THIRD EDITION HUMAN PHYSIOLOGY AN INTEGRATED APPROACH Dee










![Secondary Active Transport: Uses Kinetic Energy of [ion] • Cotransports • [Ion ] restored Secondary Active Transport: Uses Kinetic Energy of [ion] • Cotransports • [Ion ] restored](https://slidetodoc.com/presentation_image_h/dffcd80f15372b9af71360ad74999512/image-28.jpg)














- Slides: 62

THIRD EDITION HUMAN PHYSIOLOGY AN INTEGRATED APPROACH Dee Unglaub Silverthorn, Ph. D. Chapter 5 Membrane Dynamics Power. Point® Lecture Slide Presentation by Dr. Howard D. Booth, Professor of Biology, Eastern Michigan University Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

About this Chapter • Cell membrane structures and functions • Membranes form fluid body compartments • Membranes as barriers and gatekeepers • How products move across membranes • Distribution of water and solutes in cells & the body • Chemical and electrical imbalances • Membrane permeability and changes Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

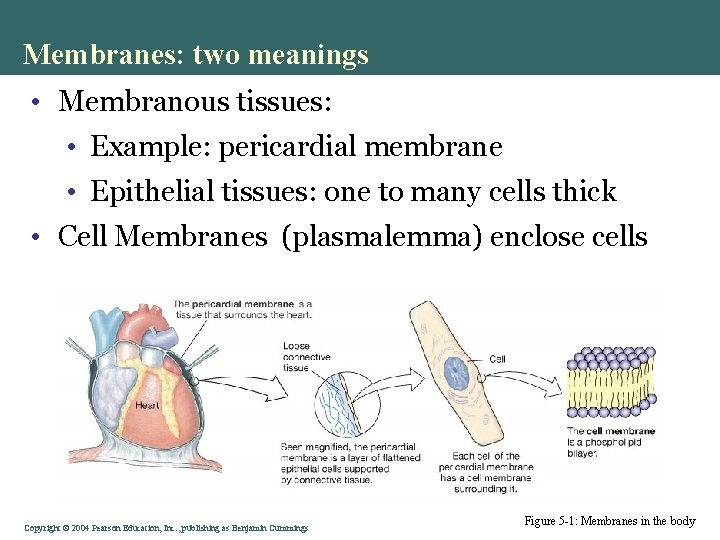
Membranes: two meanings • Membranous tissues: • Example: pericardial membrane • Epithelial tissues: one to many cells thick • Cell Membranes (plasmalemma) enclose cells Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -1: Membranes in the body

Cell Membranes: Overview • Cell structure & support • Barrier isolates cell (impermeable) • Chemically • Physically • Regulates exchange (semipermeable) • Cell communication Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

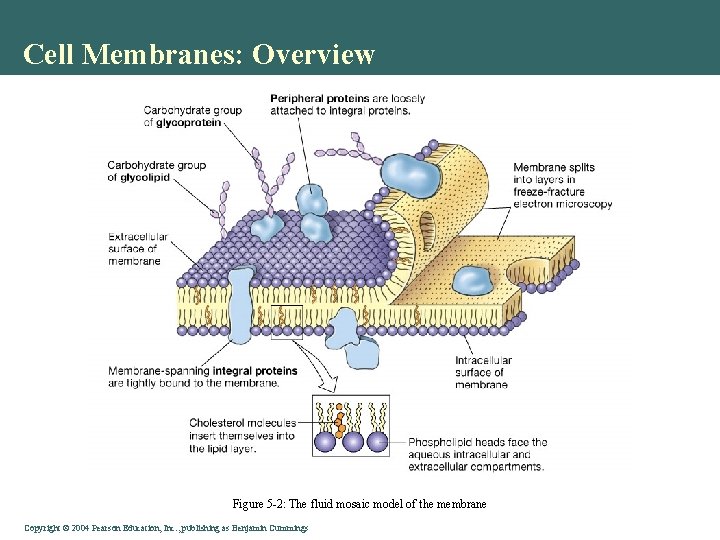
Cell Membranes: Overview Figure 5 -2: The fluid mosaic model of the membrane Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Membrane Structure • Phospholipid bilayer and cholesterol • Membrane proteins • Peripheral (associated) • Integral Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Structural Membrane Proteins: Membrane-Spanning • Structure • Cell polarity • Phosphorylation • Extracellular matrix Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

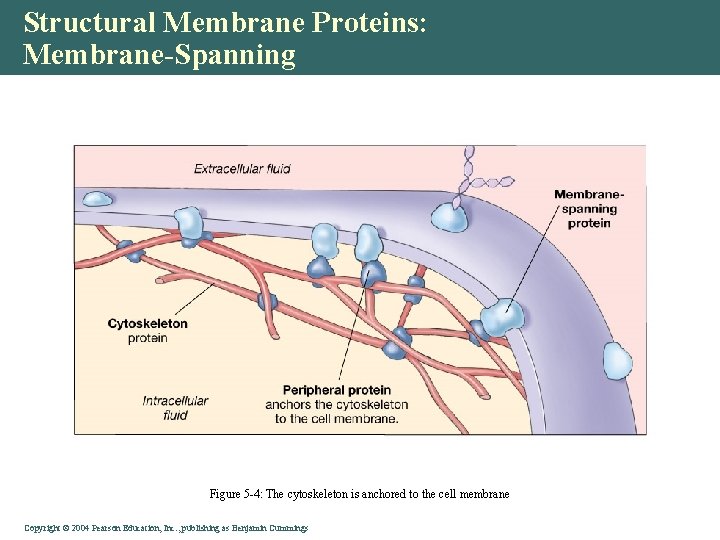
Structural Membrane Proteins: Membrane-Spanning Figure 5 -4: The cytoskeleton is anchored to the cell membrane Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

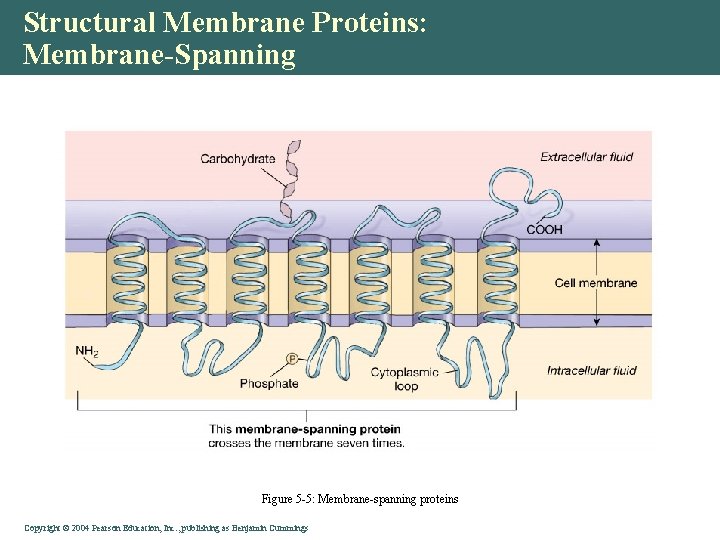
Structural Membrane Proteins: Membrane-Spanning Figure 5 -5: Membrane-spanning proteins Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

