Tim Murphy UBC Nov 3 2017 Databinge tech

Tim Murphy UBC Nov. 3, 2017 Databinge tech talks Optogenetics The "far-fetched" possibility of using light for selectively controlling precise neural activity (action potential) patterns within subtypes of cells in the brain was thought of by Francis Crick in his Kuffler Lectures at the University of California in San Diego in 1999. Crick, F. (December 1999). "The impact of molecular biology on neuroscience". Philosophical Transactions of the Royal Society B. 354 (1392): 2021– 25. PMC 1692710 reely accessible. PMID 10670022. doi: 10. 1098/rstb. 1999. 0541.

Discovery of phototaxis Deisseroth Nature Neuroscience 2015

Chlamydomonas unicellular phototaxic algae • Chlamydomonas is a genus of green algae consisting of unicellular flagellates, found in stagnant water One of the many striking features of Chlamydomonas is that it contains ion channels, (channelrhodopsins), that are directly activated by light. https: //en. wikipedia. org/wiki/Chlamydomonas
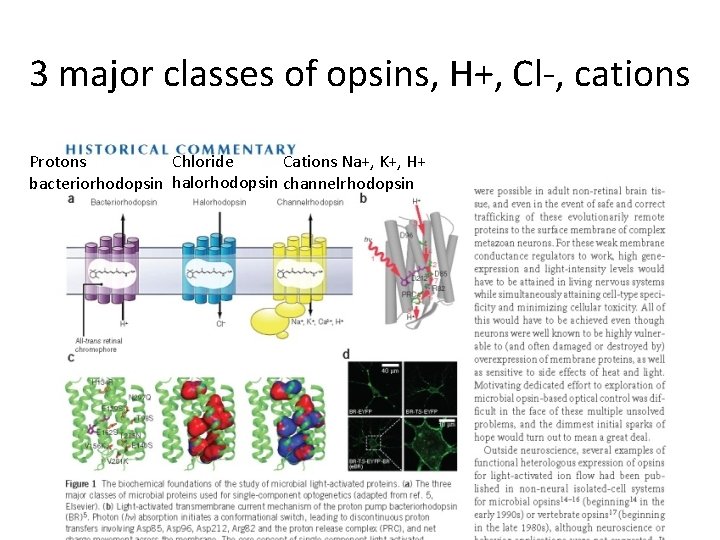
3 major classes of opsins, H+, Cl-, cations Chloride Cations Na+, K+, H+ Protons bacteriorhodopsin halorhodopsin channelrhodopsin
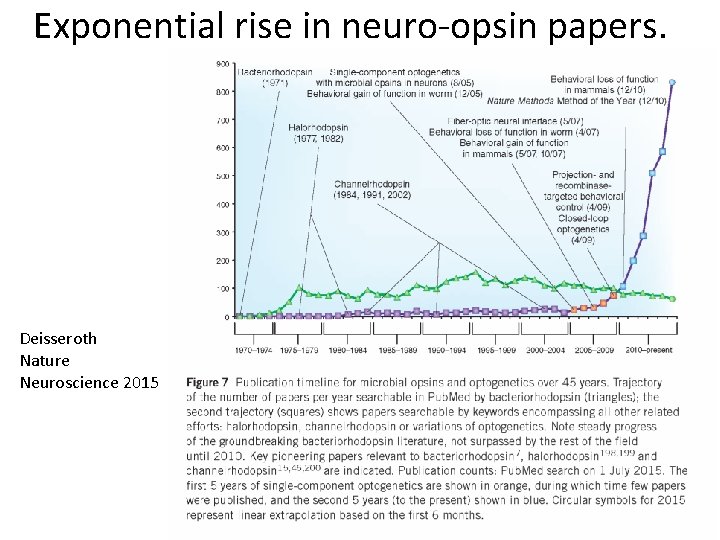
Exponential rise in neuro-opsin papers. Deisseroth Nature Neuroscience 2015
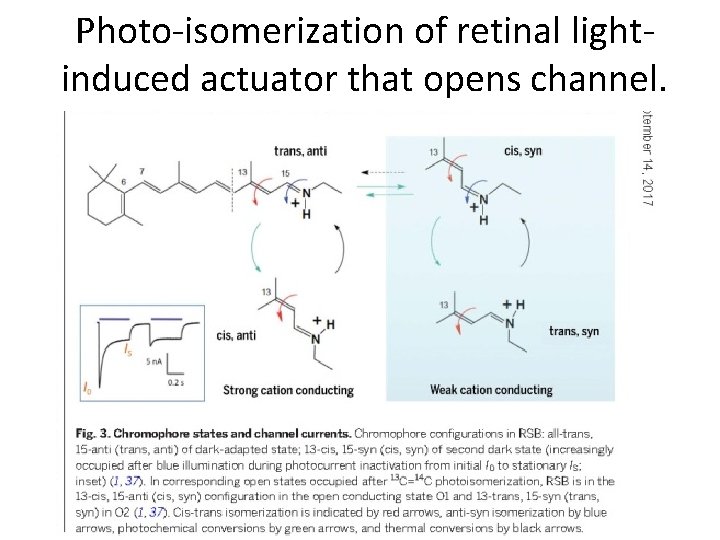
Photo-isomerization of retinal lightinduced actuator that opens channel.
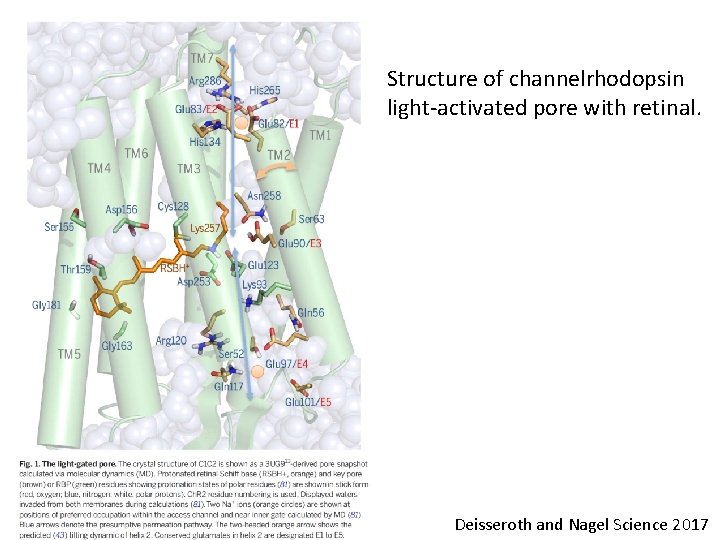
Structure of channelrhodopsin light-activated pore with retinal. Deisseroth and Nagel Science 2017

