This slide show has been prepared under fair

This slide show has been prepared under fair use exemption of the U. S. Copyright Law and are restricted from further use.

Chemical math Density & Dimensional Analysis

Scientific Measurement: � Most scientific studies and experiments use a standard system of metric units called: Le Système International d’Unités or SI for short. • SI is a modern version of the metric system based on a decimal system that uses the number 10 as the base unit.

Scientific Measurement SI Units of Measurement: Use SI units — based on the metric system Length Mass Volume Time Temperature Meter, m Kilogram, kg Liter, L Seconds, s Celsius degrees, ˚C kelvins, K

Scientific Measurement Weight & Mass – Weight is a measure of the gravitational force on an object. – Weight varies with location depending on gravitational force. – Weight is a force, and the SI unit force is the Newton (N) – Mass is the amount of matter in an object. Kilogram (kg) – The mass of an object, unlike weight, does not change with an object’s position.
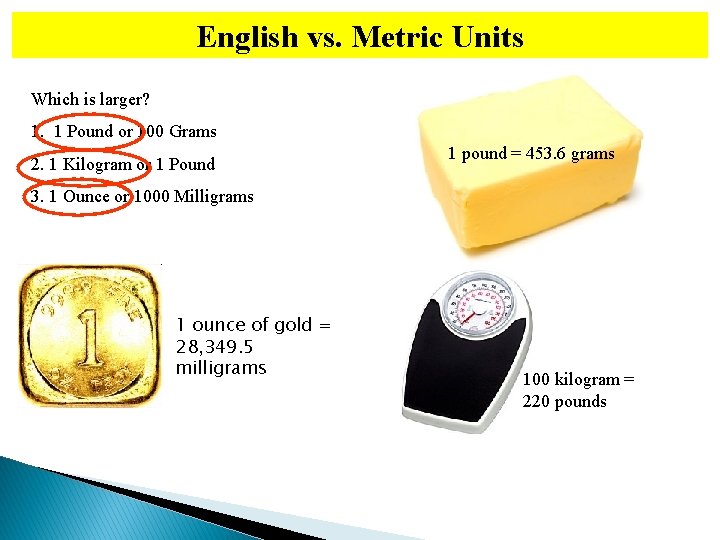
English vs. Metric Units Which is larger? 1. 1 Pound or 100 Grams 2. 1 Kilogram or 1 Pound 1 pound = 453. 6 grams 3. 1 Ounce or 1000 Milligrams 1 ounce of gold = 28, 349. 5 milligrams 100 kilogram = 220 pounds

MASS Units Mass refers to the amount of matter in an object. The base unit of mass in the metric system in the kilogram and is represented by kg. Standard: 1 kilogram is equal to the mass of the International Prototype Kilogram (IPK), a platinum-iridium cylinder kept by the BIPM at Sèvres, France. Kilogram Prototype

Measuring Mass We will be using triple-beam balances to find the mass of various objects. The objects are placed on the scale and then you move the weights on the beams until you get the lines on the right-side of the scale to match up. Once you have balanced the scale, you add up the amounts on each beam to find the total mass. What would be the mass of the object measured in the picture? _______ + _______ = ____ g

Measuring Mass – Triple-Beam Balance 1 st – Place the film canister on the pan. 2 nd – Slide the large rider (weight) to the right until the arm drops below the line. Move the rider back one groove. Make sure it “locks” into place. 3 rd – Repeat this process with the top weight. When the arm moves below the line, back it up one groove. 4 th – Slide the small weight on the front beam until the lines match up. 5 th – Add the amounts on each beam to find the total mass to the nearest tenth of a gram. Click here to try an online activity.

Scientific Measurement Area & Volume: – Area is the amount of surface included within a set of boundaries and is expressed in square units of length, such as square meters (m 2). – The amount of space occupied by an object is the object’s volume. • The SI units for volume are derived from the SI units used to measure length. • The basic SI unit of volume for a regularly shaped, solid object is the cubic meter (m 3). • SI measurements for liquid volumes are usually made in milliliters (m. L) or liters (L).

English vs. Metric Units 1 fl oz = 29. 573 ml Which is larger? A. 1 liter or 1 gallon B. 1 liter or 1 quart 1 - 12 -oz can of soda would equal approximately 355 ml. C. 1 milligram or 1 fluid ounce 1 quart = 0. 946 liters 1 gallon = 3. 79 liters It would take approximately 3 ¾ 1 liter bottles to equal a gallon.