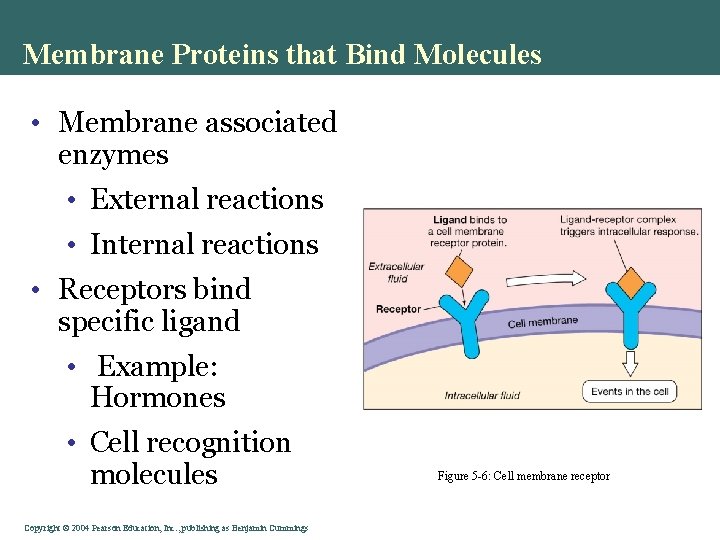
Membrane Proteins that Bind Molecules • Membrane associated enzymes • External reactions • Internal reactions • Receptors bind specific ligand • Example: Hormones • Cell recognition molecules Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -6: Cell membrane receptor

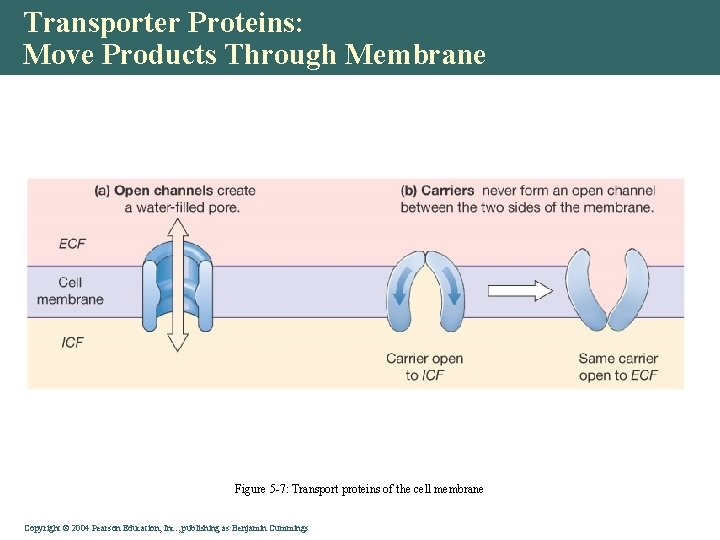
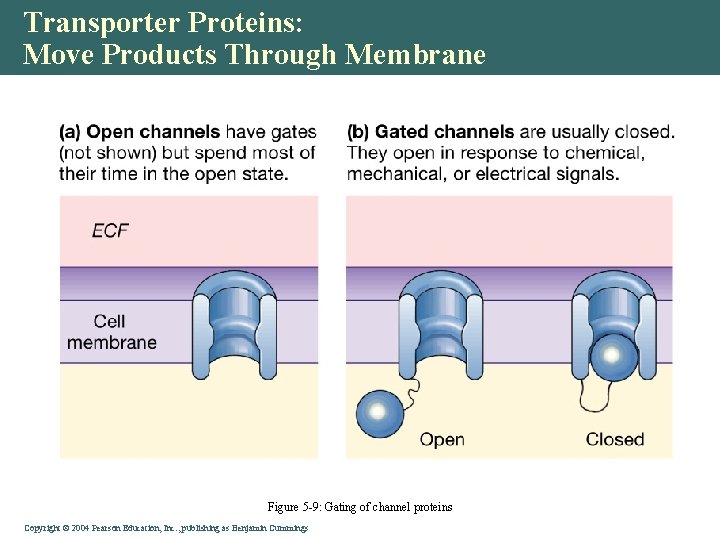
Transporter Proteins: Move Products Through Membrane • Channel proteins • Open • Gated • Carrier proteins • Bind to substrate • Slower transport Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Transporter Proteins: Move Products Through Membrane Figure 5 -7: Transport proteins of the cell membrane Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Transporter Proteins: Move Products Through Membrane Figure 5 -9: Gating of channel proteins Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

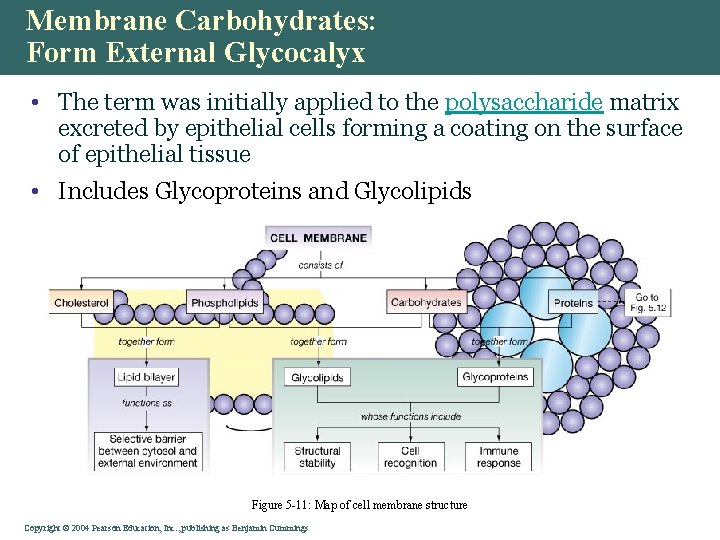
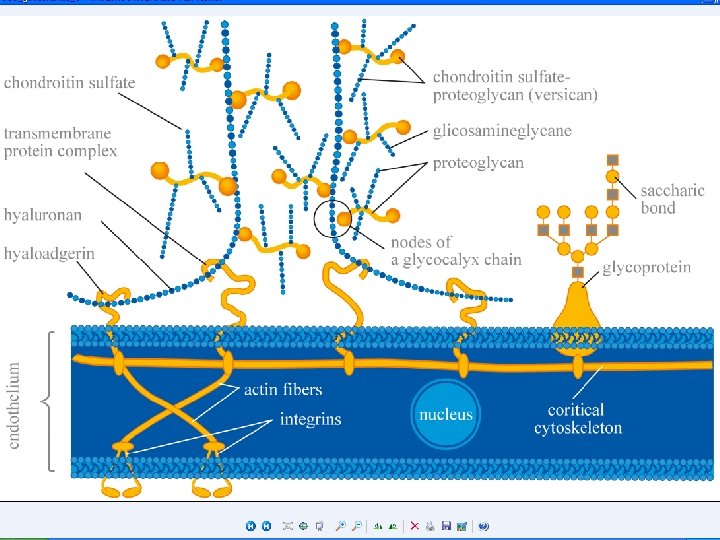
Membrane Carbohydrates: Form External Glycocalyx • The term was initially applied to the polysaccharide matrix excreted by epithelial cells forming a coating on the surface of epithelial tissue • Includes Glycoproteins and Glycolipids Figure 5 -11: Map of cell membrane structure Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

