Threedimensional microfabricated bioreactor and closedloop control system Alex

Three-dimensional microfabricated bioreactor and closed-loop control system Alex Makowski Michael Hwang Jenny Lu Dr. John Wikswo

Ultimate Project Goals l Computer modulated growth of tissue structures within microfluidic devices. l Possible uses include drug testing and dose determination, toxicity levels, and biocompatibility issues.

Problem Statement- Design progress is blocked on three distinct fronts: l l l Sensors are required to provide computer with necessary information. Bioreactor design limits the quantity and quality of cell morphology within the device. Previously used cells (primary human fibroblasts) do not easily form tissue-like structures.

Subsequent Requirements: l l l Choose or design an appropriate sensor for p. H measurements (most needed to determine cell metabolism and health). Choose a cell line that will exhibit observable morphological change under successful conditions. Redesign the bioreactor to incorporate new cell line and maximize efficiency of p. H sensors.
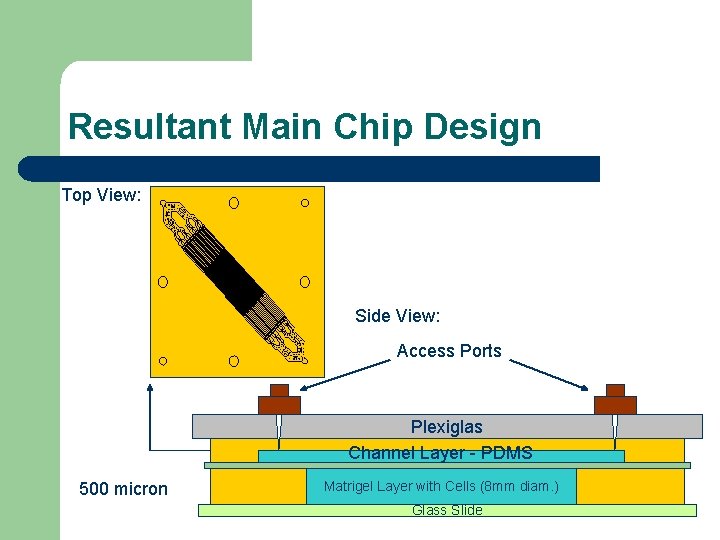
Resultant Main Chip Design Top View: Side View: Access Ports Plexiglas Channel Layer - PDMS 500 micron Matrigel Layer with Cells (8 mm diam. ) Glass Slide
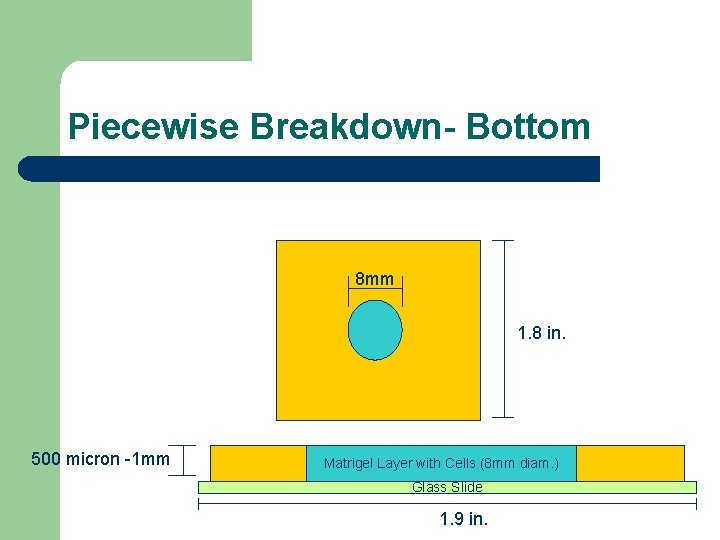
Piecewise Breakdown- Bottom 8 mm 1. 8 in. 500 micron -1 mm Matrigel Layer with Cells (8 mm diam. ) Glass Slide 1. 9 in.

Piecewise Breakdown- Top l l Filter layer – 10 micron thick x 1. 9 inches square (4. 7 micron pores) Channel Layer – Dimensions (1. 9 inch square 3 -5 mm thickness) – Channels: 60 -100 microns deep 6 mm across 15 micron wide channels Plexiglas Layer (machine shop made) – Dimensions (1. 9 inch square 5 -7 mm thick) Port Assemblies-Upchurch Scientific – Interface with Tygon tubing (500 micron) Plexiglas Channel Layer - PDMS