VOLUME Units Volume is the amount of space an object takes up. The base unit of volume in the metric system in the liter and is represented by L or l. Standard: 1 liter is equal to one cubic decimeter

Measuring Volume We will be using graduated cylinders to find the volume of liquids and other objects. Read the measurement based on the bottom of the meniscus or curve. When using a real cylinder, make sure you are eye-level with the level of the water. What is the volume of water in the cylinder? _____m. L What causes the Meniscus? A concave meniscus occurs when the molecules of the liquid attract those of the container. The glass attracts the water on the sides.

10 m. L Graduate What is the volume of liquid in the graduate? 6 _2 _ m. L

100 m. L graduated cylinder What is the volume of liquid in the graduate? 52 _ _. 7 _ m. L

Self Test Examine the meniscus below and determine the volume of liquid contained in the graduated cylinder. The cylinder contains: 7 _ _6. 0 _ m. L

Measuring Solid Volume 9 cm 8 cm We can measure the volume of regular object using the formula length x width x height. _____ X _____ = _____ 10 cm We can measure the volume of irregular object using water displacement. Amount of H 2 O with object = ______ Amount of H 2 O without object = ______ Difference = Volume = ______ Click here for an online activity about volume. Choose Lessons Volume & Displacement

Scientific Measurement Time: – Time is the interval between two events and is usually measured with a watch or clock. – The SI unit of Time is the Second (s).

Scientific Measurement Temperature: – Temperature is a measure of the average vibrations of the particles that make up a material. – A mass made up of particles that vibrate quickly has a higher temperature than a mass whose particles vibrate more slowly. – Temperature is measured in degrees with a thermometer. • In the US, temperature is typically measured on the Fahrenheit (F) scale. • In science, temperature is often measured on the Celsius (C) scale. • In SI, temperature is measured on the Kelvin (K) scale.

Scientific Measurement Temperature: The two temperature scales most often used in the world today to record daily outdoor temperatures are the Fahrenheit temperature scale (used chiefly in the United States) and the Celsius temperature scale (used almost everywhere else). Two benchmark temperatures on these scales are the boiling point and freezing point of water. On the Fahrenheit scale, the boiling point is 212 degrees, the freezing point 32 degrees. On the Celsius scale, the boiling point is 100 degrees, the freezing point 0 degrees (a little easier to remember).

To convert from Celsius to Fahrenheit, the following formula is used: F = (C × 9/5) + 32 (what do you think the 32 represents? ) That’s right! The 32 represents the freezing point on the Fahrenheit scale – the difference between 0 on the Celsius and the 32 on the Fahrenheit is 32 degrees so we have to add it in. LET’S PRACTICE F = ( C X 9/5) + 32 If C equals 10 then what does F equal? F= ( ______ X 9/5) + 32 That’s right the 10 goes there! F= (10 X 9) = ______, divided by 5 = ____, + 32 = _______ So, F = ______degrees YOU DID IT! F= 50 degrees! Is it hot, warm, cold or cool? ? ? ?

So, to convert from Fahrenheit to Celsius, the following formula is used: C = (F - 32) × 5/9 Do you see that we now need to subtract the 32 and flip the fraction to get the Celsius degree? It is easy once you get the hang of it. If the Fahrenheit degree is 45, what will the Celsius temp. be? C= ( F - 32 ) X 5/9 C= ( _____ - 32 ) X 5/9 Yep! It is 45! C= (13 ) x 5 = ______ divided by 9 You got it: 65! C= 7. 2

Now, try out your formulas by completing the table below. Round off to the nearest whole degree. Celsius 0 16 30 40 - Fahrenheit 40 75 98. 6 0

1) At what temperature (in degrees Celsius) would it have to be outside before you wore a winter coat? Why? 2) At what temperature (in degrees Celsius) would you want it to be outside before you went swimming? Why? 3) If it is 20 degrees Celsius outside, how would you describe the temperature - Hot, warm, cool, or cold? Why? 4) If it is 11 degrees Celsius outside, would you wear: (Explain why you would or would not wear each) DO THE MATH a coat? mittens or gloves? a hat or cap? snow boots?

