TIDESACS trial Comparison of TItaniumnitrideoxide coated bioactive stent

TIDES-ACS trial Comparison of TItanium-nitride-oxide coated bioactive stent to the Drug (everolimus)-Eluting Stent in Acute Coronary Syndrome Pasi P Karjalainen

Potential conflicts of interest Speaker's name: Pasi P Karjalainen I have the following potential conflicts of interest to report: Receipt of grants / research supports: Abbott Receipt of honoraria or consultation fees: Astra. Zeneca Biotronik Boston Scientific Medtronic St. Jude Medical
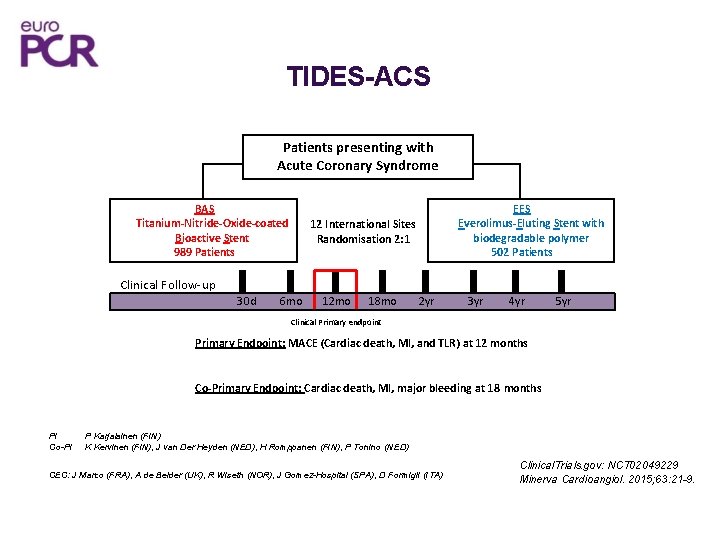
TIDES-ACS Patients presenting with Acute Coronary Syndrome BAS Titanium-Nitride-Oxide-coated Bioactive Stent 989 Patients EES Everolimus-Eluting Stent with biodegradable polymer 502 Patients 12 International Sites Randomisation 2: 1 Clinical Follow-up 30 d 6 mo 12 mo 18 mo 2 yr 3 yr 4 yr 5 yr Clinical Primary endpoint Primary Endpoint: MACE (Cardiac death, MI, and TLR) at 12 months Co-Primary Endpoint: Cardiac death, MI, major bleeding at 18 months PI Co-PI P Karjalainen (FIN) K Kervinen (FIN), J van Der Heyden (NED), H Romppanen (FIN), P Tonino (NED) CEC: J Marco (FRA), A de Belder (UK), R Wiseth (NOR), J Gomez-Hospital (SPA), D Formigli (ITA) Clinical. Trials. gov: NCT 02049229 Minerva Cardioangiol. 2015; 63: 21 -9.
