THROMBOPHILIA Abdulkareem Almomen MD FRCPC KSUMED 341 17


THROMBOPHILIA Abdulkareem Almomen, MD, FRCPC KSU-MED 341 17 -04 - 2011 (13 -05 -1432)

THROMBOPHILIA Pre-Thrombotic States, Thrombogenic States, Hypercoagulable States

Hemostasis Blood must be fluid Must coagulate (clot) at appropriate time Rapid Localized Reversible Thrombosis = inappropriate coagulation

3 Major systems involved Vessel wall Endothelium (anti-thrombotic) Platelets Coagulation system coagulation factors, natural anticoagulants & fibrinolysis
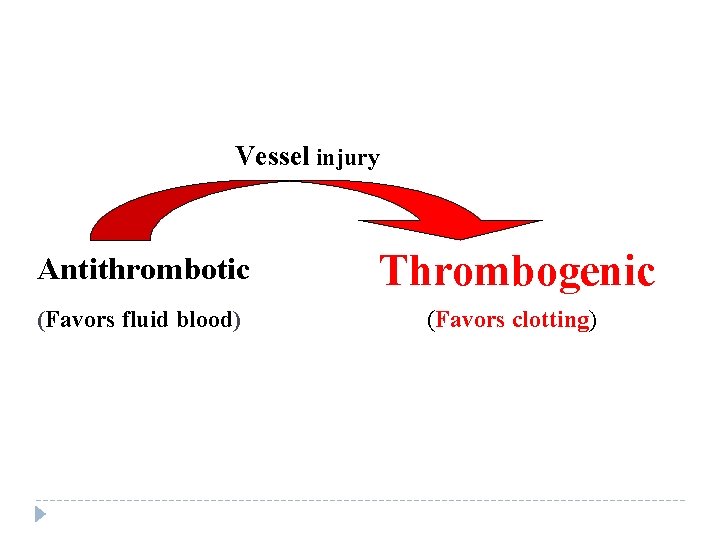
Vessel injury Antithrombotic Thrombogenic (Favors fluid blood) (Favors clotting)

Antithrombotic Properties of the Endothelium • Anti-platelet properties Healthy endothelium does not bind platelets – Produce PGI-2 (prostacyclin) and NO (Nitric Oxide), which inhibit platelet binding – Produce ADP-ase which counters the platelet aggregating effects of ADP

Antithrombotic Properties of the Endothelium (cont. ) Anticoagulant properties Produce Heparin-like proteoglycans which activate anti-thrombin Produce Thrombomodulin which make a complex with thrombin (TM. T complex ) and activates protein C , Produce t. PA which activates fibrinolysis by activating plasminogen to plasmin

Prothrombotic Properties of the Endothelium • Synthesis of von Willebrand factor • Release of tissue factor • Production of plasminogen activator inhibitors (PAI) • Membrane phospholipids bind and facilitate activation of clotting factors via Ca++ bridges
Procoagulant Anticoagulant
Procoagulant Anticoagulant