Chemogenetics • The term chemogenetics has been used to describe the processes by which macromolecules can be engineered to interact with previously unrecognized small molecules has been around for 25 years or more only recently involved neuroscience. • DREADDs, Designer Receptor Exclusively Activated by Designer Drugs. Roth • For reference Chemogenetics as a term was originally coined to describe the observed effects of mutations on chalcone isomerase activity on substrate specificities in the flowers of Dianthus caryophyllus. [1] This Method is very similar to Optogenetics however, it uses chemically engineered molecules and ligands instead of light and light-sensitive channels known as Opsins. https: //en. wikipedia. org/wiki/Chemogenetics

Roth Neuron 2016 The Deisseroth/Sudhof Neuronal Chemogenetics
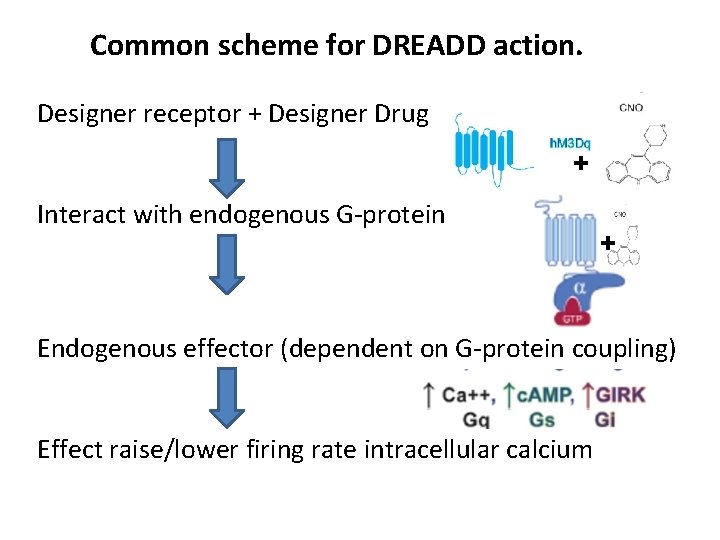
Common scheme for DREADD action. Designer receptor + Designer Drug + Interact with endogenous G-protein + Endogenous effector (dependent on G-protein coupling) Effect raise/lower firing rate intracellular calcium