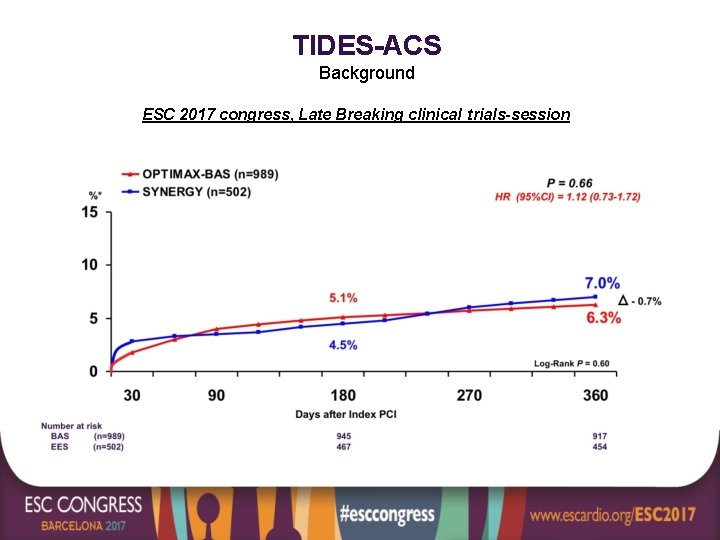
TIDES-ACS Background ESC 2017 congress, Late Breaking clinical trials-session
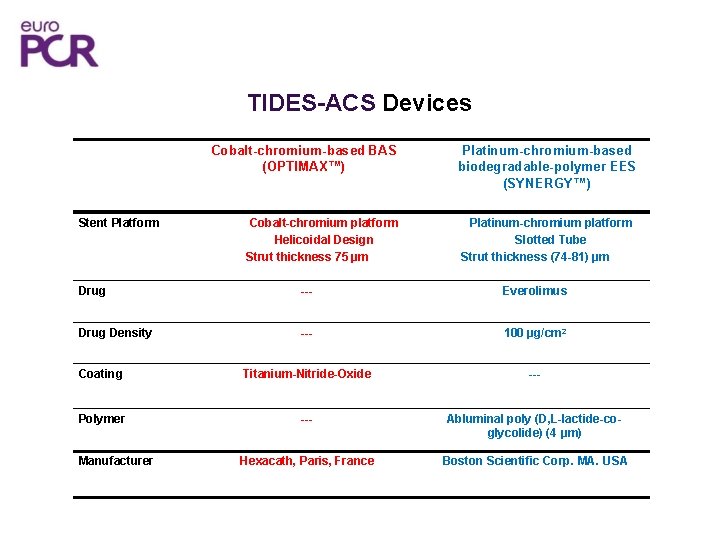
TIDES-ACS Devices Cobalt-chromium-based BAS (OPTIMAX™) Stent Platform Cobalt-chromium platform Helicoidal Design Strut thickness 75 µm Platinum-chromium-based biodegradable-polymer EES (SYNERGY™) Platinum-chromium platform Slotted Tube Strut thickness (74 -81) µm Drug --- Everolimus Drug Density --- 100 µg/cm 2 Coating Titanium-Nitride-Oxide --- Polymer --- Abluminal poly (D, L-lactide-coglycolide) (4 µm) Hexacath, Paris, France Boston Scientific Corp. MA. USA Manufacturer
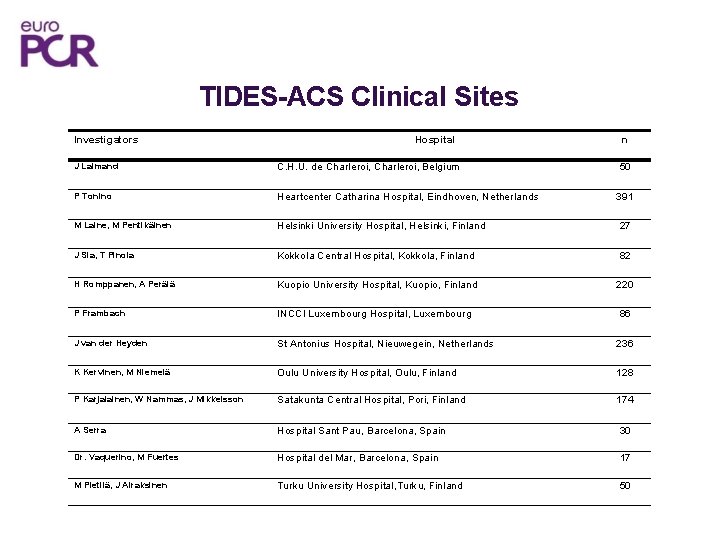
TIDES-ACS Clinical Sites Investigators Hospital n J Lalmand C. H. U. de Charleroi, Belgium 50 P Tonino Heartcenter Catharina Hospital, Eindhoven, Netherlands 391 M Laine, M Pentikäinen Helsinki University Hospital, Helsinki, Finland 27 J Sia, T Pinola Kokkola Central Hospital, Kokkola, Finland 82 H Romppanen, A Perälä Kuopio University Hospital, Kuopio, Finland 220 P Frambach INCCI Luxembourg Hospital, Luxembourg 86 