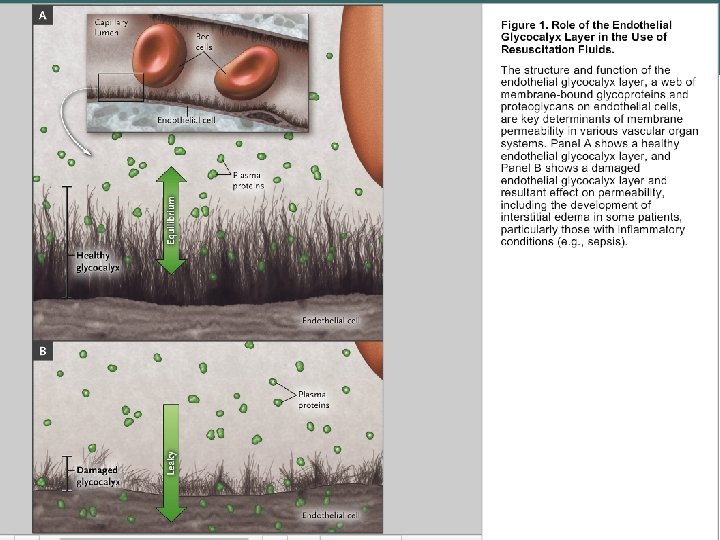
The Glycocalyx • glycocalyx — carbohydrate-rich peripheral zone of the external surface coating of the membrane in most eukaryotic cells. • Description • The outer surface of cells is covered with lipopolysaccharide "hairs" consisting of proteoglycans, glycoproteins and glycolipids, which are called “glycocalyx” Carbohydrate components of the glycocalyx include both compounds covalently bound to proteins or, to a lesser extent, to lipids on the cell surface, and additional glycoproteins and polysaccharides which are non-covalently attached to them. Some of the adsorbed macromolecules are components of the extracellular matrix, which makes it difficult to distinguish between such matrices and the glycocalyx with the cell membrane. Glycocalyx is considered as a protective layer on the vessel wall against pathogenic effects, a network barrier to the movement of molecules. It is assumed that the endothelial glycocalyx has a definite ultrastructure and may be connected with the cytoskeleton to serve as a mechanochemical transducer of blood flow effects (shear stress) into other processes of cell signaling. Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Functions of the Glycocalyx • Protection: Cushions the plasma membrane and protects it from chemical injury • Immunity to infection: Enables the immune system to recognize and selectively attack foreign organisms • Defense against cancer: Changes in the glycocalyx of cancerous cells enable the immune system to recognize and destroy them • Transplant compatibility: Forms the basis for compatibility of blood transfusions, tissue grafts, and organ transplants • Cell adhesion: Binds cells together so that tissues do not fall apart • Inflammation regulation: Glycocalyx coating on endothelial walls in blood vessels prevents leukocytes from rolling/binding in healthy states[4] • Fertilization: Enables sperm to recognize and bind to eggs • Embryonic development: Guides embryonic cells to their destinations in the body Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

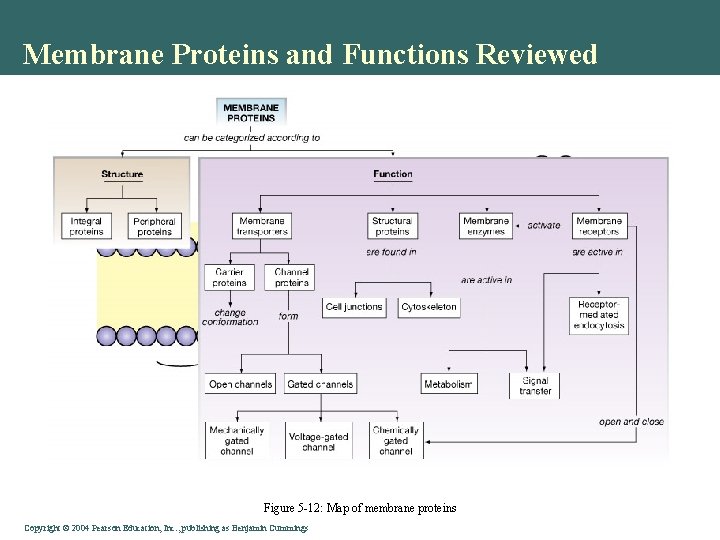
Membrane Proteins and Functions Reviewed Figure 5 -12: Map of membrane proteins Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

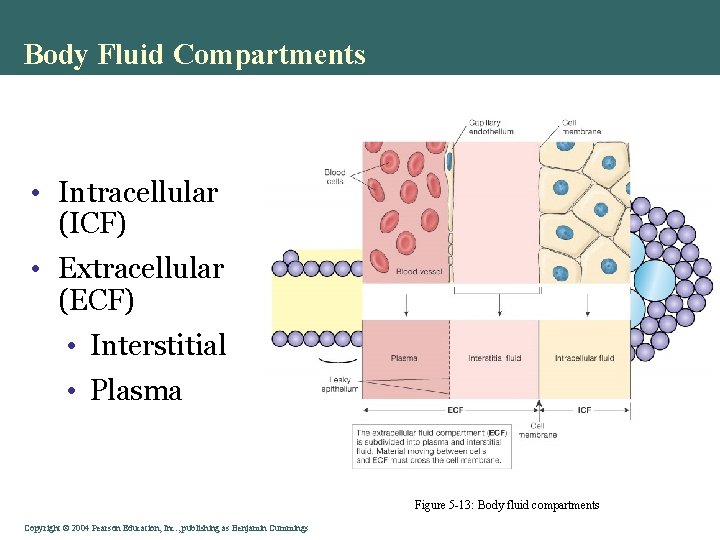
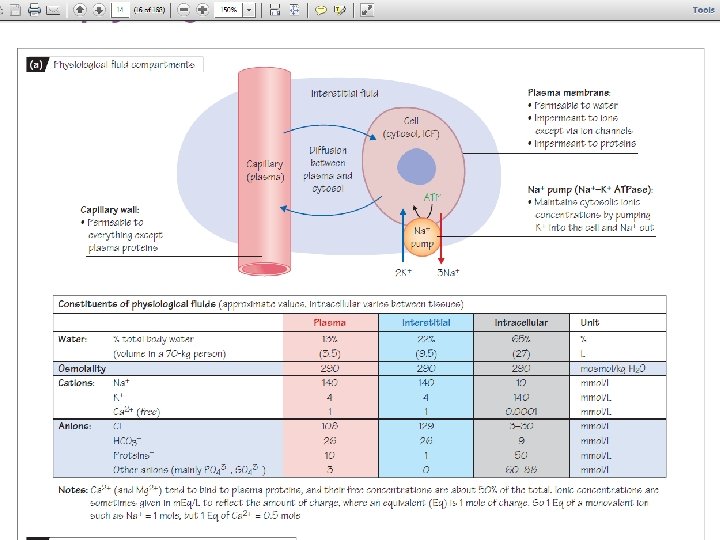
Body Fluid Compartments • Intracellular (ICF) • Extracellular (ECF) • Interstitial • Plasma Figure 5 -13: Body fluid compartments Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

