. 24 D µc = 0. 00 D µa = 0. 69 D µb = 0. 00 D µc = 2. 42 D

Ab initio calculations: CF 2 CHCl-HCCH MP 2/6 -311++G(2 d, 2 p) 0 cm-1 23 cm-1 168 cm-1 453 cm-1 A = 2637 MHz B = 1013 MHz C = 732 MHz A = 2069 MHz B = 1485 MHz C = 1043 MHz A = 2306 MHz B = 1063 MHz C = 727 MHz A = 7080 MHz B = 657 MHz C = 623 MHz µa = 1. 15 D µb = 0. 74 D µc = 0. 00 D µa = 0. 05 D µb = 0. 16 D µc = 1. 21 D µa = 1. 22 D µb = 0. 52 D µc = 0. 00 D µa = 0. 02 D µb = 1. 11 D µc = 0. 00 D

FTMW spectroscopy • 1% ethylene, 1% HCCH in Ar • Obtained chirped pulse spectrum in the 5. 6 – 18. 1 GHz region • Collected higher resolution spectrum in the 5 – 21 GHz region with a Balle-Flygare spectrometer • cis-CHFCHF-HCCH • 5 isotopologues studied, all in natural abundance: CHFCHF-HCCH, CHF 13 CHF-HCCH, CHFCHF-H 13 CCH, CHFCHF-HC 13 CCH • CF 2 CHCl-HCCH • 4 isotopologues studied in natural abundance: CF 2 CH 35 Cl-HCCH, CF 2 CH 37 Cl-HCCH using H 13 CCH: CF 2 CH 35 Cl-H 13 CCH, CF 2 CH 35 Cl-HC 13 CHFCHF
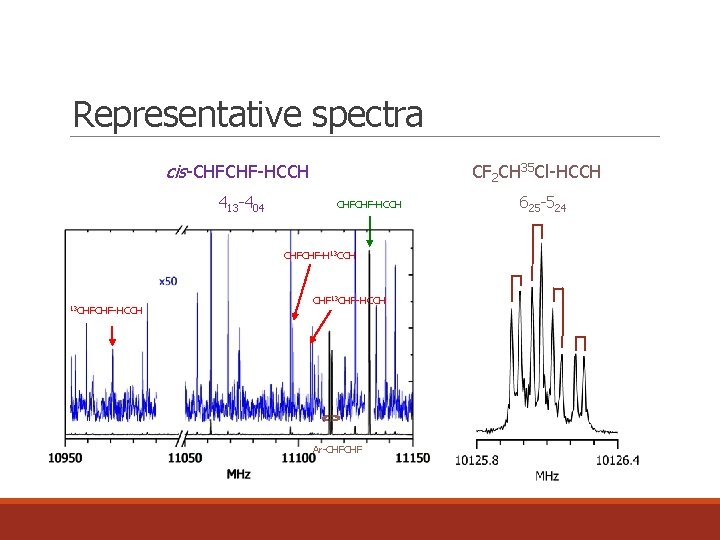
Representative spectra cis-CHFCHF-HCCH 413 -404 CF 2 CH 35 Cl-HCCH CHFCHF-H 13 CCH 13 CHFCHF-HCCH CHF 13 CHF-HCCH Ar-CHFCHF 625 -524

Spectroscopic constants: cis-CHFCHF-HCCH A / MHz B / MHz C / MHz J / 10 -3 MHz JK / 10 -3 MHz J / 10 -3 MHz K / 10 -3 MHz JK / 10 -6 MHz KJ / 10 -6 MHz K / 10 -6 MHz # transitions # a type # b type J range Ka range rms / k. Hz CHFCHF-HCCH CHF 13 CHF-HCCH 13 CHFCHF-HCCH CHFCHF-H 13 CCH CHFCHF-HC 13 CH 11705. 66697(35) 1066. 27667(13) 978. 01549(12) 1. 45299(48) 27. 9085(77) 866. 595(68) 0. 171395(46) 8. 881(55) 0. 0080(15) 0. 723(44) 27. 