J van der Heyden St Antonius Hospital, Nieuwegein, Netherlands 236 K Kervinen, M Niemelä Oulu University Hospital, Oulu, Finland 128 P Karjalainen, W Nammas, J Mikkelsson Satakunta Central Hospital, Pori, Finland 174 A Serra Hospital Sant Pau, Barcelona, Spain 30 Dr. Vaquerino, M Fuertes Hospital del Mar, Barcelona, Spain 17 M Pietilä, J Airaksinen Turku University Hospital, Turku, Finland 50
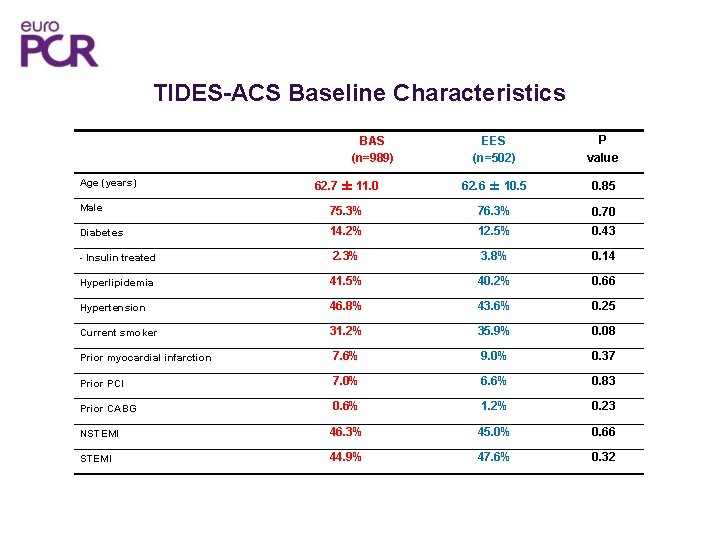
TIDES-ACS Baseline Characteristics EES (n=502) P value 62. 7 ± 11. 0 62. 6 ± 10. 5 0. 85 Male 75. 3% 76. 3% 0. 70 Diabetes 14. 2% 12. 5% 0. 43 - Insulin treated 2. 3% 3. 8% 0. 14 Hyperlipidemia 41. 5% 40. 2% 0. 66 Hypertension 46. 8% 43. 6% 0. 25 Current smoker 31. 2% 35. 9% 0. 08 Prior myocardial infarction 7. 6% 9. 0% 0. 37 Prior PCI 7. 0% 6. 6% 0. 83 Prior CABG 0. 6% 1. 2% 0. 23 NSTEMI 46. 3% 45. 0% 0. 66 STEMI 44. 9% 47. 6% 0. 32 BAS (n=989) Age (years)
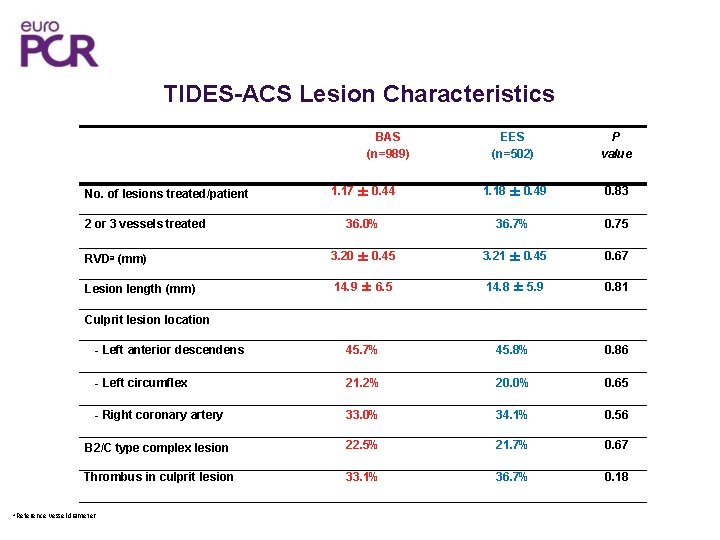