Virchow’s Triad Pathogenesis of a Thrombus Endothelial injury Abnormal blood flow Hypercoagulability Genetic acquired
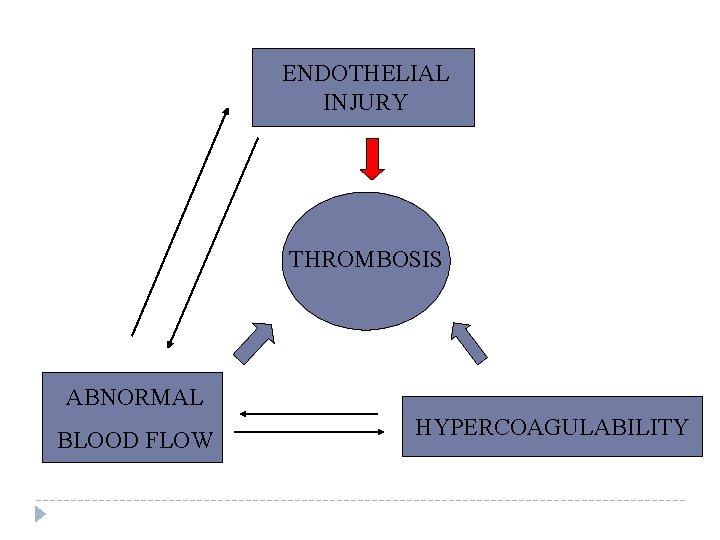
ENDOTHELIAL INJURY THROMBOSIS ABNORMAL BLOOD FLOW HYPERCOAGULABILITY
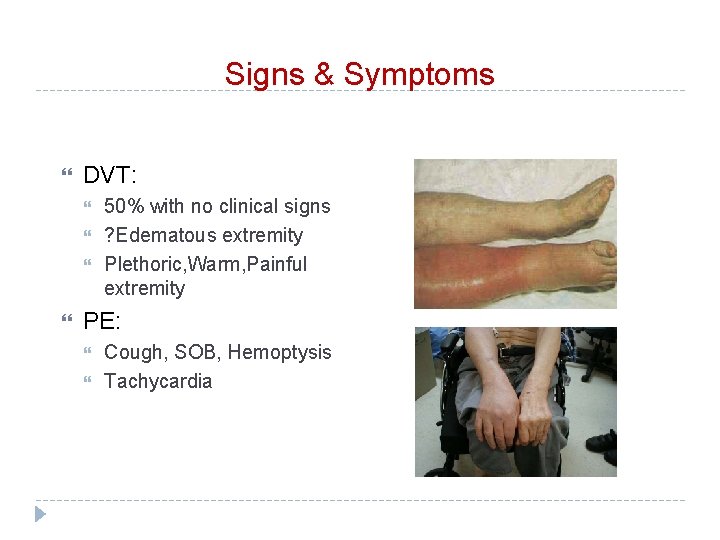
Signs & Symptoms DVT: 50% with no clinical signs ? Edematous extremity Plethoric, Warm, Painful extremity PE: Cough, SOB, Hemoptysis Tachycardia
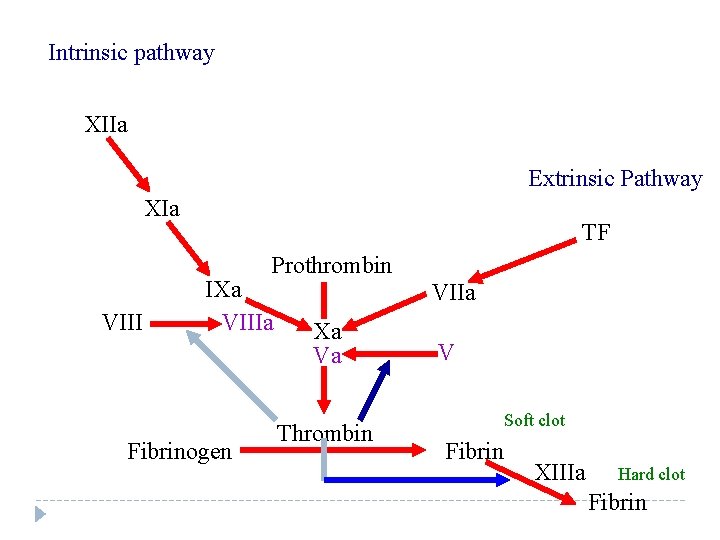
Intrinsic pathway XIIa Extrinsic Pathway XIa TF Prothrombin VIII IXa VIIIa Fibrinogen VIIa Xa Va Thrombin V Soft clot Fibrin XIIIa Hard clot Fibrin

Physiologic Inhibitors of coagulation Antithrombin Activated Protein C + protein S Inactivates Va and VIIIa (via proteolysis) Thrombomodulin Binds to thrombin activate Protein C

