This course material is supported by the Higher

This course material is supported by the Higher Education Restructuring Fund allocated to ELTE by the Hungarian Government Kinetics of liquid phase reactions Diffusion Controlled and Activation Controlled Reactions Ernő Keszei Eötvös Loránd University, Institute of Chemistry

Historical overview Author(s), year, journal Content Ø Smoluchowski, 1917 Growth of colloid particles via (Z. Phys. Chem. 92, 129) aggregation from homogeneous solution Ø Debye, 1942 Any reactants, (Trans. Electrochem. Soc. 82, 265) any interaction potentials Ø Chandrasekhar, 1943 Applicability of the molecular (Rev. Mod. Phys. 15, 1) description of diffusion Ø Collins & Kimball, 1949 (For colloid particle growth: ) (J. Colloid Sci. 4, 425) singularity at t = 0 at r = r. AB (infinite reaction rate)

Historical overview Author(s), year, journal Content Ø Noyes, 1961 First proper treatment at t = 0 (Prog. React. Kinet. , 1, 129) (molecular description instead of a continuum treatment) Ø Wilemski & Fixman, 1973 Mathematical generalisation of theory (J. Chem. Phys. 58, 4009) and a critical overview of previous papers Ø Green & Pilling, 1982 General stochastic description; (J. Phys. Chem. 86, 1318) molecular level simulation Starink, 2014 extended volume concept combined with (Thermochimica Acta, 596, 109) soft impingement of diffusion fields

Reactions in solution vs. gas phase reactions Property in solution in gas phase Distance between adjacent molecules very small large Translational motion hindered free Encounter of reactant molecules not too frequent Separation of adjacent molecules hindered free Energy transfer from product easy rare event molecules with high energy
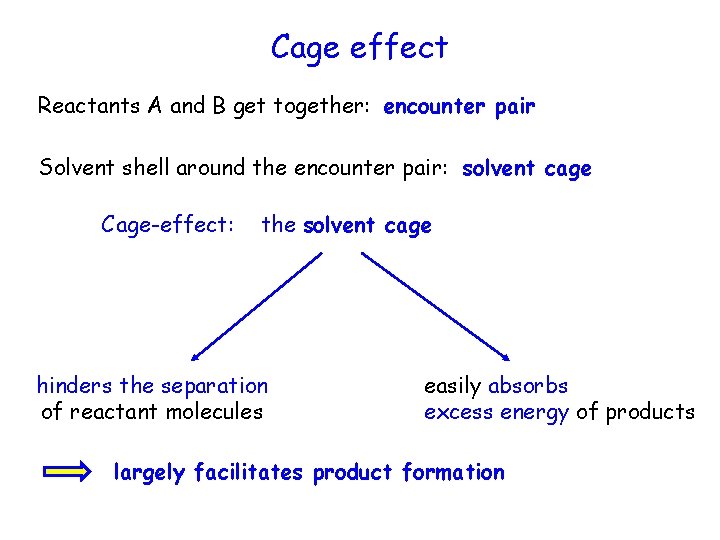
Cage effect Reactants A and B get together: encounter pair Solvent shell around the encounter pair: solvent cage Cage-effect: the solvent cage hinders the separation of reactant molecules easily absorbs excess energy of products largely facilitates product formation

Formal kinetic description A+B diffusion, k d diffusion, k – d {AB} reaction, k r product(s)

Formal kinetic description Consider {AB} as a QSS species and apply QSSA: rearrange substitute
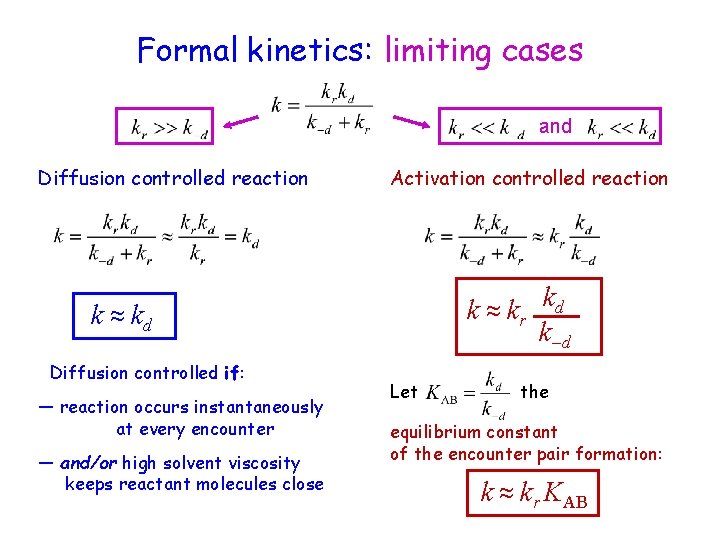
Formal kinetics: limiting cases and Diffusion controlled reaction Activation controlled reaction k ≈ kr kd k– d k ≈ kd Diffusion controlled if: — reaction occurs instantaneously at every encounter — and/or high solvent viscosity keeps reactant molecules close Let the equilibrium constant of the encounter pair formation: k ≈ kr KAB