Naming DREADDs and controls, what is significance of the controls? h. M 3 D(Gs)= human, muscarinic 3, DREADD, Gs h. M 4 D(Gi)= human, muscarinic 4, DREADD, Gi Also known as h. M 4 Di
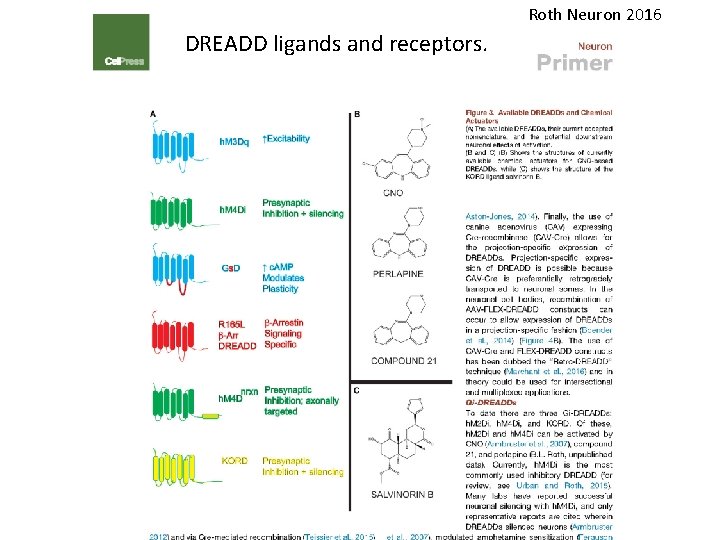
Roth Neuron 2016 DREADD ligands and receptors.
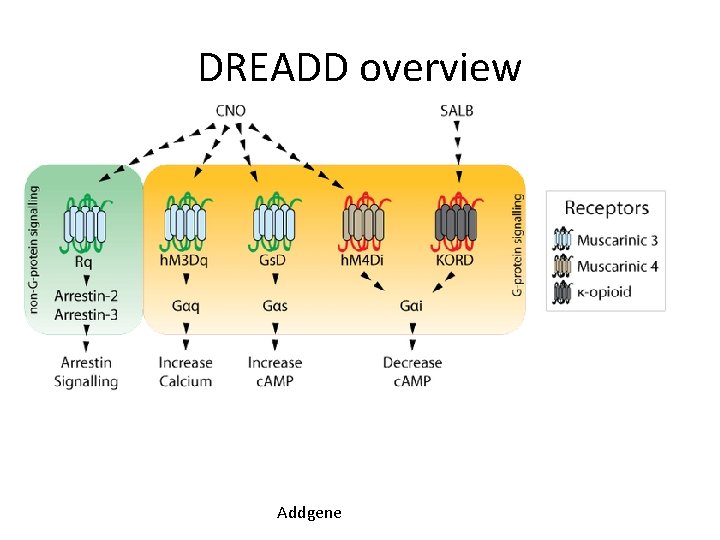
DREADD overview Addgene
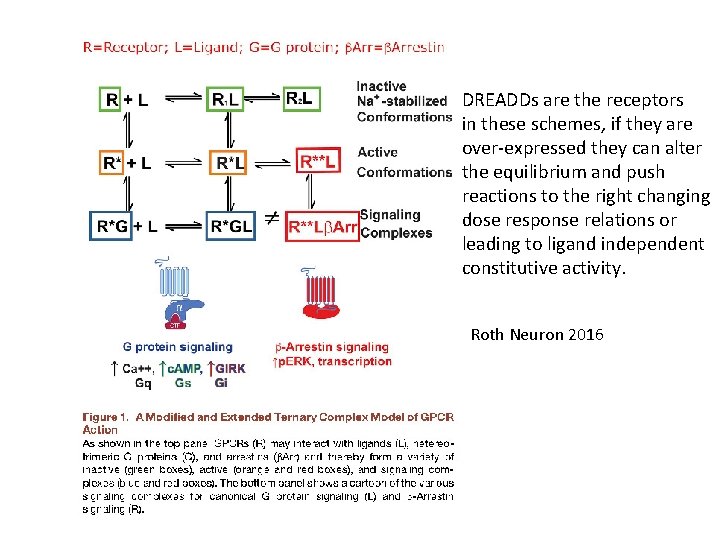
DREADDs are the receptors in these schemes, if they are over-expressed they can alter the equilibrium and push reactions to the right changing dose response relations or leading to ligand independent constitutive activity. Roth Neuron 2016
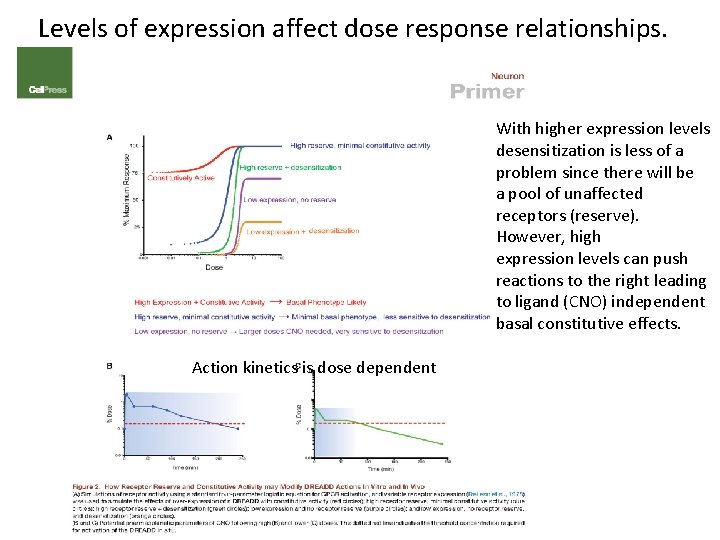
Levels of expression affect dose response relationships. With higher expression levels desensitization is less of a problem since there will be a pool of unaffected receptors (reserve). However, high expression levels can push reactions to the right leading to ligand (CNO) independent basal constitutive effects. Action kinetics is dose dependent
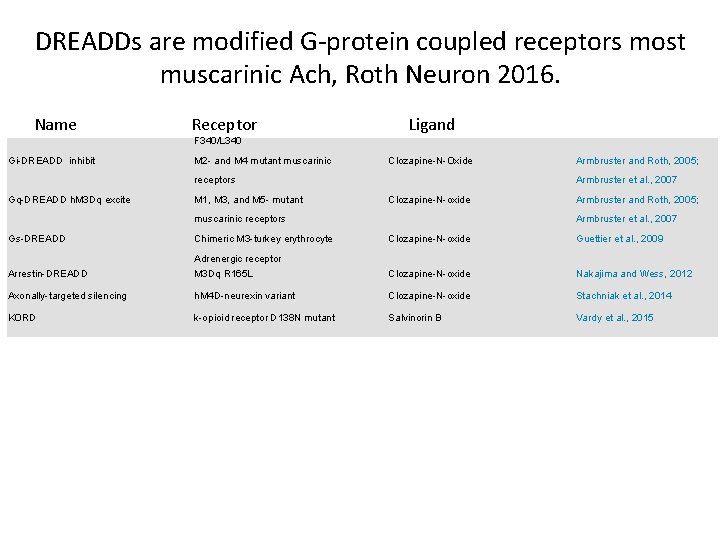
DREADDs are modified G-protein coupled receptors most muscarinic Ach, Roth Neuron 2016. Name Receptor Ligand F 340/L 340 Gi-DREADD inhibit M 2 - and M 4 mutant muscarinic Clozapine-N-Oxide receptors Gq-DREADD h. M 3 Dq excite M 1, M 3, and M 5 - mutant Armbruster et al. , 2007 Clozapine-N-oxide muscarinic receptors Gs-DREADD Chimeric M 3 -turkey erythrocyte Armbruster and Roth, 2005; Armbruster et al. , 2007 Clozapine-N-oxide Guettier et al. , 2009 Adrenergic receptor Arrestin-DREADD M 3 Dq R 165 L Clozapine-N-oxide Nakajima and Wess, 2012 Axonally-targeted silencing h. M 4 D-neurexin variant Clozapine-N-oxide Stachniak et al. , 2014 KORD k-opioid receptor D 138 N mutant Salvinorin B Vardy et al. , 2015
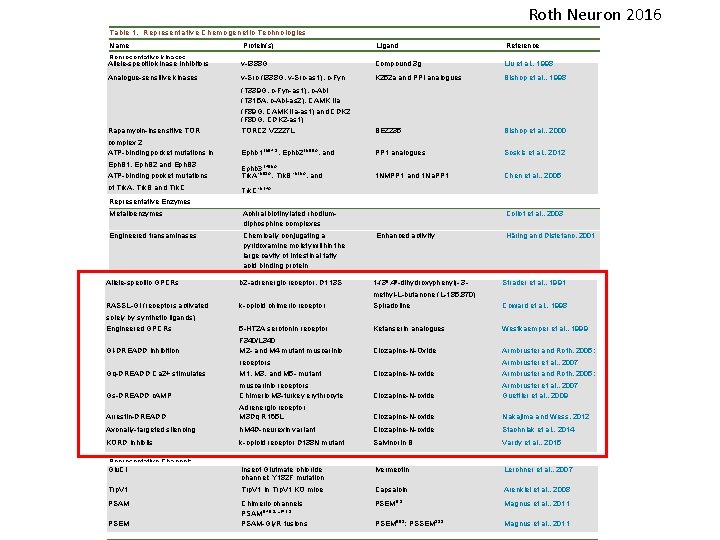