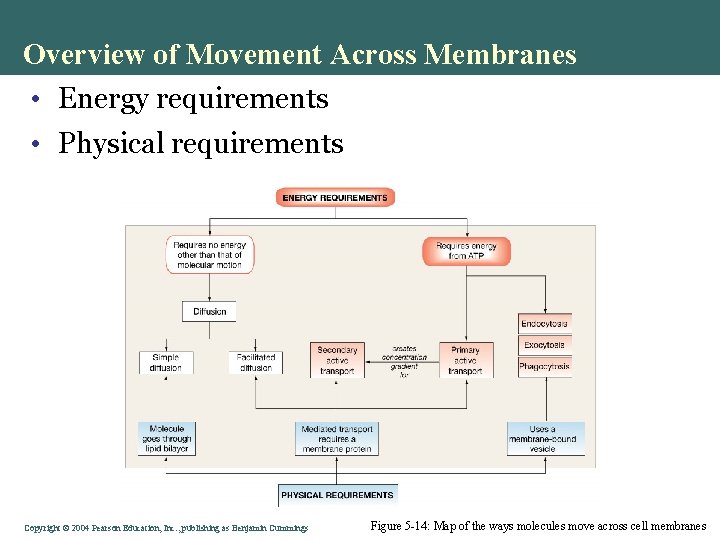
Overview of Movement Across Membranes • Energy requirements • Physical requirements Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -14: Map of the ways molecules move across cell membranes

Diffusion: Passive & down a concentration gradient • Stops at equilibrium • Rate factors: membrane, temperature, distance, & size Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -16: Fick’s law of diffusion

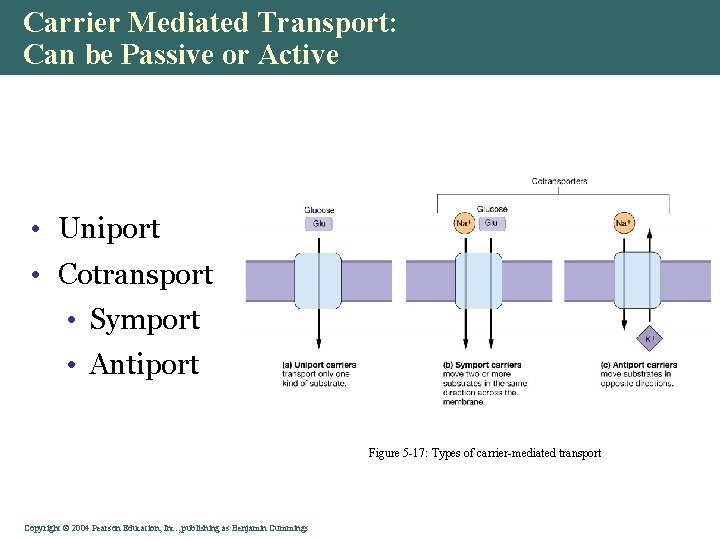
Carrier Mediated Transport: Can be Passive or Active • Uniport • Cotransport • Symport • Antiport Figure 5 -17: Types of carrier-mediated transport Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

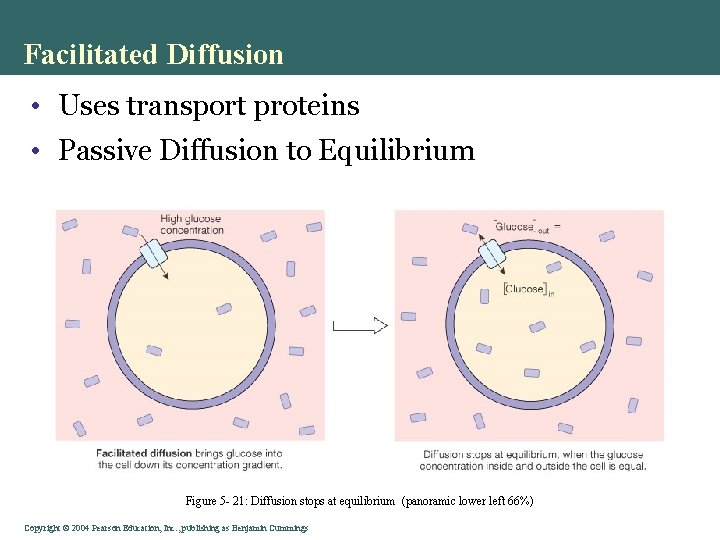
Facilitated Diffusion • Uses transport proteins • Passive Diffusion to Equilibrium Figure 5 - 21: Diffusion stops at equilibrium (panoramic lower left 66%) Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

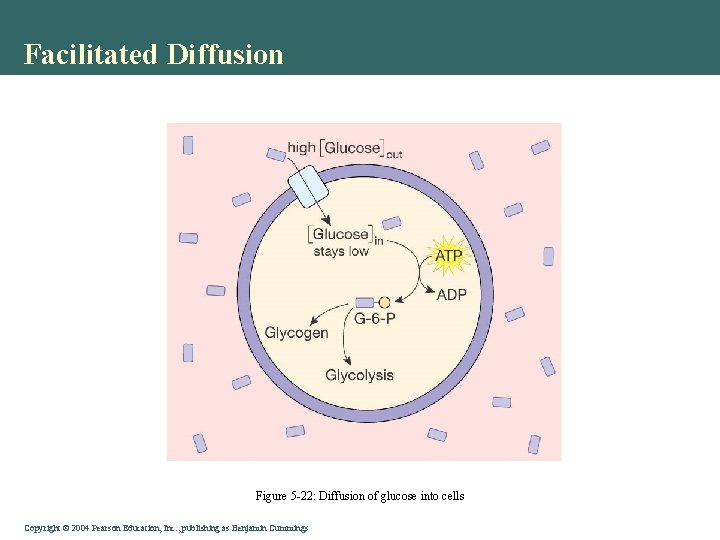
Facilitated Diffusion Figure 5 -22: Diffusion of glucose into cells Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