TIDES-ACS Lesion Characteristics EES (n=502) P value 1. 17 ± 0. 44 1. 18 ± 0. 49 0. 83 36. 0% 36. 7% 0. 75 RVDa (mm) 3. 20 ± 0. 45 3. 21 ± 0. 45 0. 67 Lesion length (mm) 14. 9 ± 6. 5 14. 8 ± 5. 9 0. 81 - Left anterior descendens 45. 7% 45. 8% 0. 86 - Left circumflex 21. 2% 20. 0% 0. 65 - Right coronary artery 33. 0% 34. 1% 0. 56 B 2/C type complex lesion 22. 5% 21. 7% 0. 67 Thrombus in culprit lesion 33. 1% 36. 7% 0. 18 BAS (n=989) No. of lesions treated/patient 2 or 3 vessels treated Culprit lesion location a Reference vessel diameter
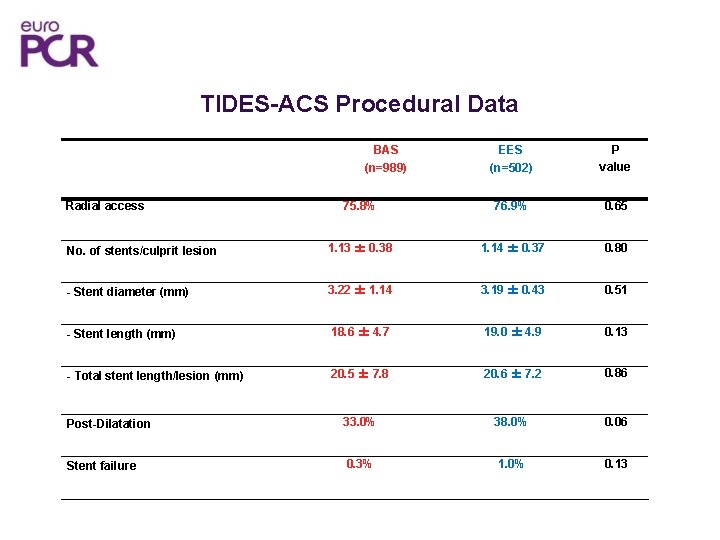
TIDES-ACS Procedural Data EES (n=502) P value 75. 8% 76. 9% 0. 65 No. of stents/culprit lesion 1. 13 ± 0. 38 1. 14 ± 0. 37 0. 80 - Stent diameter (mm) 3. 22 ± 1. 14 3. 19 ± 0. 43 0. 51 - Stent length (mm) 18. 6 ± 4. 7 19. 0 ± 4. 9 0. 13 - Total stent length/lesion (mm) 20. 5 ± 7. 8 20. 6 ± 7. 2 0. 86 Post-Dilatation 33. 0% 38. 0% 0. 06 Stent failure 0. 3% 1. 0% 0. 13 BAS (n=989) Radial access
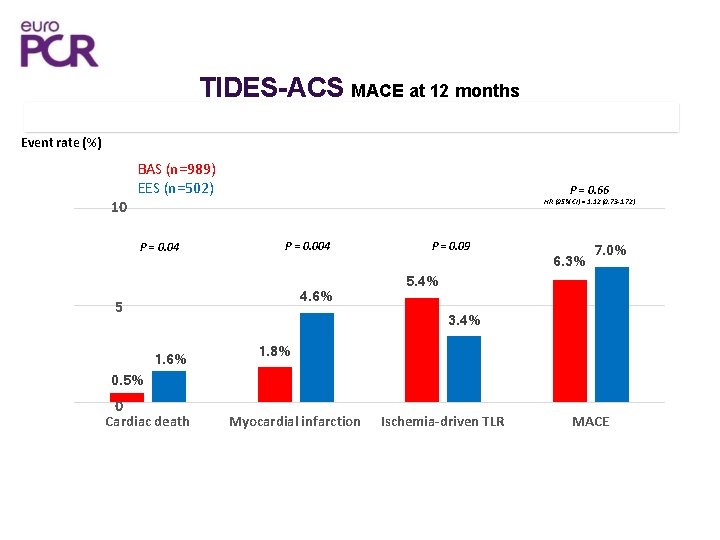
TIDES-ACS MACE at 12 months 15 Event rate (%) 10 BAS (n=989) (989) OPTIMAX-BAS EES (n=502) (502) SYNERGY-EES P = 0. 04 P = 0. 66 HR (95% CI) = 1. 12 (0. 73 -1. 72) P = 0. 004 4. 6% 5 0. 5% 0 P = 0. 09 5. 4% 6. 3% 7. 0% 3. 4% 1. 6% Cardiac death 1. 8% Myocardial infarction Ischemia-driven TLR MACE