Roth Neuron 2016 Table 1. Representative Chemogenetic Technologies Name Protein(s) Ligand Reference Representative kinases Allele-specific kinase inhibitors v-I 388 G Compound 3 g Liu et al. , 1998 Analogue-sensitive kinases v-Src (I 338 G, v-Src-as 1), c-Fyn K 252 a and PPI analogues Bishop et al. , 1998 Rapamycin-insensitive TOR (T 339 G, c-Fyn-as 1), c-Abl (T 315 A, c-Abl-as 2), CAMK IIa (F 89 G, CAMK IIa-as 1) and CDK 2 (F 80 G, CDK 2 -as 1) TORC 2 V 2227 L BEZ 235 Bishop et al. , 2000 complex 2 ATP-binding pocket mutations in Ephb 1 T 697 G, Ephb 2 T 699 A, and PP 1 analogues Soskis et al. , 2012 Eph. B 1, Eph. B 2 and Eph. B 3 ATP-binding pocket mutations Ephb 3 T 706 A Trk. AF 592 A, Trk. BF 616 A, and 1 NMPP 1 and 1 Na. PP 1 Chen et al. , 2005 of Trk. A, Trk. B and Trk. C F 617 A Trk. C Representative Enzymes Metalloenzymes Achiral biotinylated rhodium- diphosphine complexes Engineered transaminases Chemically conjugating a pyridoxamine moiety within the large cavity of intestinal fatty acid binding protein Representative GPCRs Allele-specific GPCRs Collot et al. , 2003 Enhanced activity Ha¨ ring and Distefano, 2001 b 2 -adrenergic receptor, D 113 S 1 -(30 , 40 -dihydroxyphenyl)- 3 - Strader et al. , 1991 RASSL-Gi (receptors activated k-opioid chimeric receptor methyl-L-butanone (L-185, 870) Spiradoline Coward et al. , 1998 solely by synthetic ligands) Engineered GPCRs 5 -HT 2 A serotonin receptor Ketanserin analogues Westkaemper et al. , 1999 Gi-DREADD inhibition F 340/L 340 M 2 - and M 4 mutant muscarinic Clozapine-N-Oxide Armbruster and Roth, 2005; Gq-DREADD Ca 2+ stimulates receptors M 1, M 3, and M 5 - mutant Clozapine-N-oxide Armbruster et al. , 2007 Armbruster and Roth, 2005; Gs-DREADD c. AMP muscarinic receptors Chimeric M 3 -turkey erythrocyte Clozapine-N-oxide Armbruster et al. , 2007 Guettier et al. , 2009 Arrestin-DREADD Adrenergic receptor M 3 Dq R 165 L Clozapine-N-oxide Nakajima and Wess, 2012 Axonally-targeted silencing h. M 4 D-neurexin variant Clozapine-N-oxide Stachniak et al. , 2014 KORD inhibits k-opioid receptor D 138 N mutant Salvinorin B Vardy et al. , 2015 Insect Glutmate chloride channel; Y 182 F mutation Ivermectin Lerchner et al. , 2007 Trp. V 1 in Trp. V 1 KO mice Capsaicin Arenkiel et al. , 2008 Representative Channels Glu. Cl Trp. V 1 PSAM PSEM Chimeric channels PSAMQ 79 G, L 141 S PSAM-Gly. R fusions PSEM 9 S PSEM 89 S; PSSEM 22 S Magnus et al. , 2011