Scientific Measurement Scientific Notation � Scientific Notation: is used to express numbers that are very large or small. Scientists this type of shorthand to express the number as a multiplier and a power of 10. – In scientific notation, a number is expressed as a value between 1 and 10 multiplied by a power of 10. – The power of 10 is the number of places the decimal point must be shifted so that only a single digit remains either to the left or right of the decimal point.

Scientific Measurement Scientific Notation – If the decimal point must be shifted to the left, the exponent of 10 is Positive. – If the decimal point in a number must be shifted to the right, then the exponent of 10 is Negative. For example: 90 000 000 = 9 X 1010 5 974 200 000 000 000 = 0. 000001 = 1 X 10 -10 5. 9742 X 1024

Density - the volume which is occupied by a specific mass of a material, usually given in units of grams per cubic centimeters or grams per milliliter. D = m/v

Density of Liquid The density of a liquid can be determined by massing the liquid and then measuring the exact volume.

Displacement/Solid Density � The volume of a solid can be difficult to measure, so its density is determined using a different method. The volume is found by measuring the volume of a liquid that the solid displaces. In the example below, the volume of the compound is 17. 2 m. L Volume of water= 15. 0 ml Volume of water + compound = 32. 2 ml

Math problems � To solve all math problems in this class you will use the “u, k, p, s” method. � u: unknown � k: known � p: plan � s: solve

worksheet 1 problem 1 What is the density of carbon dioxide gas if 0. 196 g occupies a volume of 100 m. L? U: density Carbon dioxide k: mass CO 2 = 0. 196 g; vol. CO 2 =100 m. L p: d= m v s: d = 0. 196 g = 0. 00196 g/m. L 100 m. L 2 E -3 g/m. L

worksheet 1 problem 2 A block of wood 3. 0 cm on each side has a mass of 27 g. What is the density of this block? u: dwood k: m = 27 g length = 3. 0 cm p: d= m volume = l * w * h v s: volume = 3. 0 cm * 3. 0 cm = 27 cm 3 dwood = 27 g = 3 1. 0 g/cm 27 cm 3

worksheet 1 problem 3 An irregularly shaped stone was lowered into a graduated cylinder holding a volume of water equal to 2. 0 m. L. The height of the water rose to 7. 0 m. L. If the mass of the stone was 25 g. What was its density? u: dstone= k: v 1=2. 0 m. L; v 2= 7. 0 m. L; m = 25 g p: d= m vol = v 2 – v 1 v S: vol = 7. 0 m. L – 2. 0 m. L = 5. 0 m. L d = 25 g 5. 0 m. L = 5. 0 g/m. L

worksheet 1 problem 4 A 10. 0 cm 3 sample of copper has a mass of 89. 6 g. What is the density of copper? u: dcopper= k: v = 89. 6 cm 3; m = 10. 0 g p: d= m v S: d = 89. 6 g = 8. 96 g/cm 3 10. 0 cm 3

worksheet 1 problem 5 Silver has a density of 10. 5 g/cm 3 and gold has a density of 19. 3 g/cm 3. What would have a greater mass, 5 cm 3 of silver or 5 cm 3 of gold? u: m. Ag , m. Au k: v. Ag=5 cm 3; v. Au= 5 cm 3; d = 10. 5 g/cm 3 p: d= m v S: 10. 5 g = m. Ag 19. 3 g = m. Au cm 3 5. 0 cm 3 10. 5 g ∙ (5. 0 cm 3) = m. Ag ∙ (cm 3) cm 3 52. 5 g = m. Ag cm 3 19. 3 g ∙ (5. 0 cm 3)= m. Au ∙ (cm 3) cm 3 96. 5 g = m. Au 5 g of gold cm 3

worksheet 1 problem 6 Five m. L of ethanol has a mass of 3. 9 g, and 5. 0 m. L of benzene has a mass of 4. 4 g. Which liquid is denser? u: dethanol , dbenzene k: vethanol=5 m. L; vbenzene= 5 m. L; methanol =3. 9 g mbenzene = 5. 0 g p: d= m v S: dethanol= 3. 9 g dbenzene = 4. 4 g 5. 0 m. L dethanol = 0. 78 g/m. L dbenzene = 0. 88 g/m. L Benzene is denser

worksheet 1 problem 7 A sample of iron has the dimensions of 2 cm x 3 cm x 2 cm. If the mass of this rectangular-shaped object is 94 g, what is the density of iron? u: diron k: m = 94 g l=3 cm, w=2 cm, h=2 cm p: d= m volume = l * w * h v s: volume = 3 cm * 2 cm = 12 cm 3 dwood = 94 g = 7. 8 g/cm 3 12 cm 3 7. 8 g/cm 3