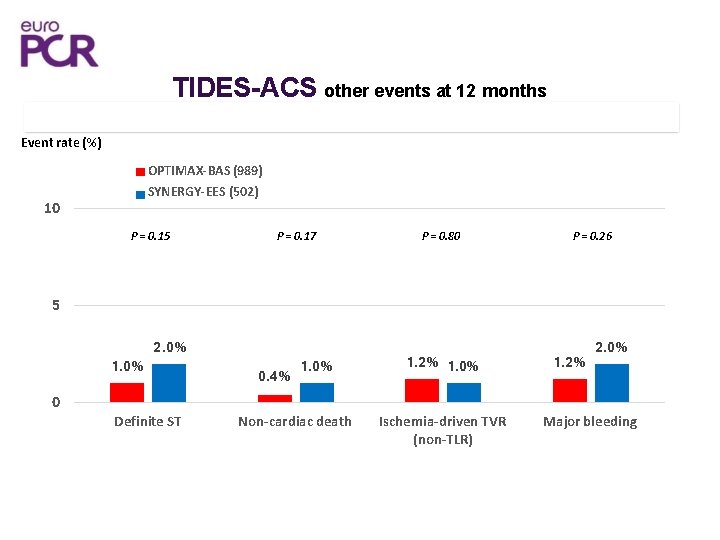
TIDES-ACS other events at 12 months 15 Event rate (%) OPTIMAX-BAS (989) SYNERGY-EES (502) 10 P = 0. 15 P = 0. 17 P = 0. 80 P = 0. 26 5 1. 0% 2. 0% 0. 4% 1. 0% 1. 2% 2. 0% 0 Definite ST Non-cardiac death Ischemia-driven TVR (non-TLR) Major bleeding
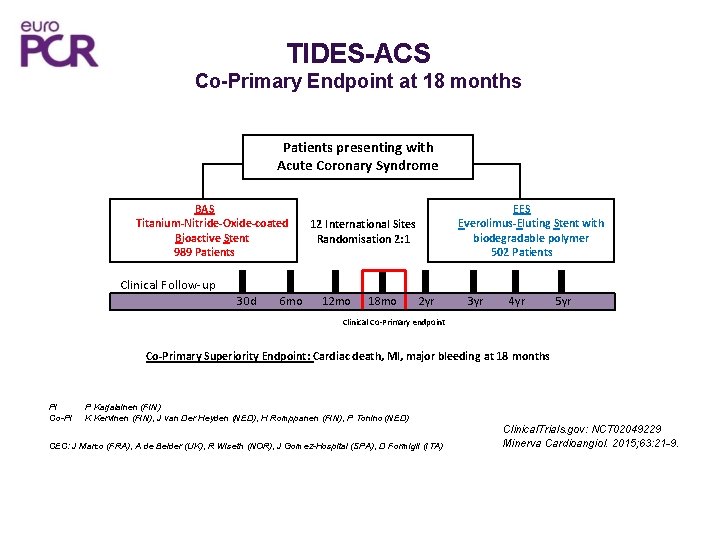
TIDES-ACS Co-Primary Endpoint at 18 months Patients presenting with Acute Coronary Syndrome BAS Titanium-Nitride-Oxide-coated Bioactive Stent 989 Patients EES Everolimus-Eluting Stent with biodegradable polymer 502 Patients 12 International Sites Randomisation 2: 1 Clinical Follow-up 30 d 6 mo 12 mo 18 mo 2 yr 3 yr 4 yr 5 yr Clinical Co-Primary endpoint Co-Primary Superiority Endpoint: Cardiac death, MI, major bleeding at 18 months PI Co-PI P Karjalainen (FIN) K Kervinen (FIN), J van Der Heyden (NED), H Romppanen (FIN), P Tonino (NED) CEC: J Marco (FRA), A de Belder (UK), R Wiseth (NOR), J Gomez-Hospital (SPA), D Formigli (ITA) Clinical. Trials. gov: NCT 02049229 Minerva Cardioangiol. 2015; 63: 21 -9.