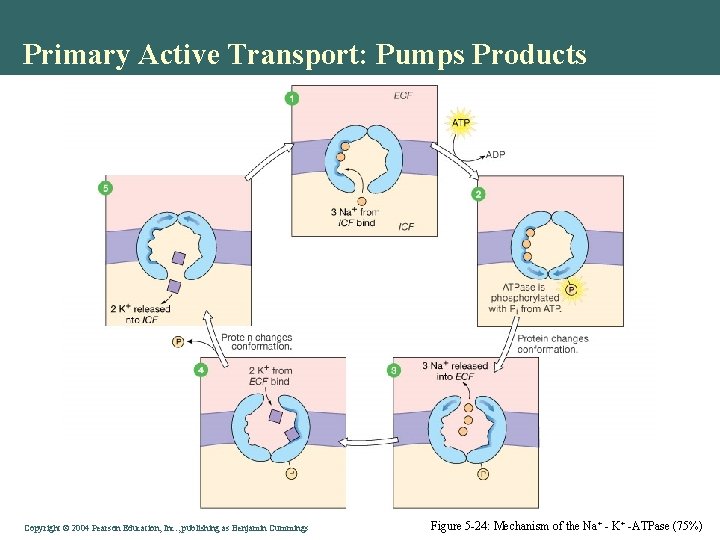
Primary Active Transport: Pumps Products • Uses ATP to move products • Up a concentration gradient Figure 5 -23: The Na+ - K+ -ATPase Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Primary Active Transport: Pumps Products Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -24: Mechanism of the Na + - K+ -ATPase (75%)
![Secondary Active Transport Uses Kinetic Energy of ion Cotransports Ion restored Secondary Active Transport: Uses Kinetic Energy of [ion] • Cotransports • [Ion ] restored](https://slidetodoc.com/presentation_image_h/dffcd80f15372b9af71360ad74999512/image-28.jpg)
Secondary Active Transport: Uses Kinetic Energy of [ion] • Cotransports • [Ion ] restored • using ATP Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -25: Sodium-glucose symporter

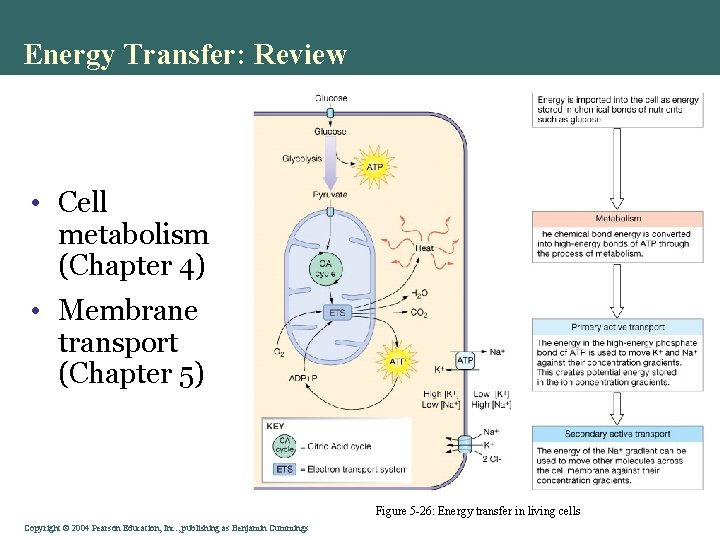
Energy Transfer: Review • Cell metabolism (Chapter 4) • Membrane transport (Chapter 5) Figure 5 -26: Energy transfer in living cells Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

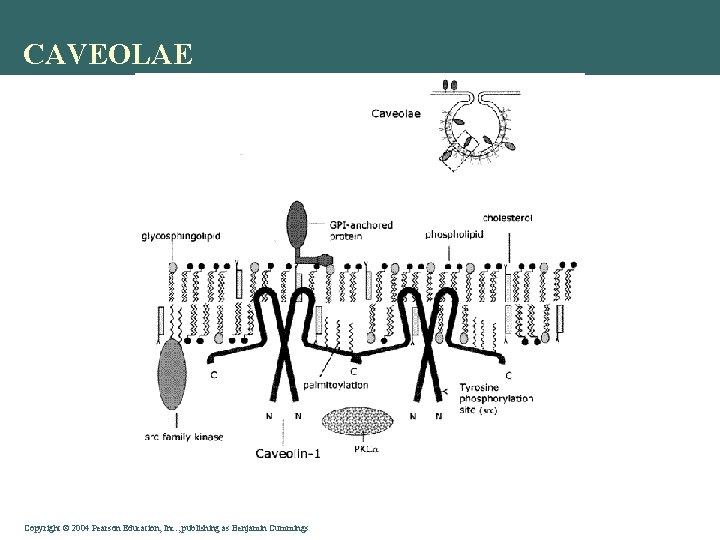
Endocytosis Pathways • Clathrin-mediated endocytosis is mediated by small (approx. 100 nm in diameter) vesicles that have a morphologically characteristic coat made up of a complex of proteins that are mainly associated with the cytosolic protein clathrin. Clathrin-coated vesicles (CCVs) are found in virtually all cells and form domains of the plasma membrane termed clathrin-coated pits. Coated pits can concentrate large extracellular molecules that have different receptors responsible for the receptor-mediated endocytosis of ligands, e. g. low density lipoprotein, transferrin, growth factors, antibodies and many others. • Caveolae are the most common reported non-clathrin-coated plasma membrane buds, which exist on the surface of many, but not all cell types. They consist of the cholesterol-binding protein caveolin (Vip 21) with a bilayer enriched in cholesterol and glycolipids. Caveolae are small (approx. 50 nm in diameter) flask-shape pits in the membrane that resemble the shape of a cave (hence the name caveolae). They can constitute up to a third of the plasma membrane area of the cells of some tissues, being especially abundant in smooth muscle, type I pneumocytes, fibroblasts, adipocytes, and endothelial cells. Uptake of extracellular molecules is also believed to be specifically mediated via receptors in caveolae. • Macropinocytosis, which usually occurs from highly ruffled regions of the plasma membrane, is the invagination of the cell membrane to form a pocket, which then pinches off into the cell to form a vesicle (0. 5– 5 µm in diameter) filled with a large volume of extracellular fluid and molecules within it (equivalent to ~100 CCVs). The filling of the pocket occurs in a non-specific manner. The vesicle then travels into the cytosol and fuses with other vesicles such as endosomes and lysosomes. • Phagocytosis is the process by which cells bind and internalize particulate matter larger than around 0. 75 µm in diameter, such as small-sized dust particles, cell debris, micro-organisms and even apoptotic cells, which only occurs in specialized cells. These processes involve the uptake of larger membrane areas than clathrin-mediated endocytosis and caveolae pathway. Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Vesicles in Membrane Transport • Transcytosis: Moves some molecules and large proteins and particles via endocytosis and exocytosis across cell membrane • Phagosome: The vesicle formed via internalization, in phagocytic cells. Binds and internalizes particles > 0. 75 microns • Phagocytes: An Actin-mediated process. Examples, immune cells • Clatherin-Mediated Endocytosis • Caveolae: Non-Clatherin coated but have caveolin Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

