ThreeDimensional ClosedLoop Microfabricated Bioreactor Michael Hwang Jenny Lu
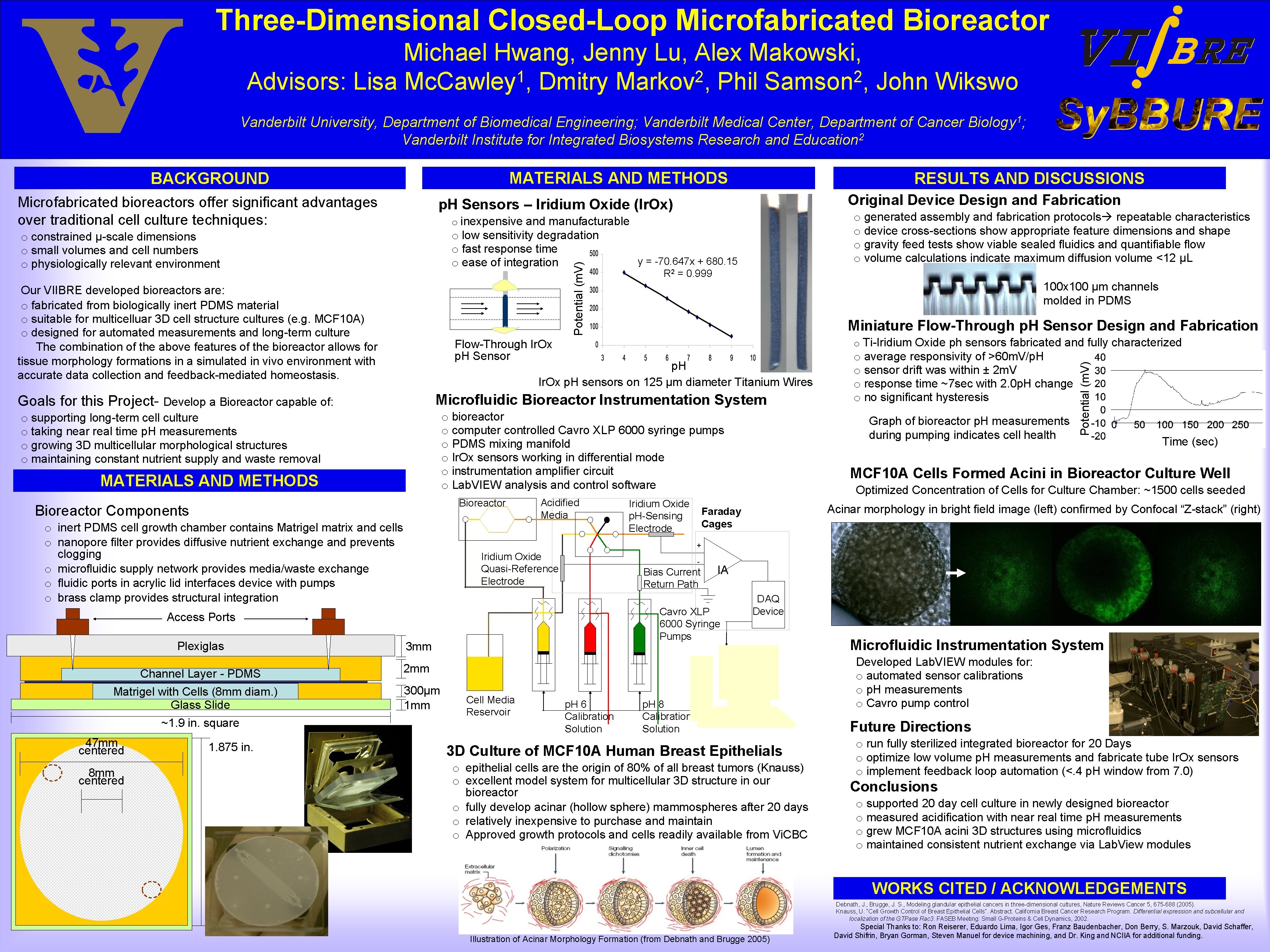
Three-Dimensional Closed-Loop Microfabricated Bioreactor Michael Hwang, Jenny Lu, Alex Makowski, 1 2 2 Advisors: Lisa Mc. Cawley , Dmitry Markov , Phil Samson , John Wikswo Vanderbilt University, Department of Biomedical Engineering; Vanderbilt Medical Center, Department of Cancer Biology 1; Vanderbilt Institute for Integrated Biosystems Research and Education 2 MATERIALS AND METHODS Microfabricated bioreactors offer significant advantages over traditional cell culture techniques: o low sensitivity degradation o fast response time o ease of integration Potential (m. V) Bioreactor Components o inert PDMS cell growth chamber contains Matrigel matrix and cells o nanopore filter provides diffusive nutrient exchange and prevents clogging o microfluidic supply network provides media/waste exchange o fluidic ports in acrylic lid interfaces device with pumps o brass clamp provides structural integration Acidified Media Iridium Oxide p. H-Sensing Electrode o average responsivity of >60 m. V/p. H o sensor drift was within ± 2 m. V o response time ~7 sec with 2. 0 p. H change o no significant hysteresis Graph of bioreactor p. H measurements during pumping indicates cell health Time (sec) MCF 10 A Cells Formed Acini in Bioreactor Culture Well Optimized Concentration of Cells for Culture Chamber: ~1500 cells seeded Acinar morphology in bright field image (left) confirmed by Confocal “Z-stack” (right) Faraday Cages + Iridium Oxide Quasi-Reference Electrode - Bias Current Return Path IA Cavro XLP 6000 Syringe Pumps Access Ports 8 mm centered o Ti-Iridium Oxide ph sensors fabricated and fully characterized o bioreactor o computer controlled Cavro XLP 6000 syringe pumps o PDMS mixing manifold o Ir. Ox sensors working in differential mode o instrumentation amplifier circuit o Lab. VIEW analysis and control software Bioreactor 1. 875 in. Miniature Flow-Through p. H Sensor Design and Fabrication Microfluidic Bioreactor Instrumentation System MATERIALS AND METHODS 47 mm centered 100 x 100 μm channels molded in PDMS p. H Ir. Ox p. H sensors on 125 μm diameter Titanium Wires o supporting long-term cell culture o taking near real time p. H measurements o growing 3 D multicellular morphological structures o maintaining constant nutrient supply and waste removal ~1. 9 in. square y = -70. 647 x + 680. 15 R 2 = 0. 