Non-physiologic inhibitors of coagulation Vitamin K antagonists (in vivo only) Ca chelators (in vitro only) EDTA Citrate Oxalate * Heparin (in vivo and in vitro)
Clot removal
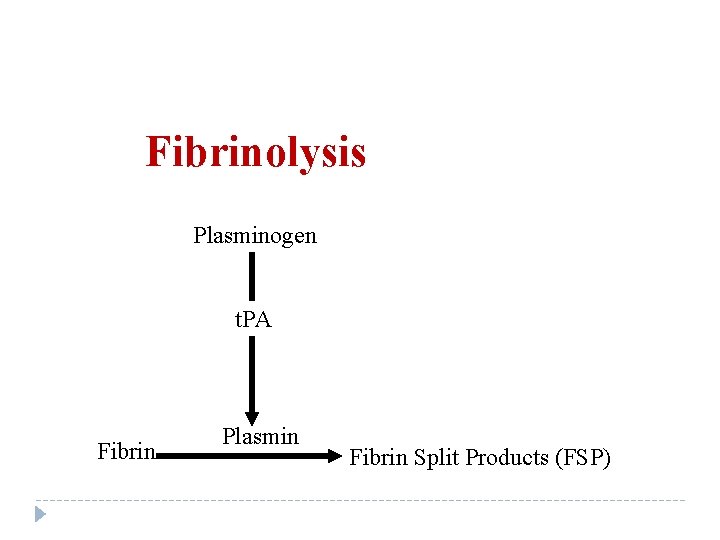
Fibrinolysis Plasminogen t. PA Fibrin Plasmin Fibrin Split Products (FSP)

Inhibitors of fibrinolysis Plasminogen activator inhibitors (PAIs) a 2 -antiplasmin (serpin)
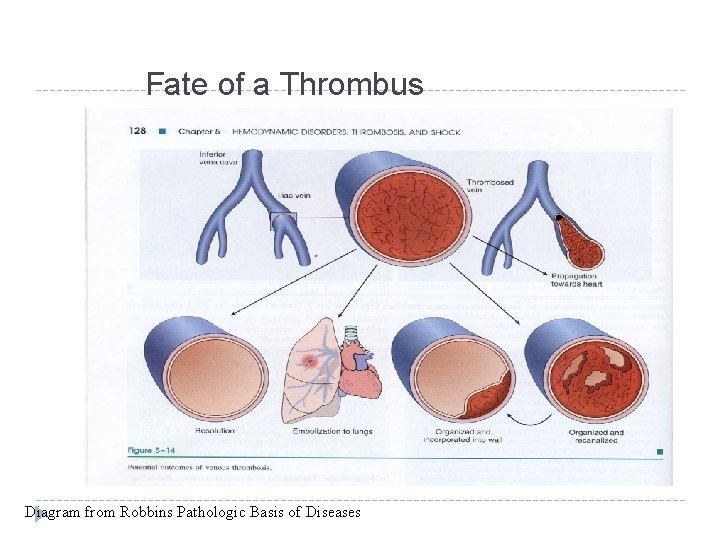
Fate of a Thrombus Diagram from Robbins Pathologic Basis of Diseases

Hereditary Thrombophilias Protein C pathway Factor V Leiden Protein C deficiency Protein S deficiency Prothrombin G 20210 A mutation Antithrombin deficiency Hyperhomocystinemia C 677 T MTHFR mutation

Factor V Leiden Mutation in Factor V Protein C/S complex Impaired anticoagulation 5 -11% of white Europeans Heterozygous Autosomal dominant Homozygous rare
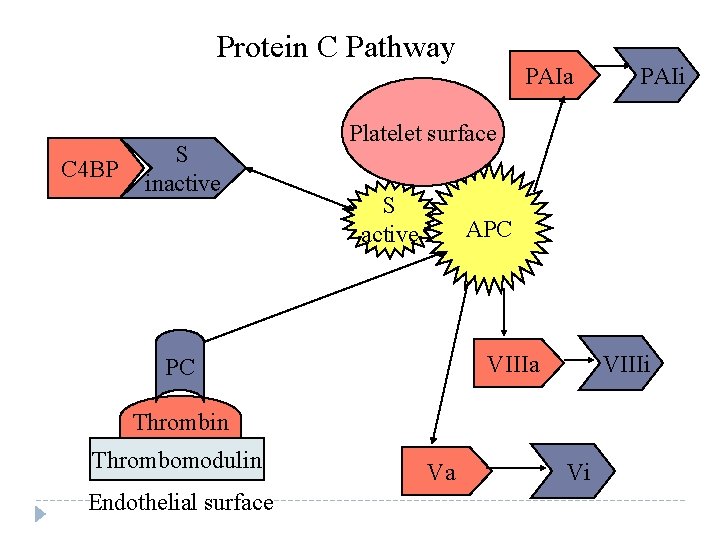
Protein C Pathway C 4 BP S inactive PAIa Platelet surface S active APC VIIIa PC VIIIi Thrombin Thrombomodulin Endothelial surface PAIi Va Vi