Ultimate Assembly • Final Bioreactor assemblies include: • Brass clamp • Acrylic and Port Assemblies • PDMS Layers (containing cells) Viibre Repository: Schaffer
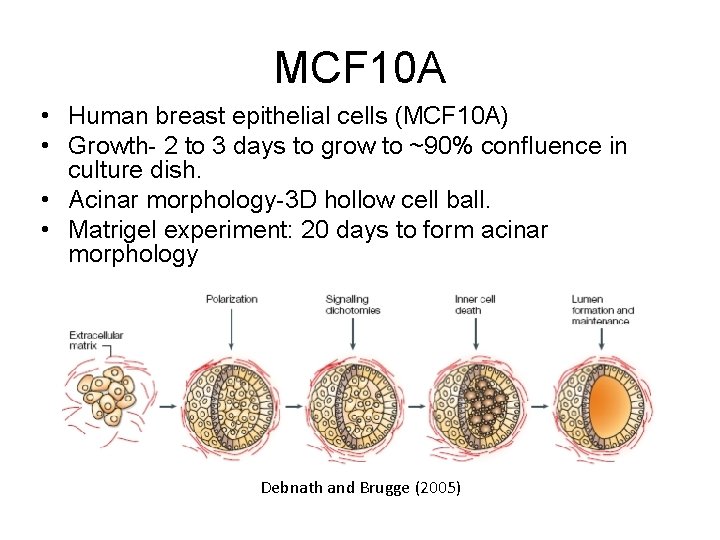
MCF 10 A • Human breast epithelial cells (MCF 10 A) • Growth- 2 to 3 days to grow to ~90% confluence in culture dish. • Acinar morphology-3 D hollow cell ball. • Matrigel experiment: 20 days to form acinar morphology Debnath and Brugge (2005)
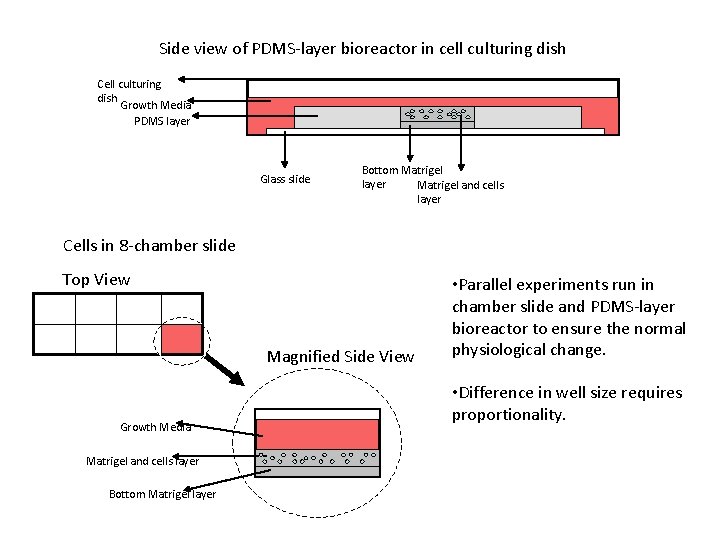
Side view of PDMS-layer bioreactor in cell culturing dish Cell culturing dish Growth Media PDMS layer Glass slide Bottom Matrigel layer Matrigel and cells layer Cells in 8 -chamber slide Top View Magnified Side View Growth Media Matrigel and cells layer Bottom Matrigel layer • Parallel experiments run in chamber slide and PDMS-layer bioreactor to ensure the normal physiological change. • Difference in well size requires proportionality.

Problems and Solutions P 1: Matrigel migrated from the well of bioreactor to the surface of PDMS layer. S 1: Let the second layer of Matrigel with cells sit for 1 hour instead of 20 minutes to polymerize. P 2: Some cells migrated to the surface of PDMS layer and some even to the cell culturing dish. S 2: The final bioreactor has a filter that prevents the cells from migrating out of the well. Current Progress • Cells in the chamber slides grew for more than 20 days to form into acinar morphology • Cells in bioreactors are staying inside of the well and forming spheriods, and they are 3 weeks old and still alive
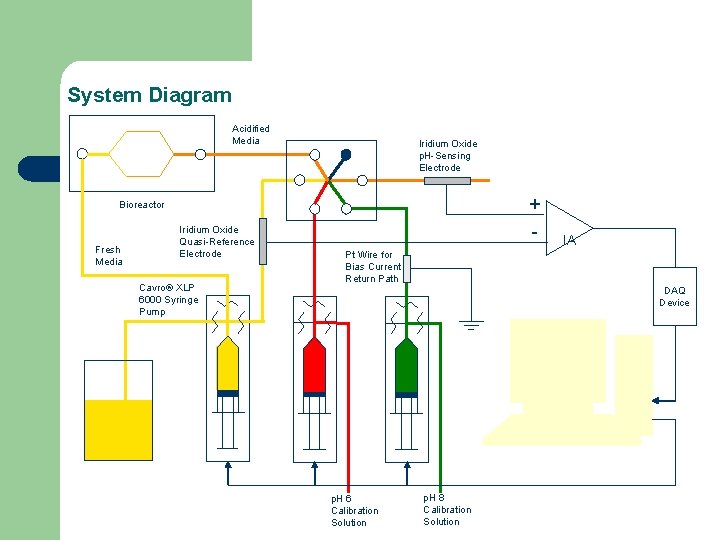
System Diagram Acidified Media Iridium Oxide p. H-Sensing Electrode + Bioreactor Fresh Media Iridium Oxide Quasi-Reference Electrode Cavro® XLP 6000 Syringe Pump - IA Pt Wire for Bias Current Return Path DAQ Device p. H 6 Calibration Solution p. H 8 Calibration Solution
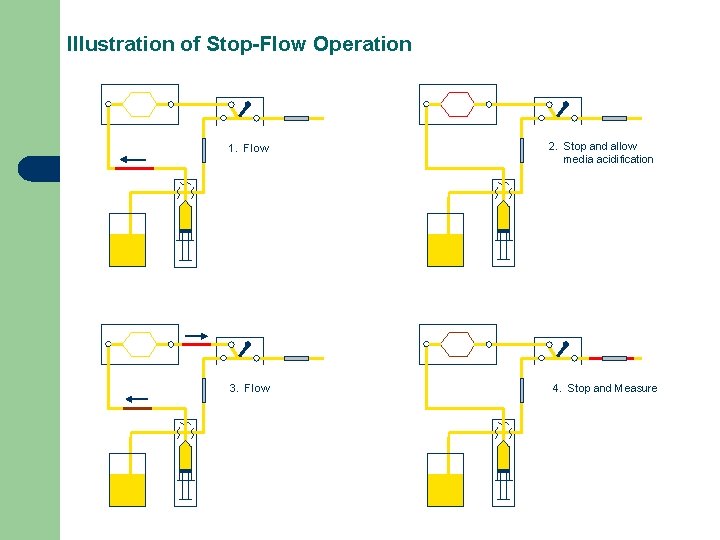
Illustration of Stop-Flow Operation 1. Flow 3. Flow 2. Stop and allow media acidification 4. Stop and Measure