Inhibitory coupling DREADDs Gi • “Gi-DREADDs to date there are three Gi-DREADDs: h. M 2 Di, h. M 4 Di, and KORD. Of these, h. M 2 Di and h. M 4 Di can be activated by CNO (Armbruster et al. , 2007), compound 21, and perlapine (B. L. Roth, unpublished data). Currently, h. M 4 Di is the most commonly used inhibitory DREADD (for review, see Urban and Roth, 2015). Many labs have reported successful neuronal silencing with h. M 4 Di, and only representative reports are cited wherein DREADDs silenced neurons (Armburster et al. 2012). ” h. M 2 and h. M 4 are human muscarinic receptors M 2 and M 4 • From Roth 2016 Neuron
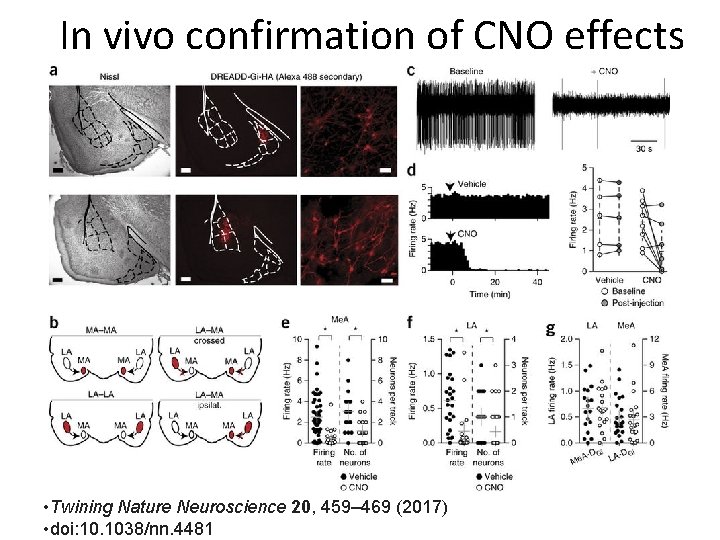
In vivo confirmation of CNO effects • Twining Nature Neuroscience 20, 459– 469 (2017) • doi: 10. 1038/nn. 4481