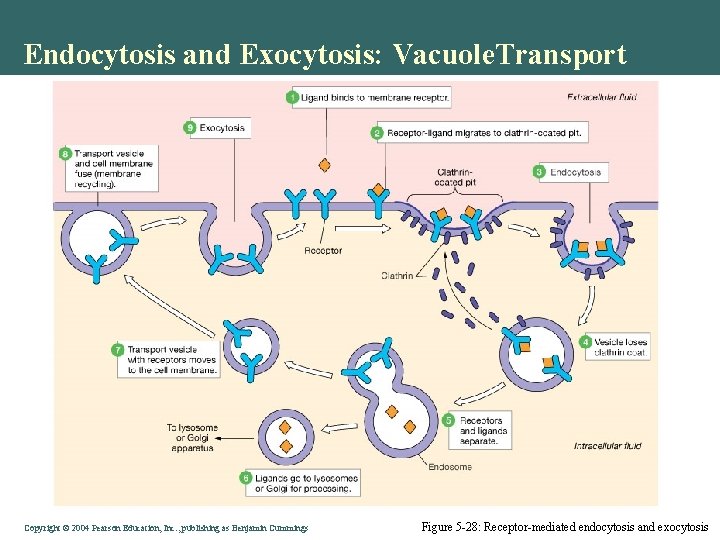
Endocytosis and Exocytosis: Vacuole. Transport • Pinocytosis: non-selective • Receptor mediated: specific substrate Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Endocytosis and Exocytosis: Vacuole. Transport Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -28: Receptor-mediated endocytosis and exocytosis

CAVEOLAE Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

CAVEOLAE Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

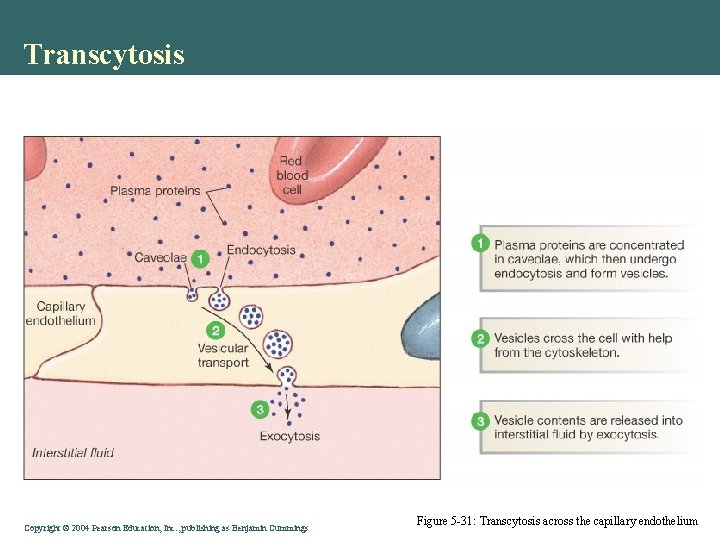
Transcytosis Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -31: Transcytosis across the capillary endothelium

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

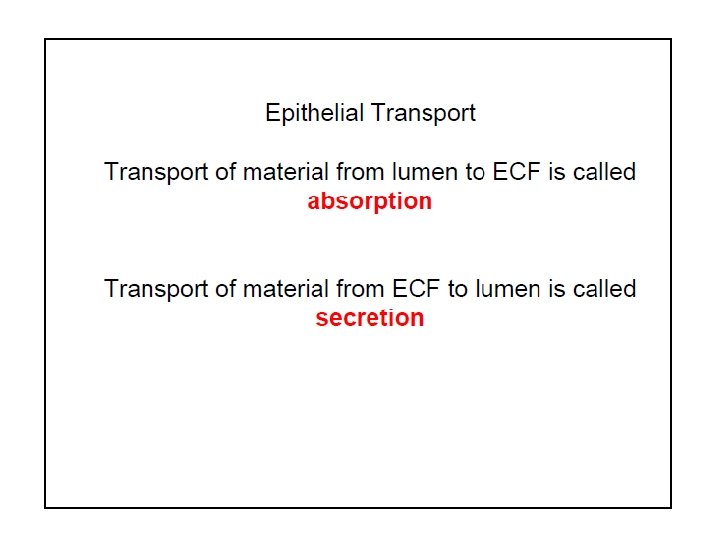
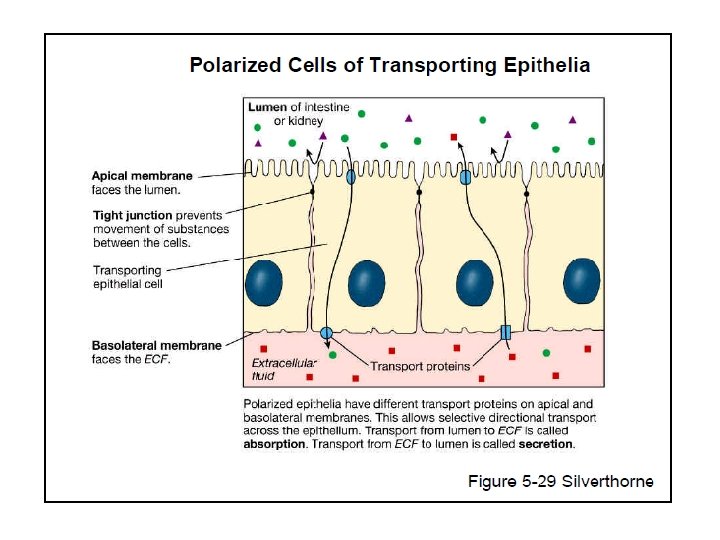
Transepithelial Transport • Cross two membranes • Apical • Basolateral • Absorption • Secretion Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

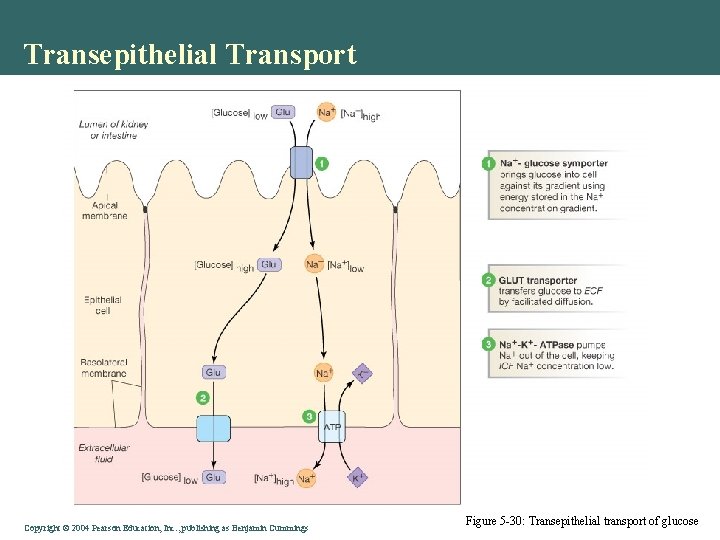
Transepithelial Transport Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -30: Transepithelial transport of glucose