Rate constants: orders of magnitude Solvent: water at 25 ºC — activation energy of diffusion: ~ 15 k. J/mol („large” activation energy of reactions: well above 20 – 30 k. J/mol) — kd ~ 109 – 1010 dm 3 mol– 1 s– 1 — k– d ~ 1010 – 1011 s– 1 (as KAB ~ 10– 1 dm 3 mol– 1) — kr >> k– d : if kr > 1012 s– 1 (diffusion control) — kr << k– d : if kr < 109 s– 1 (activation control)

Conditions to solve the diffusion equation Approximations in the differential equation of diffusion: Both A and B are neutral (uncharged) molecules B is in a large excess compared to A; i. e. [B] ≈ constant As [A] is small compared to [B], thus individual molecules of A can react independently The radial distribution of B around a molecule A can be substituted by the ensemble average over large volumes As a consequence, the reaction system can be considered as many independent spherically symmetrical „cells” with a central molecule A surrounded by many molecules B

Conditions to solve the diffusion equation B B B A r B B r. AB : reaction radius (A and B react immediately within this distance) [B] : bulk concentration of B at : [B]r = 0 (inner boundary condition) at : [B]r = [B] (outer boundary condition) Distribution of [B]r between [B]r = 0 and [B]r = [B] : a smooth function Concentration gradient: Diffusive flux:

The diffusion equation Radial distribution of concentration of B around a central atom A: zero from r = 0 to r. AB , [B] at r = ∞, and it is differentiable between the two. General diffusion flux: (1 dimension) surface concentration gradient diffusion coefficient For the spherically symmetric diffusion in the – r direction (←): (the surface of a sphere) (DAB = DA + DB : relative diffusion coefficient)

Solving the diffusion equation readily provides the average radial concentration integration At r = r. AB , [B]r = 0, thus the flux : As there is no production or consumption of B at r ≥ r. AB , the flux should not depend on r.

Solving the diffusion equation Let us substitute into Thus, the radial concentration profile is

Radial concentration profile diffusion controlled reaction (a): activation controlled reaction (b): Pilling & Seakins 1995

Diffusion controlled reaction rate — the radial diffusion flux of molecules B is constant (around all A) until r. AB, where they immediately react — the radial diffusion flux of B is the same around all A molecules Thus, the reaction rate is the same as the rate of diffusion around all A’s: The rate equation for the reaction: Comparing the two: As the unit of DAB is m 2 s– 1, the unit of k′ is m 3 s– 1 The diffusion controlled rate constant k in dm 3 mol– 1 s– 1 unit:

DAB , the relative diffusion coefficient How do we know DAB ? pure water solution of A B pure water A solution of B Ionic species: Ø DAB ~ 2 x 10− 9 m 2 s− 1 Ø r. AB ~ 0. 5 nm k ~ 8 x 109 dm 3 mol− 1 s− 1 Rule of thumb: k > 109 dm 3 mol− 1 s− 1 indicates diffusion control

Typical diffusion controlled reactions Ø Recombination (of radicals, atoms): small or zero activation energy e. g. I+I I 2 (in hexane: k = 1. 3 x 1010 dm 3 mol− 1 s− 1) Ø Acid-base reactions e. g. H+ + OH− H 2 O (in water: k = 4 x 1011 dm 3 mol− 1 s− 1) Ø Excitation quenching e. g. naphtalene hν naphtalene* biacetyl naphtalene + biacetyl* (continuous excitation: k = 2. 2 x 1010 dm 3 mol− 1 s− 1)

Diffusion controlled reactions of ions Ø Brownian motion: can be simulated by discrete displacements („hopping”) Ø If particles are charged (ions: r − 2 potential dependence) hopping is not completely random diffusion term migration term