Excitatory coupling DREADDs Gq “When the original DREADDs were invented, three Gq-coupled DREADDs were created, each of which was based on a different human muscarinic receptor m 1, m 3 and m 5: h. M 1 Dq, h. M 3 Dq, and h. M 5 Dq (Armbruster et al. , 2007). All three Gq-DREADDs are activated by low n. M concentrations of CNO and mobilize intracellular calcium (Armbruster et al. , 2007). All three Gq. DREADDs could be used as excitatory DREADDs, although h. M 3 Dq appears to be most frequently used. ” Roth Neuron 2016 How is mobilization of intracellular calcium linked to excitation of neurons? Can we be certain this will happen in all neurons expressing this DREADD?
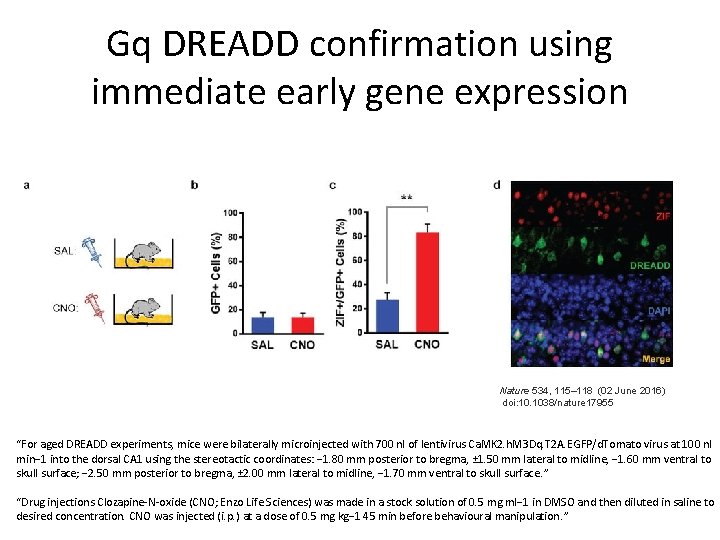
Gq DREADD confirmation using immediate early gene expression Nature 534, 115– 118 (02 June 2016) doi: 10. 1038/nature 17955 “For aged DREADD experiments, mice were bilaterally microinjected with 700 nl of lentivirus Ca. MK 2. h. M 3 Dq. T 2 A. EGFP/d. Tomato virus at 100 nl min− 1 into the dorsal CA 1 using the stereotactic coordinates: − 1. 80 mm posterior to bregma, ± 1. 50 mm lateral to midline, − 1. 60 mm ventral to skull surface; − 2. 50 mm posterior to bregma, ± 2. 00 mm lateral to midline, − 1. 70 mm ventral to skull surface. ” “Drug injections Clozapine-N-oxide (CNO; Enzo Life Sciences) was made in a stock solution of 0. 5 mg ml− 1 in DMSO and then diluted in saline to desired concentration. CNO was injected (i. p. ) at a dose of 0. 5 mg kg− 1 45 min before behavioural manipulation. ”