TIDES-ACS at 18 months Statistical considerations Co-Primary Superiority Endpoint at 18 months including * Myocardial Infarction (MI) * Cardiac Death * Major Bleeding - Expected event rate in SYNERGY vs. OPTIMAX; 6. 5% vs. 3. 5% * Sample size needed (α=5%; β=80%), N = 1484 (2: 1) TIDES-ACS

Bleeding Academic Research Consortium (BARC) Mehran et al. Circulation 2001; 123: 2736 -47 Type 3 a -Overt bleeding + hemoglobin drop of 3 to <5 g/dl -Any transfusion with overt bleeding Type 3 b - Overt bleeding + hemoglobin drop > 5 g/dl - Cardiac tamponade - Bleeding requiring surgical intervention for control -Bleeding requiring intravenous vasoactive agents Type 3 c - Intracranial hemorrhage - Subcategories confirmed by autopsy or imaging or lumbar puncture - Intraocular bleed compromising vision Type 4: CABG-related bleeding Perioperative intracranial bleeding within 48 h Reoperation after closure of sternotomy for the purpose of controlling bleeding Transfusion of >5 U whole blood or packed red blood cells within 48 -h period Chest tube output >2 L within a 24 -h period Type 5: fatal bleeding Type 5 a - Probable fatal bleeding; no autopsy or imaging confirmation but clinically suspicious Type 5 b - Definite fatal bleeding; overt bleeding or autopsy or imaging confirmation
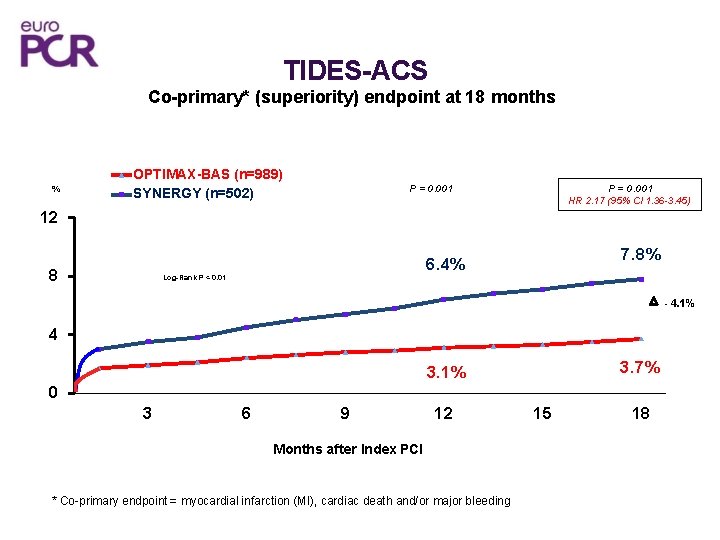