999 Flow-Through Ir. Ox p. H Sensor Goals for this Project- Develop a Bioreactor capable of: Matrigel with Cells (8 mm diam. ) Glass Slide o generated assembly and fabrication protocols repeatable characteristics o device cross-sections show appropriate feature dimensions and shape o gravity feed tests show viable sealed fluidics and quantifiable flow o volume calculations indicate maximum diffusion volume <12 μL o inexpensive and manufacturable Our VIIBRE developed bioreactors are: o fabricated from biologically inert PDMS material o suitable for multicelluar 3 D cell structure cultures (e. g. MCF 10 A) o designed for automated measurements and long-term culture The combination of the above features of the bioreactor allows for tissue morphology formations in a simulated in vivo environment with accurate data collection and feedback-mediated homeostasis. Channel Layer - PDMS Original Device Design and Fabrication p. H Sensors – Iridium Oxide (Ir. Ox) o constrained μ-scale dimensions o small volumes and cell numbers o physiologically relevant environment Plexiglas RESULTS AND DISCUSSIONS Potential (m. V) BACKGROUND 3 mm DAQ Device 2 mm 300μm 1 mm Cell Media Reservoir p. H 6 Calibration Solution p. H 8 Calibration Solution 3 D Culture of MCF 10 A Human Breast Epithelials o epithelial cells are the origin of 80% of all breast tumors (Knauss) o excellent model system for multicellular 3 D structure in our bioreactor o fully develop acinar (hollow sphere) mammospheres after 20 days o relatively inexpensive to purchase and maintain o Approved growth protocols and cells readily available from Vi. CBC Microfluidic Instrumentation System Developed Lab. VIEW modules for: o automated sensor calibrations o p. H measurements o Cavro pump control Future Directions o run fully sterilized integrated bioreactor for 20 Days o optimize low volume p. H measurements and fabricate tube Ir. Ox sensors o implement feedback loop automation (<. 4 p. H window from 7. 0) Conclusions o supported 20 day cell culture in newly designed bioreactor o measured acidification with near real time p. H measurements o grew MCF 10 A acini 3 D structures using microfluidics o maintained consistent nutrient exchange via Lab. View modules WORKS CITED / ACKNOWLEDGEMENTS Debnath, J. , Brugge, J. S. , Modeling glandular epithelial cancers in three-dimensional cultures, Nature Reviews Cancer 5, 675 -688 (2005). Knauss, U. “Cell Growth Control of Breast Epithelial Cells”. Abstract. California Breast Cancer Research Program. Differential expression and subcellular and localization of the GTPase Rac 3. FASEB Meeting: Small G-Proteins & Cell Dynamics, 2002. Illustration of Acinar Morphology Formation (from Debnath and Brugge 2005) Special Thanks to: Ron Reiserer, Eduardo Lima, Igor Ges, Franz Baudenbacher, Don Berry, S. Marzouk, David Schaffer, David Shifrin, Bryan Gorman, Steven Manuel for device machining, and Dr. King and NCIIA for additional funding.
- Slides: 1