Controls off-target actions • “CNO may be metabolized via back-transformation to clozapine—especially in guinea pigs, humans (Jann et al. , 1994), and nonhuman primates (unpublished observations). ” not suitable for human • “care should be taken to ensure that clozapine-like sideeffects (e. g. , hypotension, sedation, and anticholinergic syndrome) do not occur by keeping the dose as small as possible and by always performing appropriate controls (e. g. , administering CNO to animals expressing GFP or similarly irrelevant protein). ” Roth 2016 Neuron • Use same construct that does not express DREADD and dose accordingly with CNO.

Schemes for DREADD expression and action.
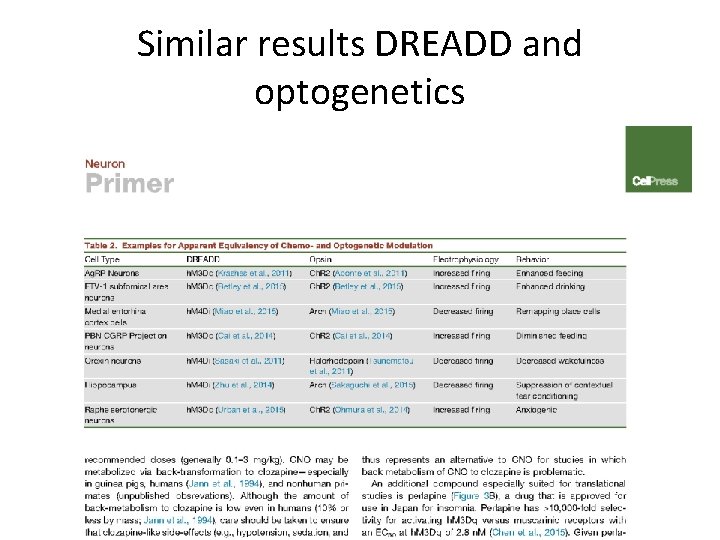
Similar results DREADD and optogenetics

Gomez et al. 2017 Science

CNO converts to clozapine in rat brain CNO can convert to clozapine in vivo (6– 12). Therefore, to demonstrate that systemic CNO injections lead to (i) clozapine conversion and (ii) DREADD occupancy via converted clozapine, we injected rats expressing h. M 4 Di and GFP in the right and left motor cortex, respectively, with intravenous [11 C]CNO (~2 to 4 nmol/kg) and harvested their blood and brain ~35 to 40 min later. In extracts from brain hemispheres receiving AAV-GFP, [11 C]CNO accounted for ~76% of radioactivity detected (other metabolites accounted for ~24%), and no [11 C]clozapine could be detected (Fig. 2, N and P). In extracts from brain hemispheres receiving AAV-h. M 4 Di, [11 C]CNO accounted for ~44%, whereas [11 C]clozapine accounted for ~31% of total radioactivity detected (other metabolites accounted for ~25%) (Fig. 2, O and P). Gomez et al. 2017 Science
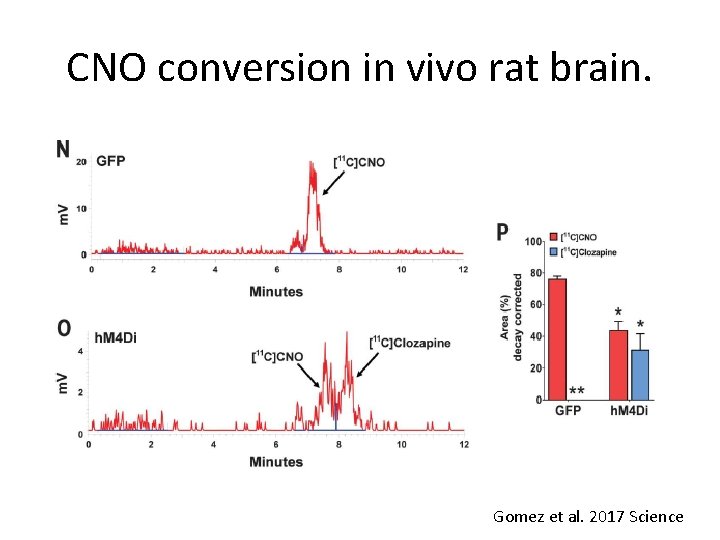
CNO conversion in vivo rat brain. Gomez et al. 2017 Science