Prothrombin G 20210 A mutation Mutation in promotor 150 -200% in prothrombin levels 2 -3% of Europeans Heterozygous autosomal dominant Homozygous similar to Factor V
MTHFR and Thrombosis Hyperhomocysteinemia implicated in both arterial and venous thrombosis Why is homocysteine thrombogenic? Theories: Direct toxicity to endothelial cells Inhibits Protein C activation Promotes endothelial tissue factor expression Surpresses endothelial cell surface heparin sulfate

Hyperhomocysteinemia Atherosclerosis, NTD, thromboembolism Severe – homozygous 1 in 200, 000 -355, 000 Cystathionine -synthase Mild to moderate – Heterozygotes for C S mutation Homozygous for 667 C-T MTHFR (11%)
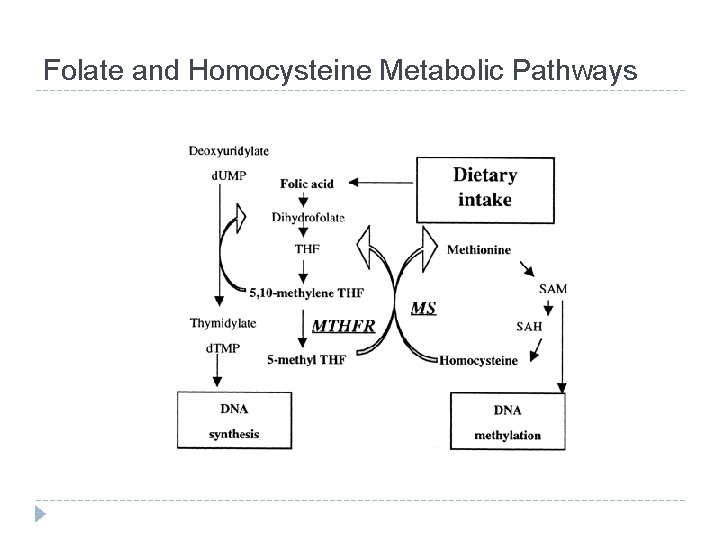
Folate and Homocysteine Metabolic Pathways
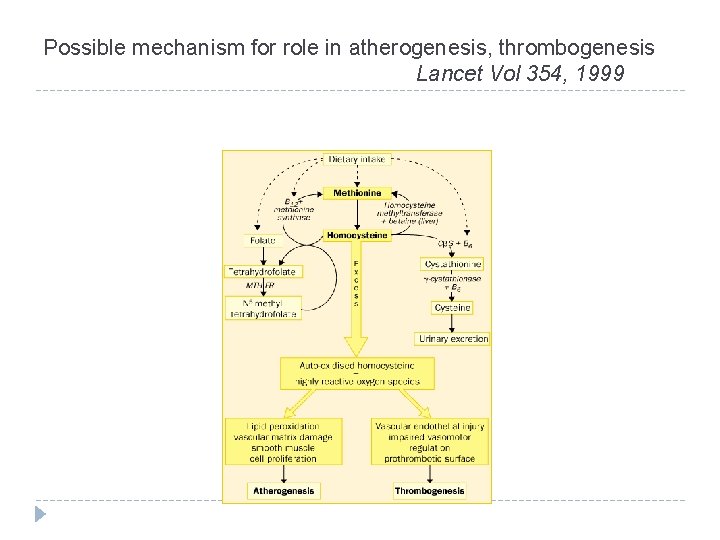
Possible mechanism for role in atherogenesis, thrombogenesis Lancet Vol 354, 1999

AT Deficiency Multiple mutations Most thrombogenic disorder Type I Levels and activity Type II Activity