64(39) 98 58 40 0 – 14 0– 4 1. 05 11678. 68329(61) 11538. 15510(78) 11668. 43706(54) 11601. 35226( 94) 1065. 80015(14) 1059. 85138(16) 1047. 24162(13) 1038. 28714(20) 977. 454979(89) 971. 43530(11) 961. 730707(91) 953. 70149(13) 1. 45295(87) 1. 4369(10) 1. 38497(87) 1. 4163(12) 27. 644(27) 27. 148(36) 26. 618(20) 28. 298(46) [866. 595] 0. 17066(54) 0. 16904(64) 0. 15808(54) 0. 16579(85) [8. 881] A = 5076 MHz A = 11664 MHz B = 1392 MHz [ 0. 0080] B = 1082 MHz [ 0. 0080] C = 1164 MHz C = 990 MHz [0. 723] 6/311++G(2 d, 2 p) [ 27. 64] -1 -1 MP 2 32 16 cm 0 cm 31 34 29 -1 -1 19 point corrected 18 0 cm 21 17 27 cm Zero 13 13 12 12 BSSE corrected 0 cm-1 10 cm-1 0– 8 0– 2 0– 1 40 cm-1 MP 4 0 cm-1 1. 55 1. 80 1. 55 2. 12 CCSD 39 cm-1 0 cm-1 CCSD(T) 22 cm-1 0 cm-1
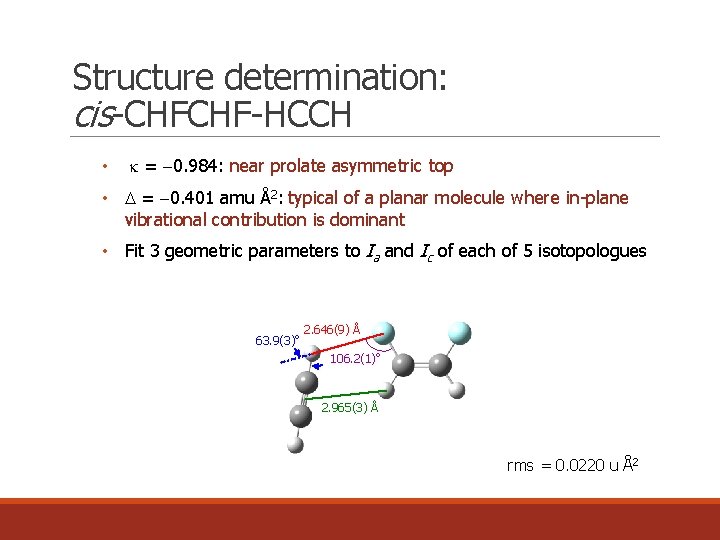
Structure determination: cis-CHFCHF-HCCH • k = 0. 984: near prolate asymmetric top • = 0. 401 amu Å2: typical of a planar molecule where in-plane vibrational contribution is dominant • Fit 3 geometric parameters to Ia and Ic of each of 5 isotopologues 63. 9(3)o 2. 646(9) Å 106. 2(1)o 2. 965(3) Å rms = 0. 0220 u Å2
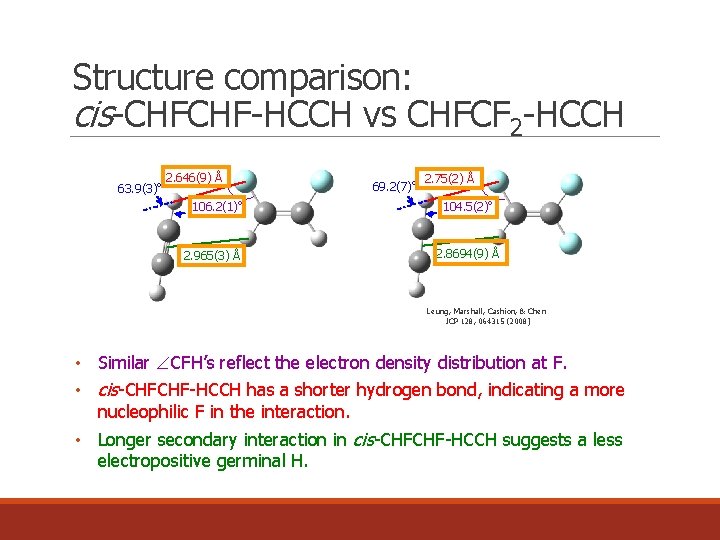
Structure comparison: cis-CHFCHF-HCCH vs CHFCF 2 -HCCH 63. 9(3)o 2. 646(9) Å 106. 2(1)o 2. 965(3) Å 69. 2(7)o 2. 75(2) Å 104. 5(2)o 2. 8694(9) Å Leung, Marshall, Cashion, & Chen JCP 128, 064315 (2008) • Similar CFH’s reflect the electron density distribution at F. • cis-CHFCHF-HCCH has a shorter hydrogen bond, indicating a more nucleophilic F in the interaction. • Longer secondary interaction in cis-CHFCHF-HCCH suggests a less electropositive germinal H.