Density worksheet 2 problem 1 An aluminum bar with a volume of 35 cm 3 has a mass of 94. 5 g. What is the density of aluminum based on this information? u: density Al k: m= 94. 5 g; v=35 cm 3 p: d= m v s: d = 94. 5 g = 2. 7 g/cm 3 35 cm 3

Density worksheet 2 problem 2 A glass tube that holds exactly 75. 0000 m. L is filled with liquid mercury metal whose mass is shown to be 1020. 0011 g. From these data, what is the density of mercury metal? u: density Hg k: m= 1020. 0011 g; v=75. 0000 m. L p: d= m v s: d = 1020 g = 13. 6 g/m. L 75 m. L

Density worksheet 2 problem 3 The mass of 155 m. L of ethyl alcohol is found to be exactly 122. 5 g. What is the density of ethyl alcohol? u: density of ethyl alcohol k: m= 122. 5 g; v=155 m. L p: d= m v s: d = 122. 5 g = 0. 790 g/m. L 155 m. L

Density worksheet 2 problem 4 A block of ice with a mass of 519. 0 g is found to occupy a volume of 565. 8 cm 3. What is the density of the ice? u: density H 2 O(s) k: m= 519. 0 g; v=565. 8 cm 3 p: d= m v s: d = 519. 0 g = 0. 9173 g/cm 3 565. 8 cm 3

Density worksheet 2 problem 5 Find the density of a sample of silicon which has a volume of 235. 7 cm 3 and a mass of 570. 44 g. u: density Si k: m= 570. 44 g; v=235. 7 cm 3 p: d= m v s: d = 570. 44 g = 2. 42 g/cm 3 235. 7 cm 3

Density worksheet 2 problem 6 A solid cube of impure zinc metal, 3. 50 cm on a side, has a density of 6. 95 g/cm 3. What is the mass of the cube? u: mass of Zn k: d= 6. 95 g/m. L; l(per side)=3. 50 cm p: d= m volume=l*w*h v s: volume =3. 50 cm*3. 50 cm = 42. 88 cm 3 6. 95 g = m = cm 3 42. 88 cm 3 (6. 95 g)(42. 88 cm 3) = m (cm 3) cm 3 m = 176 g

Density worksheet 2 problem 7 An irregularly shaped piece of gold (density = 19. 3 g/cm 3) has a mass of 428. 4 g. What is the volume of this piece of gold? u: volume of Au k: d= 19. 3 g/m. L; m=428. 4 g p: d= m v s: 19. 3 g = 428. 4 g = m. L v (19. 3 g)(v) = m. L (428. 4 g) 19. 3 g 22. 2 cm 3 Au = v

Dimensional Analysis At times you need to convert from one unit to another, but you do not have a “formula” in which to plug the numbers into. You can use the relationship between any two known units. These are called conversion factors. For example, there are 60 minutes in one hour, so the relationship is 1 hour=60 minutes or 1 hour or 60 mins. 1 hour
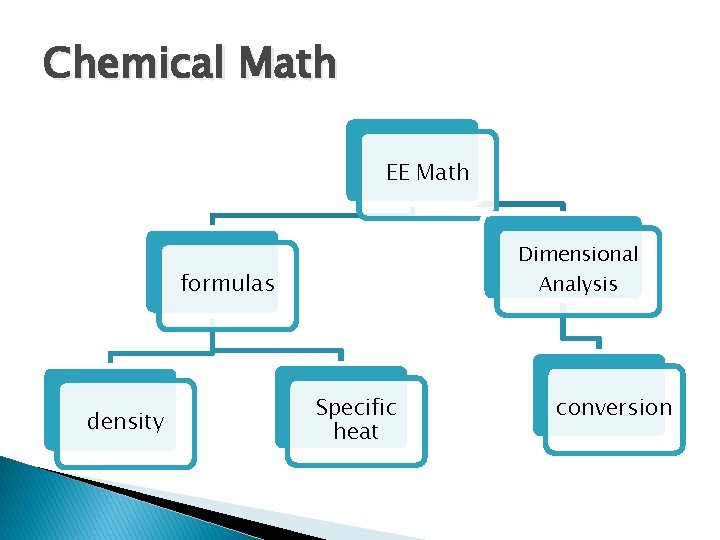
Chemical Math EE Math Dimensional Analysis formulas density Specific heat conversion