Issues with CNO conversion • Clozapine 1000 x higher affinity than CNO for DREADDs, clozapine crosses BBB better. • Process of conversion is slow prolonging effects making behavioral assessment more difficult, does not convert much in rodent blood. • Strangely conversion dependent on expression of DREADD • However, all is not bad since affinities at DREADDs is higher could use clozapine alone.
From Roth 2016 Neuron “Because of the potential for back-metabolism of CNO to clozapine and other clozapine metabolites in non-rodent species—including the pharmacologically diverse compound Ndesmethyl-clozapine (NDMC) (Davies et al. , 2005)—we have developed new non-CNO chemical actuators (Chen et al. , 2015). The first of these—Compound 21 (Figure 3 B)—has minimal off-target activity and exquisite selectivity for activating h. M 3 Dq versus muscarinic and other GPCRs (Chen et al. , 2015). Preliminary studies indicate that Compound 21 has equivalent potency in studies in vivo when compared with CNO (unpublished data). Compound 21 likely cannot be metabolized via normal routes to clozapine or any related compound”
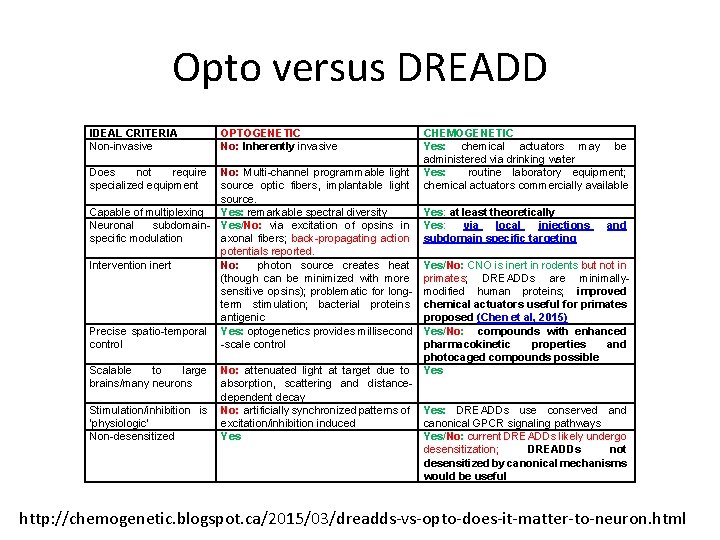
Opto versus DREADD IDEAL CRITERIA Non-invasive Does not require specialized equipment Capable of multiplexing Neuronal subdomainspecific modulation Intervention inert Precise spatio-temporal control Scalable to large brains/many neurons Stimulation/inhibition is ‘physiologic’ Non-desensitized OPTOGENETIC No: Inherently invasive CHEMOGENETIC Yes: chemical actuators may be administered via drinking water No: Multi-channel programmable light Yes: routine laboratory equipment; source optic fibers, implantable light chemical actuators commercially available source. Yes: remarkable spectral diversity Yes: at least theoretically Yes/No: via excitation of opsins in Yes: via local injections and axonal fibers; back-propagating action subdomain specific targeting potentials reported. No: photon source creates heat Yes/No: CNO is inert in rodents but not in (though can be minimized with more primates; DREADDs are minimallysensitive opsins); problematic for long- modified human proteins; improved term stimulation; bacterial proteins chemical actuators useful for primates antigenic proposed (Chen et al, 2015) Yes: optogenetics provides millisecond Yes/No: compounds with enhanced -scale control pharmacokinetic properties and photocaged compounds possible No: attenuated light at target due to Yes absorption, scattering and distancedependent decay No: artificially synchronized patterns of Yes: DREADDs use conserved and excitation/inhibition induced canonical GPCR signaling pathways Yes/No: current DREADDs likely undergo desensitization; DREADDs not desensitized by canonical mechanisms would be useful http: //chemogenetic. blogspot. ca/2015/03/dreadds-vs-opto-does-it-matter-to-neuron. html

Remaining issues with DREADDs • Uncertain about what action DREADDs will ultimately have on neurons? • Metabolism of CNO in rodent in vivo, Gomez et al. Science? • What are controls that are needed for each?

Readings: • Roth DREADDs for Neuroscientists Neuron 2016 • Deisseroth Optogenetics: 10 years of microbial opsins in neuroscience Nature Neuroscience 2015
- Slides: 32