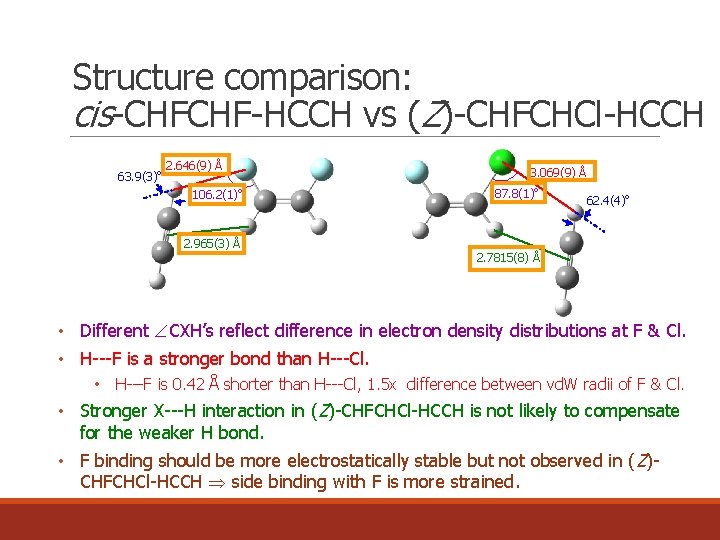
Structure comparison: cis-CHFCHF-HCCH vs (Z)-CHFCHCl-HCCH 63. 9(3)o 2. 646(9) Å 106. 2(1)o 3. 069(9) Å 87. 8(1)o 62. 4(4)o 2. 965(3) Å 2. 7815(8) Å • Different CXH’s reflect difference in electron density distributions at F & Cl. • H---F is a stronger bond than H---Cl. • H---F is 0. 42 Å shorter than H---Cl, 1. 5 x difference between vd. W radii of F & Cl. • Stronger X---H interaction in (Z)-CHFCHCl-HCCH is not likely to compensate for the weaker H bond. • F binding should be more electrostatically stable but not observed in ( Z)CHFCHCl-HCCH side binding with F is more strained.

Spectroscopic constants: CF 2 CHCl-HCCH A / MHz B / MHz C / MHz J / 10 -3 MHz JK / 10 -3 MHz J / 10 -3 MHz K / 10 -3 MHz aa / MHz bb / MHz cc / MHz | ab |/ MHz # rot. transitions (# hyperfine components) # a type # b type J range Ka range rms / k. Hz CF 2 CH 35 Cl-HCCH CF 2 CH 37 Cl-HCCH CF 2 CH 35 Cl-H 13 CCH CF 2 CH 35 Cl-HC 13 CH 2671. 139527(34) 988. 154087(16) 721. 384868(11) 0. 86170(12) 0. 22463(76) 13. 5197(15) 0. 278637(54) 2. 7811(18) 36. 22777(55) 72. 54601(64) 36. 31824(50) 20. 5874(26) 151 (799) 72 79 1 – 12 0– 5 1. 30 2598. 597739(62) 988. 042168(27) 715. 910453(18) 0. 85675(18) 0. 2303(15) 13. 0972(36) 0. 281282(82) 2. 6934(33) 28. 50483(87) 57. 12613(91) 28. 62131(69) 16. 411(29) 80 (346) 48 32 1 – 10 0– 4 1. 01 2670. 993806(80) 966. 462337(33) 709. 750972(24) 0. 81180(21) 0. 0693(15) 13. 3506(46) 0. 258054(98) 2. 7284(41) 36. 2715(18) 72. 5906(14) 36. 3192(15) 2637 MHz 20. 451(29) 1013 MHz 87 732 (341) MHz 52 35 1 – 11 0– 4 1. 30 2648. 901199(99) 964. 007725(46) 706. 846637(31) 0. 84078(26) 0. 1058(19) 12. 7986(57) 0. 26805(13) 2. 7764(50) 36. 7690(23) 73. 0885(18) 36. 3195(20) 19. 0740(51) 77 (294) 48 29 1 – 10 0– 4 1. 37 A= B= C=