Diffusion controlled reactions of ions diffusion term Rearranged: Solution of the linear differential equation: migration term

Diffusion controlled reactions of ions Potential energy of the Coulomb interaction: Onsager (escape) radius: the distance where the Coulomb interaction equals the average thermal energy: Within the Onsager radius: attraction of oppositely charged ions repulsion of ions both charged positively or negatively Comparing V(r) and rc : for Coulomb interaction

Diffusion controlled reactions of ions Solution for diffusion + migration: Substitution of
![Diffusion controlled reactions of ions Substituting [B]r = 0 at r = r. AB Diffusion controlled reactions of ions Substituting [B]r = 0 at r = r. AB](http://slidetodoc.com/presentation_image_h/ff626acd5719ba235f46784438c4f400/image-23.jpg)
Diffusion controlled reactions of ions Substituting [B]r = 0 at r = r. AB , we get: ; Expressig and calculating

Diffusion controlled reactions of ions Rate of diffusion controlled ion-ion reactions: Effective reaction radius: (for pure Coulomb interactions) Dependence of reff on solvent permittivity; r. AB= 0. 5 nm, │z. A│ = │z. B│= 1 reff / nm Solvent εr rc / nm z. A = z. B z. A = − z. B Water 78, 5 0, 7 0, 2 0, 9 Methanol 32, 6 1, 7 0, 06 1, 8 Ethanol 24, 3 2, 3 0, 02 2, 3 Dietil-ethter 4, 27 13 10− 10 13 Hexane 1, 89 29 10− 24 29

Time-dependent rate constants Up to now, there was a stationary concentration gradient. Initiating the reaction from a precursor of A by pulse radiolysis, a laser pulse, T-jump or P-jump, the initial distribution of the molecules is homogeneous; i. e. , [B]r = constant, independent of r, even if r < r. AB. In this case, the onset of reaction involves close partners first, thus the concentration profile will change until the stationary profile would form. The time-dependent concentration profile can be calculated using Fick’s 2 nd law:

Time-dependent rate constants The Laplace operator ( ) for a sphere can be given as: For an isotropic sphere: The time-dependent equation: Boundary conditions (as before): and

Time-dependent rate constants The flux depends on the radius ! The concentration profile is time-dependent the rate constant is also time-dependent The concentration profile will ″smoothen″ until the stationary profile is achieved, when the diffusion flux inwards at r = r. AB is equal to the reaction rate. Solution for the time-dependent concentration profile: error function:

Time-dependent rate constants Solution for the time-dependent flux: Relations used to get the solution:

Time-dependent rate constants Reaction rate: Rate constant: • problem : at • Solution: at short times, continuum description is not valid; (we should use molecular description below ~ 1 ps)
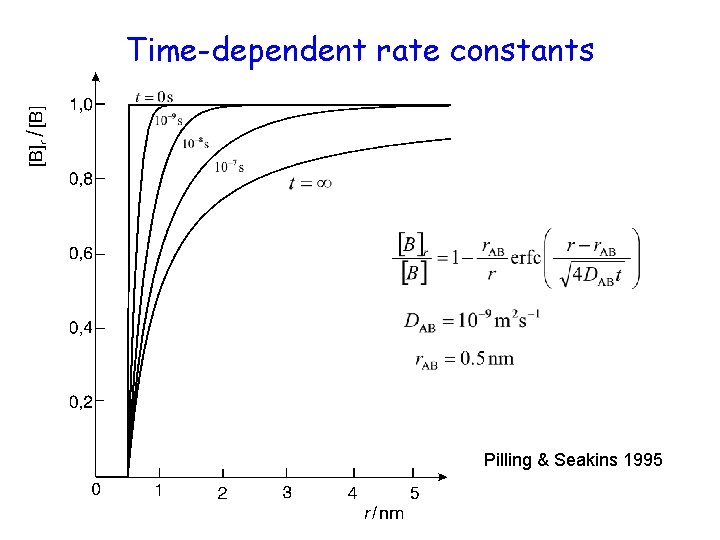
Time-dependent rate constants Pilling & Seakins 1995

From diffusion control to activation control Former inner boundary condition: at advantage: simplifies the maths; no need to deal with conditions within disadvantage: applicable only for diffusion control r. AB Let us use a more general inner boundary condition it should be applicable for: • diffusion controlled • activation controlled • and mixed control reactions A suitable modification: Let us reformulate the problem using this latter boundary condition.