Protein C / Protein S Deficiencies Protein C deficiency Type I – number and activity Type II – activity Protein S deficiency Type I – total and free forms Type II – cofactor activity Type III - free only Autosomal dominant 0. 2 -0. 5, 0. 8 prevalence
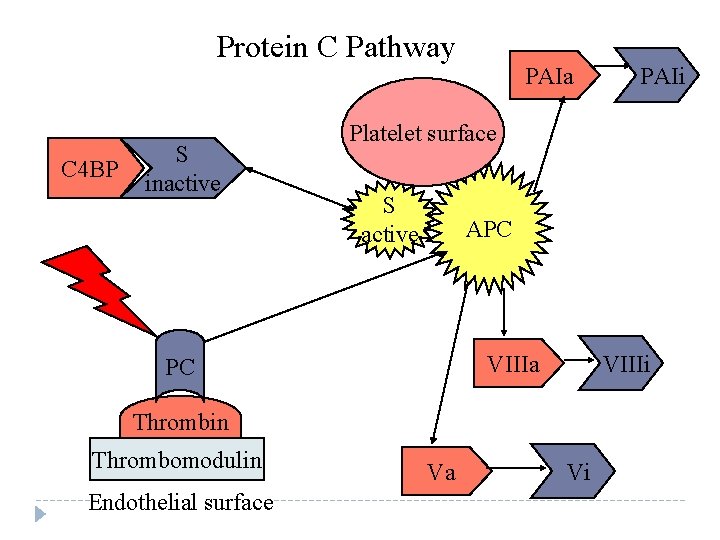
Protein C Pathway C 4 BP S inactive PAIa Platelet surface S active APC VIIIa PC VIIIi Thrombin Thrombomodulin Endothelial surface PAIi Va Vi

Antiphospholipid Antibody Syndrome Autoimmune Acquired Prothrombotic Disorder Very High Risk for recurrent thromboembolic disease both venous and arterial Indefinite duration anticoagulation recommended +/immunosuppression Strict Diagnostic Criteria

Antiphospholipid Syndrome Clinical criteria (≥ 1 must be present): 1. Vascular thrombosis: - ≥ 1 clinical episode of, objectively confirmed, arterial, venous, or small vessel thrombosis 2. Pregnancy morbidity: - ≥ 1 unexplained fetal death @ ≥ 10 weeks EGA - ≥ 1 premature birth (≤ 34 th week of gestation) due to eclampsia, severe pre-eclampsia, or placental insufficiency - ≥ 3 unexplained consecutive spontaneous abortions @ <10 weeks EGA Revised Sapporo/Sydney Criteria. JTH 2006; 4: 295 -306

Antiphospholipid Syndrome Laboratory criteria (≥ 1 must be present): LA (+) ≥ 2 occasions, at least 12 weeks apart, according to ISTH guidelines: prolonged PL-based clotting assay, lack of correction with 1: 1 mix, and correction with excess PL ACLA and/or anti-β 2 glycoprotein-I antibody: medium or high Ig. G and/or Ig. M isotype titer ≥ 2 occasions, at least 12 weeks apart Standardized ELISA assays Revised Sapporo/Sydney Criteria. JTH 2006; 4: 295 -306
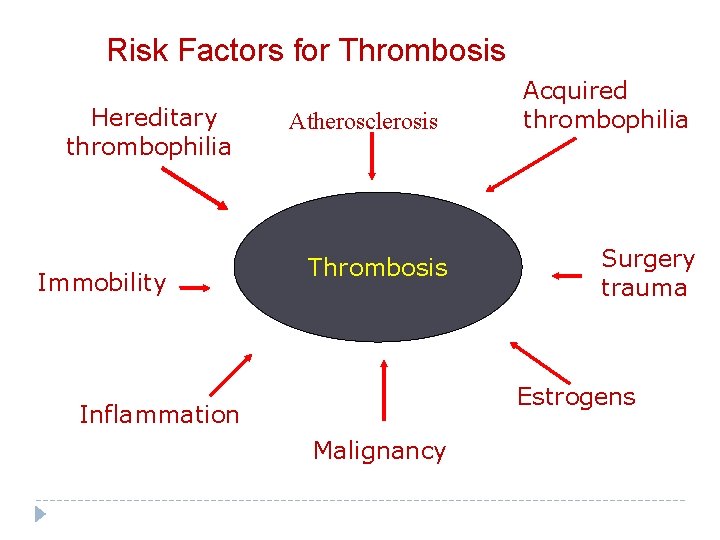
Risk Factors for Thrombosis Hereditary thrombophilia Immobility Atherosclerosis Thrombosis Acquired thrombophilia Surgery trauma Estrogens Inflammation Malignancy

Therapies/Heparin Mechanism: catalysis of AT. Neonates have lower AT levels. Monitoring: a. PTT Problems a. PTT levels based on adult therapeutic studies. Even in adults, therapeutic a. PTT may not suggest clinically sufficient anti-coag.