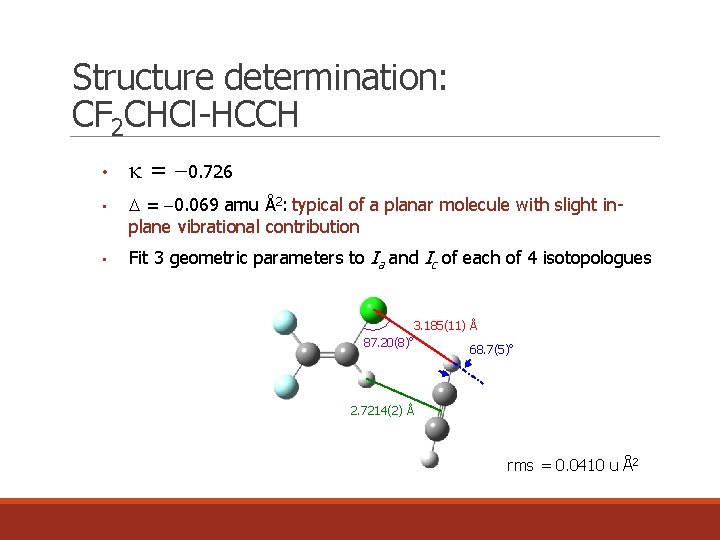
Structure determination: CF 2 CHCl-HCCH • • • k = 0. 726 = 0. 069 amu Å2: typical of a planar molecule with slight inplane vibrational contribution Fit 3 geometric parameters to Ia and Ic of each of 4 isotopologues 3. 185(11) Å o 87. 20(8) 68. 7(5)o 2. 7214(2) Å rms = 0. 0410 u Å2
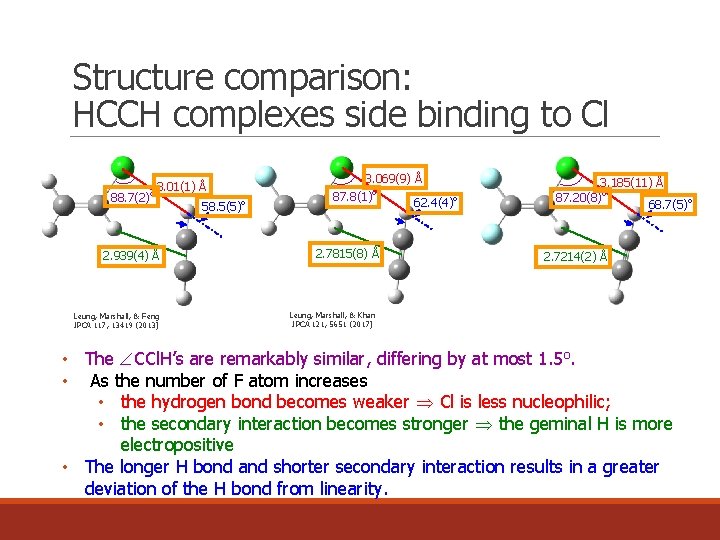
Structure comparison: HCCH complexes side binding to Cl o 88. 7(2) 3. 01(1) Å 2. 939(4) Å Leung, Marshall, & Feng JPCA 117, 13419 (2013) 58. 5(5)o 3. 069(9) Å o 87. 8(1) 2. 7815(8) Å 62. 4(4)o 3. 185(11) Å 87. 20(8)o 68. 7(5)o 2. 7214(2) Å Leung, Marshall, & Khan JPCA 121, 5651 (2017) • The CCl. H’s are remarkably similar, differing by at most 1. 5 o. • As the number of F atom increases • the hydrogen bond becomes weaker Cl is less nucleophilic; • the secondary interaction becomes stronger the geminal H is more electropositive • The longer H bond and shorter secondary interaction results in a greater deviation of the H bond from linearity.

Summary • The rotational spectra of the HCCH complexes of cis-CHFCHF and CF 2 CHCl have been observed analyzed. • HCCH binds to a geminal H, F pair in cis-CHFCHF • the preference for the geminal H, Cl pair in (Z)-CHFCHCl suggests that steric factors outweigh electrostatic factors when both F and Cl are available in ethylene for side binding modes. • HCCH binds to a geminal H, Cl pair instead of a vicinal H, F pair in CF 2 CHCl • the electron distribution about Cl makes the side binding configuration sterically more favorable, and the stability outweighs the stronger electrostatic interactions with F.
- Slides: 15