From diffusion control to activation control Diffusion flux is same as before: Reactive flux (that of molecules B at a distance through a spherical shell at r. AB : In a stationary state, the two fluxes should be equal:

From diffusion control to activation control Differential equation to solve: Solution: Boundary conditions: former present at

From diffusion control to activation control Applying the boundary conditions: formerly now Rate constant: now formerly

From diffusion control to activation control Concentration dependence of the rate constant: now formerly
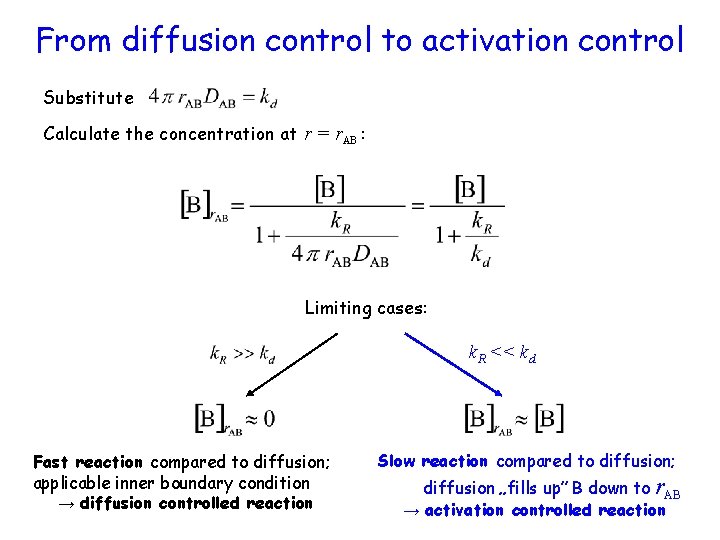
From diffusion control to activation control Substitute Calculate the concentration at r = r. AB : Limiting cases: k. R << kd Fast reaction compared to diffusion; applicable inner boundary condition → diffusion controlled reaction Slow reaction compared to diffusion; diffusion „fills up” B down to r. AB → activation controlled reaction

From diffusion control to activation control Concentration profile: If k. R ≈ kd, B molecules react relatively fast (but not immediately), diffusion cannot deliver enough B to the close vicinity of A. Adapted from Pilling & Seakins 1995

From diffusion control to activation control Substitute kd into the rate expression: k. R << kd k ≈ k. R pure activation control k. R is comparable to kd k < k. R diffusion cannot completely ”fill up” B that is consumed by the reaction k. R is the rate constant that would apply if the concentration of B at r. AB were always the same as the bulk concentration

Diffusion controlled reactions diffusion control: Stokes-Einstein equation: hydrodynamic radius of the diffusing species constant solvent viscosity Values of the constant: • a = 6: the solvent is a continuum, the spherical species is much greater than the solvent molecules, and it ″slips” • a = 4: same case but the spherical species ″sticks″ • a = 1: the diffusing (spherical) species’ size is roughly the same as the solvent molecule

Diffusion controlled reactions rs depends on solvent-reactant interactions r. AB depends on reactant-reactant interactions In case of missing information, usual approximations: If r. A and r. B are also equal:

Diffusion controlled reactions Special case; with r. A = r. B General case kd ∝ 1 / η is always a good approximation If r. A = r. B then kd does not depend on r. AB reason: increasing reaction radius also increases kd but decreases DAB Solvent 103 / kg m– 1 s– 1 Solvent hexane 0. 326 2 -propanol water 1. 00 1, 2 -ethanediol ethanol 1. 20 1, 2, 3 -propanetriol 103 / kg m– 1 s– 1 2. 26 19. 9 1490

Diffusion controlled reactions Temperature dependence: Increasing T decreases both D and kd increas e Arrhenius equation: Aqueous solutions: Ea ~ 15 k. J mol− 1 A ~ 1012 dm 3 mol− 1 s− 1 Ea for diffusion originates from more complex phenomena than in a gas including close-packedness and cooperative displacement The pre-exponencial factor A is somewhat higher than the collision number Z in a gas

Acknowledgements Figures marked as Pilling & Seakins 1995 are reproduced from M. J. Pilling, P. W. Seakins: Reaction Kinetics, Oxford University Press, 1995

END of the lecture Diffusion Controlled and Activation Controlled Reactions Thank you for your attention ! This course material is supported by the Higher Education Restructuring Fund allocated to ELTE by the Hungarian Government
- Slides: 44