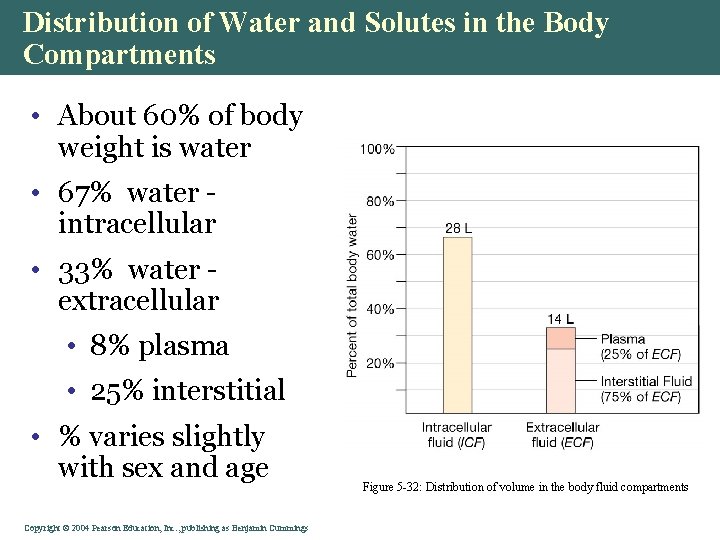
Distribution of Water and Solutes in the Body Compartments • About 60% of body weight is water • 67% water intracellular • 33% water extracellular • 8% plasma • 25% interstitial • % varies slightly with sex and age Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -32: Distribution of volume in the body fluid compartments

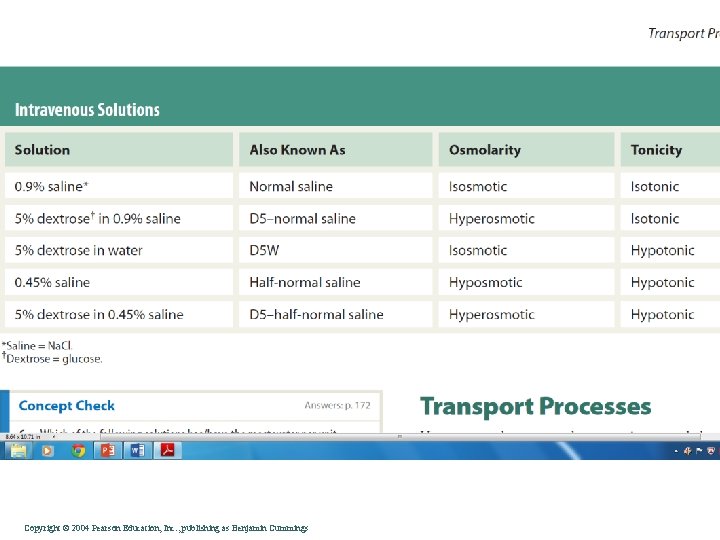
Molarity vs Molality • Molarity is defined as the number of moles of solute per liter of solution. This means that if you have a 1 M solution of some compound, evaporating one liter will cause one mole of the solute to precipitate. • Molality is defined as the number of moles of solute per kilogram of solvent. To make a 1 m solution, you'd take one mole of a substance and add it to 1 Kg of solvent. As a result, the final volume of a 1 m solution will be somewhat more than 1 L if the solvent is Water. Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Molality • Molalities are more convenient than molarities in experiments that involve significant temperature changes. Because the volume of a solution increases when its temperature increases, heating makes the solutions molarity go down- but the molality, which is based on masses rather than volumes, remains unchanged. Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Osmolarity vs Osmolality • Osmolarity is distinct from molarity because it measures moles of solute particles rather than moles of solute. The distinction arises because some compounds can dissociate in solution, whereas others cannot, • Plasma osmolality is affected by changes in water content. In comparison, the plasma osmolarity is slightly less than osmolality, because the total plasma weight (the divisor used for osmolality) excludes the weight of any solutes, while the total plasma volume (used for osmolarity) includes solute content. Otherwise, one liter of plasma would be equivalent to one kilogram of plasma, and plasma osmolarity and plasma osmolality would be equal. However, at low concentrations, the weight of the solute is negligible compared to the weight of the solvent, and osmolarity and osmolality are very similar. Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

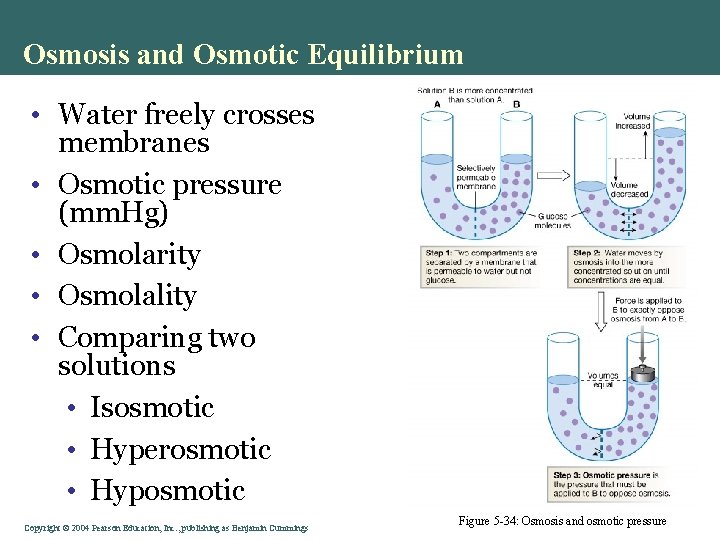
Osmosis and Osmotic Equilibrium • Water freely crosses membranes • Osmotic pressure (mm. Hg) • Osmolarity • Osmolality • Comparing two solutions • Isosmotic • Hyperosmotic • Hyposmotic Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -34: Osmosis and osmotic pressure