Dimensional Analysis � Other examples of conversion factors are: � 1 mile = 5 280 ft OR 1 mile 11 lbs = 5 kg OR 5280 ft 11 lbs 5 kg OR 5280 ft 1 mile OR 5 kg 11 lbs

Dimensional Analysis A number ### with a unit √ ### √ X √ Must equal 1 but not be the same. For all problems you MUST use Unknown, Known, Plan, Solve (note how this follows the scientific method. )

Dimensional Analysis Begin with KNOWN – what you want to convert from. Ex: 4 m = ______ cm u: cm k: 4 m p: 1 m=100 cm s: 4 m Known 100 cm 1 m
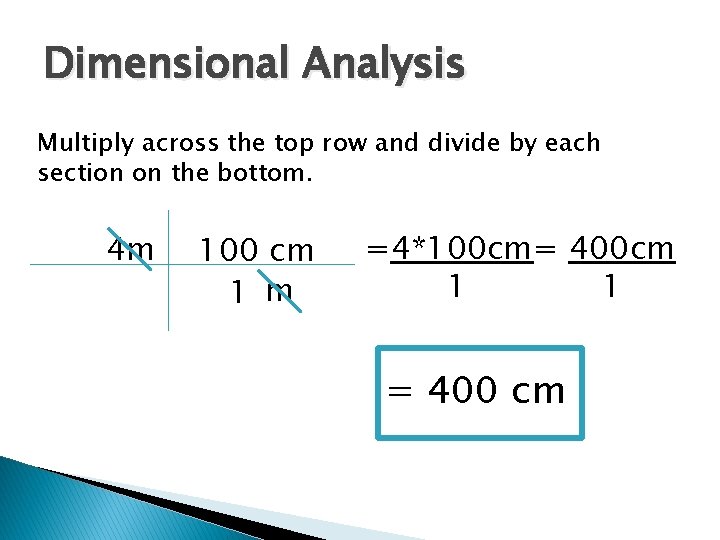
Dimensional Analysis Multiply across the top row and divide by each section on the bottom. 4 m 100 cm 1 m =4*100 cm= 400 cm 1 1 = 400 cm

Dimensional Analysis Convert 10 m to ______ Km. U: Km K: 10 m P: 1 Km= 1000 m 1 Km S: 10 meters = 10 1000 meters *1 Km 1000 = 0. 01 Km

Dimensional Analysis How much will it cost to take a trip by car. Where? Miles? Type of car? Miles per gallon? $ per gallon? u: how much does it cost to drive to Myrtle Beach k: 1 Myrtle Beach p: 1 Myrtle Beach = 250 miles; 1 gallon = $2. 50; 25 miles =1 gallon

Dimensional Analysis u: how much does it cost to drive to Myrtle Beach k: 1 Myrtle Beach p : 1 Myrtle Beach = 250 miles; 1 gallon = $3. 10; 25 miles =1 gallon 1 Myrtle Beach 250 miles 1 Myrtle Beach 1 gallon $3. 10 25 miles 1 gallon = 1 * 250 * 1 * $3. 10 ÷ 1÷ 25÷ 1 = $31

Metric prefix equivalents

Prefix >base 1 1 1 1 Exe (E) Penta (P) Tetra (T) Giga (G) Mega (M) Kilo (K) Hecto(H) Deka (D) Base = = = = Prefix < base 1018 1015 1012 109 106 103 (1000) 102 (100) 10