Stop-flow Over Continuous Flow l l More equal nutrient/metabolite exchange Easier to conceptualize/model exchange Less delay in acquiring current p. H measurement If response time of sensor is long, allows measurement of endpoint p. H rather than moving average of p. H signal (calculation of acidification rate better with former)
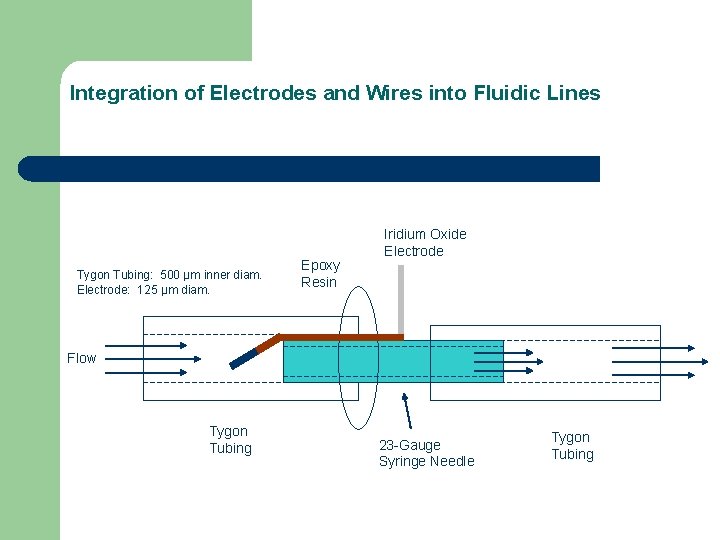
Integration of Electrodes and Wires into Fluidic Lines Tygon Tubing: 500 μm inner diam. Electrode: 125 μm diam. Epoxy Resin Iridium Oxide Electrode Flow Tygon Tubing 23 -Gauge Syringe Needle Tygon Tubing
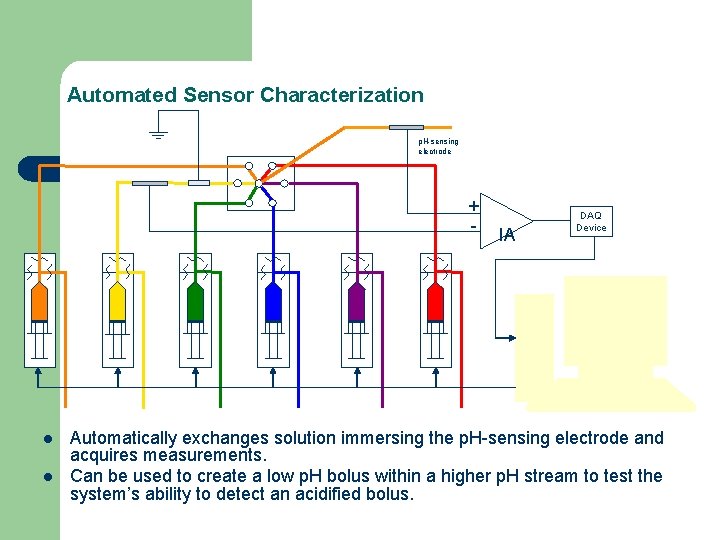
Automated Sensor Characterization p. H-sensing electrode + - l l IA DAQ Device Automatically exchanges solution immersing the p. H-sensing electrode and acquires measurements. Can be used to create a low p. H bolus within a higher p. H stream to test the system’s ability to detect an acidified bolus.
- Slides: 16