Therapies/Heparin Recommended dose 75 U/kg loading. Maintenance drip dose varies: Infants <1 yr of age 28 U/kg/hr Children > 1 yr 20 U/kg/hr Side effects (besides bleeding): Heparin induced thrombocytopenia Osteoporosis
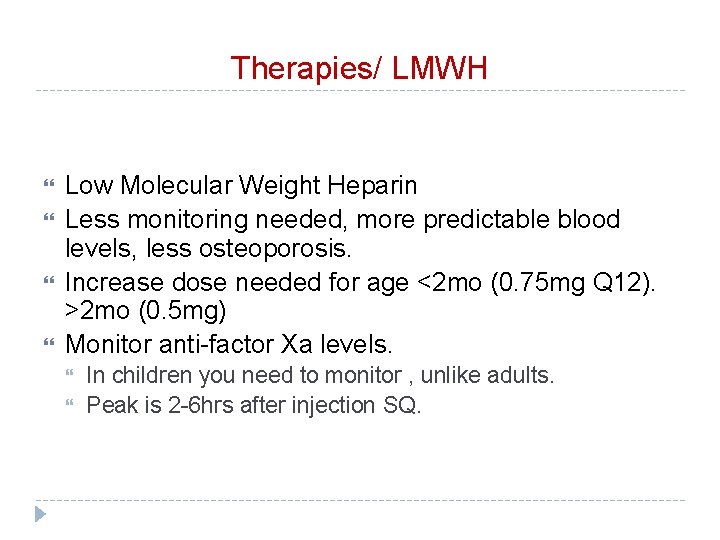
Therapies/ LMWH Low Molecular Weight Heparin Less monitoring needed, more predictable blood levels, less osteoporosis. Increase dose needed for age <2 mo (0. 75 mg Q 12). >2 mo (0. 5 mg) Monitor anti-factor Xa levels. In children you need to monitor , unlike adults. Peak is 2 -6 hrs after injection SQ.
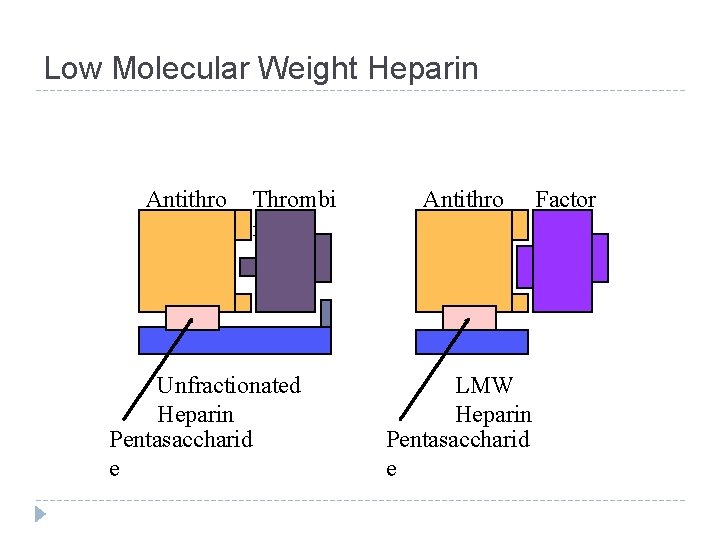
Low Molecular Weight Heparin Antithro mbin Thrombi n Unfractionated Heparin Pentasaccharid e Antithro mbin LMW Heparin Pentasaccharid e Factor Xa

Therapies/Oral-anticoagulants Impairs function of vitamin-K dependent proteins (II, VII, IX, X) plus Proteins C & S. Newborns have reduced levels of vitamin-K dependent proteins. (Shot at birth helps. ) Vitamin K added to formulas. Minimal in breast milk. New anti-coagulants: Direct anti-thrombin (Daqbigatran) Anti-Xa (Rivaroxaban)

Monitoring PTT PT/INR TT (thrombin time) Heparin level Xa activity No monitoring

Anti dotes (overdose) Stop the anti-thrombotic/anti-coagulant agent, Protamin sulfate (heparin) Plasma/ vitamin K (warfarin) Tranexamic acid (thrombolytic therapy, fibrinolysis) DDAVP (anti-platelets) r. FVIIa ( universal anti hemorrhagic) ( dose= 4000 -20000 SR )
- Slides: 43