Prefix >base Base 1 1 1 Prefix < base = 10 deci (d) = 100 centi (c) = 1000 milli (m) = 106 micro (μ) = 109 nano (n) = 1012 pico (p)
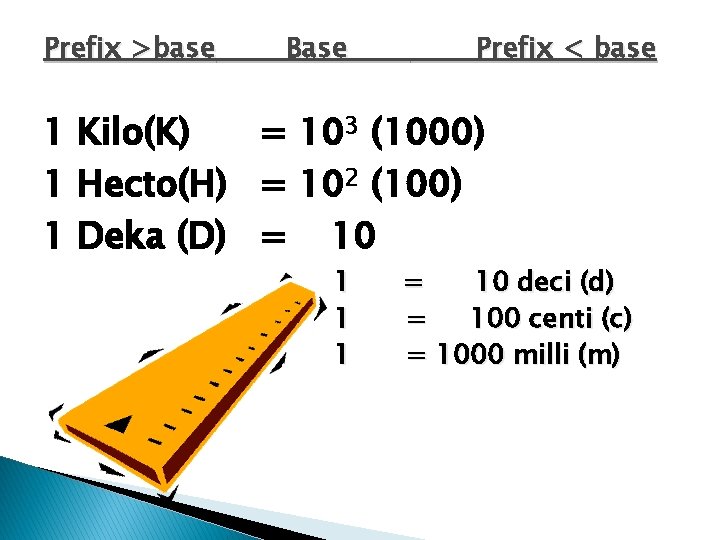
Prefix >base Base Prefix < base 1 Kilo(K) = 103 (1000) 1 Hecto(H) = 102 (100) 1 Deka (D) = 10 1 1 1 = 10 deci (d) = 100 centi (c) = 1000 milli (m)
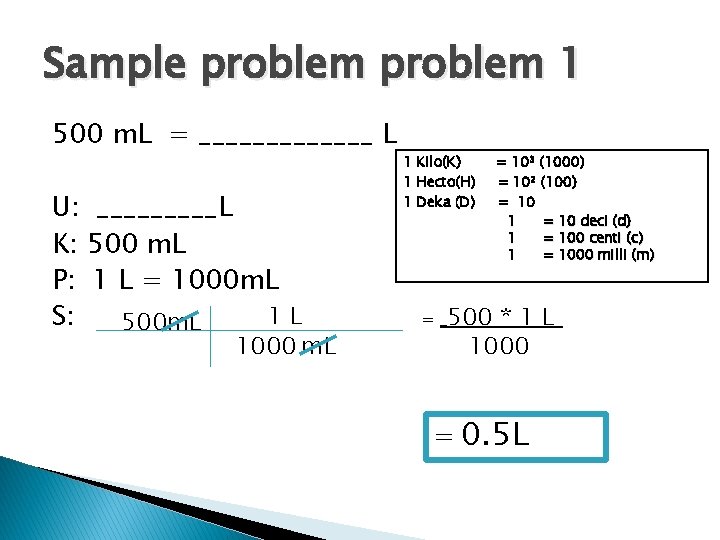
Sample problem 1 500 m. L = _______ L U: _____L K: 500 m. L P: 1 L = 1000 m. L 1 L S: 500 m. L 1000 m. L 1 Kilo(K) 1 Hecto(H) 1 Deka (D) = = 103 (1000) = 102 (100) = 10 1 = 10 deci (d) 1 = 100 centi (c) 1 = 1000 milli (m) 500 * 1 L 1000 = 0. 5 L

Sample problem 2 25 cg = _______ g U: _____g K: 25 cg P: 1 g= 100 cg 1 g S: 25 cg 100 cg 1 Kilo(K) 1 Hecto(H) 1 Deka (D) = = 103 (1000) = 102 (100) = 10 1 = 10 deci (d) 1 = 100 centi (c) 1 = 1000 milli (m) 25 * 1 g 100 = 0. 25 L