TIDES-ACS Co-primary* (superiority) endpoint at 18 months % OPTIMAX-BAS (n=989) SYNERGY (n=502) P = 0. 001 HR 2. 17 (95% CI 1. 36 -3. 45) 12 7. 8% 6. 4% 8 Log-Rank P < 0. 01 - 4. 1% 4 3. 7% 3. 1% 0 3 6 9 12 Months after Index PCI * Co-primary endpoint = myocardial infarction (MI), cardiac death and/or major bleeding 15 18
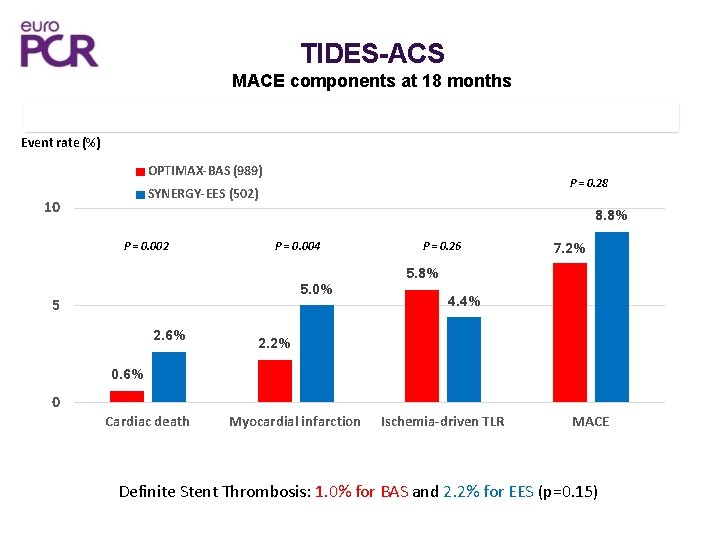
TIDES-ACS MACE components at 18 months 15 Event rate (%) OPTIMAX-BAS (989) P = 0. 28 SYNERGY-EES (502) 10 8. 8% P = 0. 002 P = 0. 004 5. 0% 5 2. 6% P = 0. 26 7. 2% 5. 8% 4. 4% 2. 2% 0. 6% 0 Cardiac death Myocardial infarction Ischemia-driven TLR MACE Definite Stent Thrombosis: 1. 0% for BAS and 2. 2% for EES (p=0. 15)
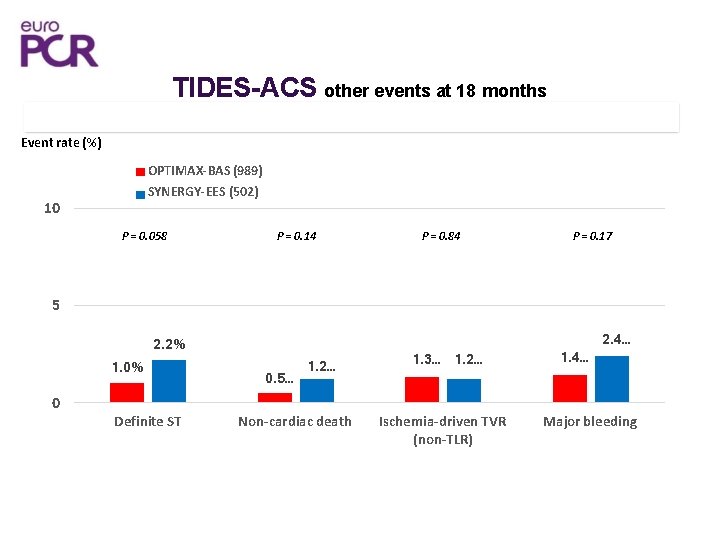
TIDES-ACS other events at 18 months 15 Event rate (%) OPTIMAX-BAS (989) SYNERGY-EES (502) 10 P = 0. 058 P = 0. 14 P = 0. 84 P = 0. 17 5 1. 0% 0 2. 2% Definite ST 0. 5… 1. 2… Non-cardiac death 1. 3… 1. 2… Ischemia-driven TVR (non-TLR) 1. 4… 2. 4… Major bleeding

TIDES-ACS Conclusions • OPTIMAX-BAS was non-inferior to platinum-chromium-based biodegradablepolymer SYNERGY-EES for the primary composite of safety and efficacy outcome (MACE) at 12 -month follow-up. Co-primary superiority endpoint at 18 months (myocardial infarction, cardiac death and major bleeding) showed “better outcome with OPTIMAX-BAS compared with SYNERGY-EES” TIDES-ACS

TIDES-ACS trial Comparison of TItanium-nitride-oxide coated bioactive stent to the Drug (everolimus)-Eluting Stent in Acute Coronary Syndrome ’’Thank you’’
- Slides: 19