Sample problem 3 400 mg = _______ kg 1 Kilo(K) 1 Hecto(H) 1 Deka (D) U: _____kg K: 400 mg P: 1 g= 1000 mg; 1000 g = 1 kg 1 g 1 kg S: 400 mg 1000 g = 103 (1000) = 102 (100) = 10 1 = 10 deci (d) 1 = 100 centi (c) 1 = 1000 milli (m) We are not to the unknown = 400 * 1 kg÷ 1000 = 0. 0004 kg
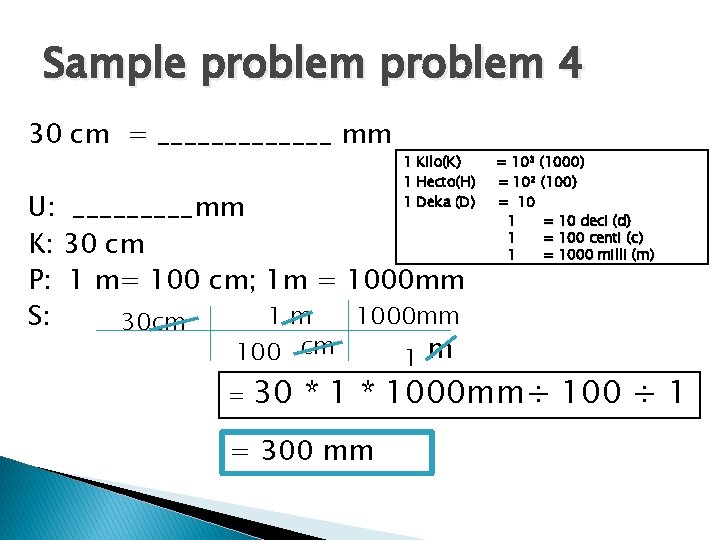
Sample problem 4 30 cm = _______ mm 1 Kilo(K) 1 Hecto(H) 1 Deka (D) U: _____mm K: 30 cm P: 1 m= 100 cm; 1 m = 1000 mm 1 m 1000 mm S: 30 cm 100 cm 1 m = = 103 (1000) = 102 (100) = 10 1 = 10 deci (d) 1 = 100 centi (c) 1 = 1000 milli (m) 30 * 1000 mm÷ 100 ÷ 1 = 300 mm
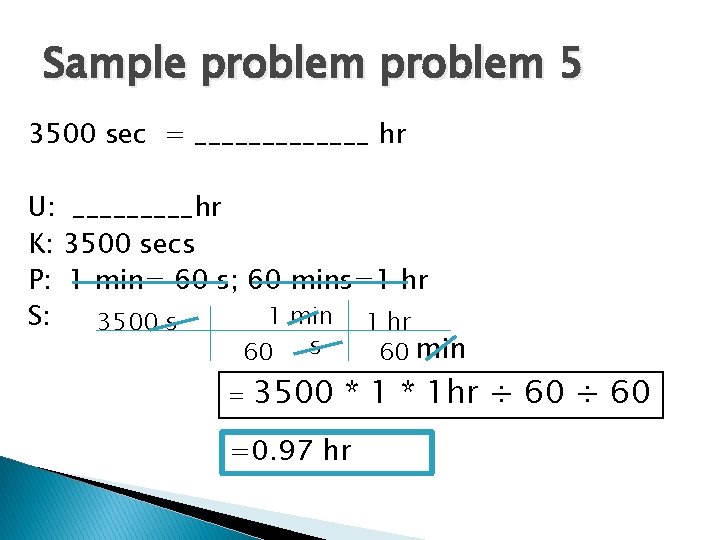
Sample problem 5 3500 sec = _______ hr U: _____hr K: 3500 secs P: 1 min= 60 s; 60 mins=1 hr 1 min 1 hr S: 3500 s 60 min = 3500 * 1 hr ÷ 60 =0. 97 hr

Sample problem 6 2 yrs = _______ secs U: _____secs K: 2 yrs P: 365 days= 1 yr; 1 day=24 hours; 1 min=60 sec 60 mins= 1 hr 24 hr 60 sec S: 60 min 365 days 2 yrs 1 min 1 yr 1 day 1 hr = 2*365*24 *60 sec÷ 1÷ 1÷ 1 =63 072 000 sec

Deep Water Horizon oil spill 4 526 668 barrels 42 gal 3. 785 L 1 barrel 1 gallon 1000 m. L 1 L = 719 604 411 960 m. L of oil spilled into the Gulf of Mexico 719 billon 604 million 411 thousand 960 gallons

Dimensional Analysis (Factor Label Method) worksheet 9 1. 2. 3. 4. 5. 6. 10000 sec 0. 0035 cg 12 lbs 90. in 11 000 hr 2 E 24 molecules 4. 2 moles 8. 100 liters 9. 4. 46 moles 10. 1. 3 E 24 7. molecules 11. 190 liters 12. 7. 5 liters

- Slides: 66