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

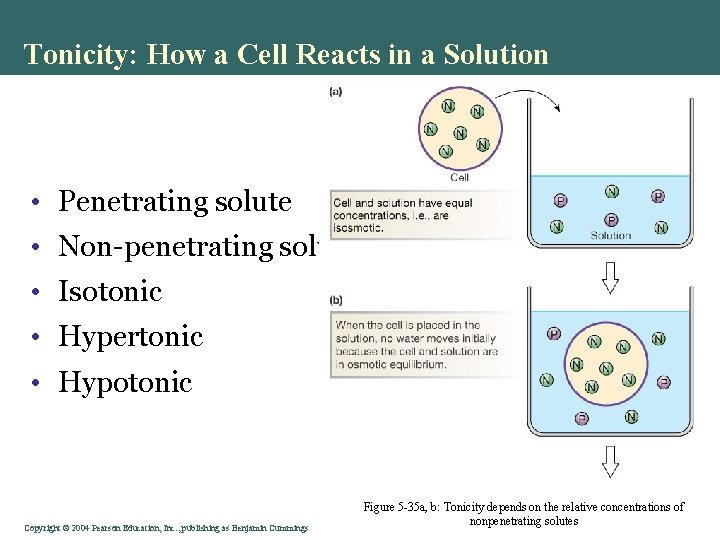
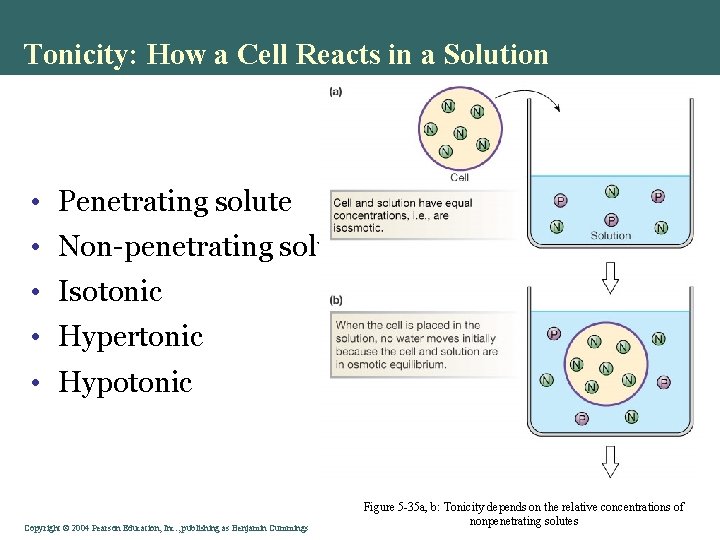
Tonicity: How a Cell Reacts in a Solution • Penetrating solute • Non-penetrating solute • Isotonic • Hypertonic • Hypotonic Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -35 a, b: Tonicity depends on the relative concentrations of nonpenetrating solutes

Tonicity: How a Cell Reacts in a Solution • Penetrating solute • Non-penetrating solute • Isotonic • Hypertonic • Hypotonic Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -35 a, b: Tonicity depends on the relative concentrations of nonpenetrating solutes

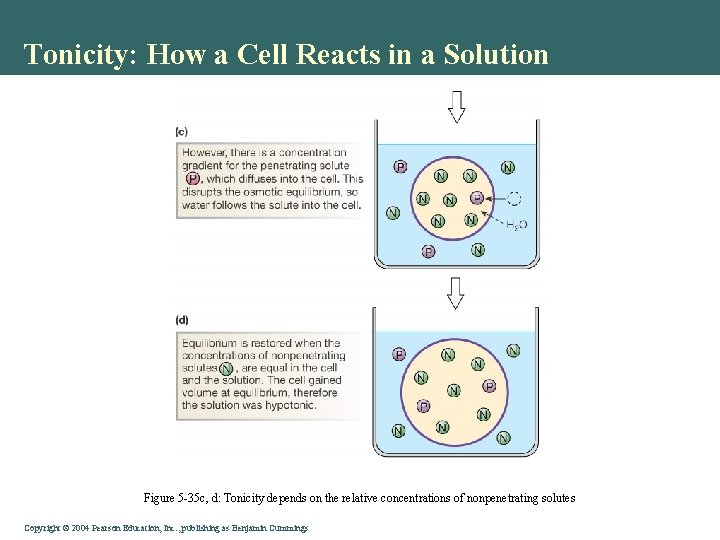
Tonicity: How a Cell Reacts in a Solution Figure 5 -35 c, d: Tonicity depends on the relative concentrations of nonpenetrating solutes Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

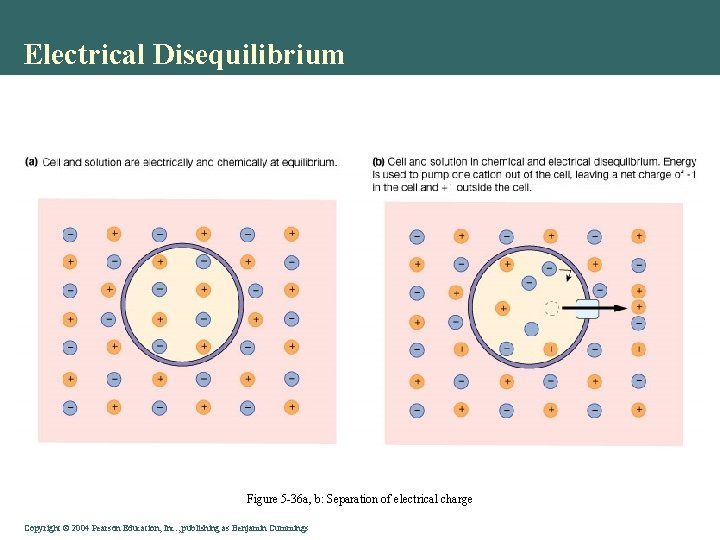
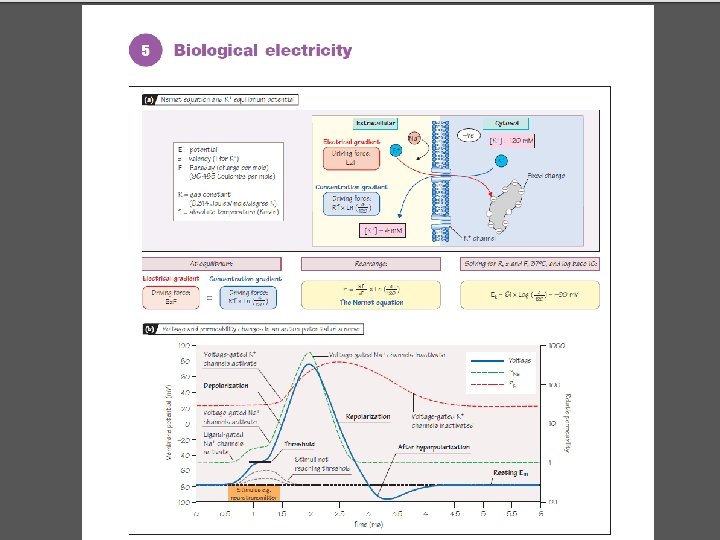
Electrical Disequilibrium • Separation of charged ions • Membrane insulates • Potential • Conduction of signal • Electrochemical gradient Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Electrical Disequilibrium Figure 5 -36 a, b: Separation of electrical charge Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

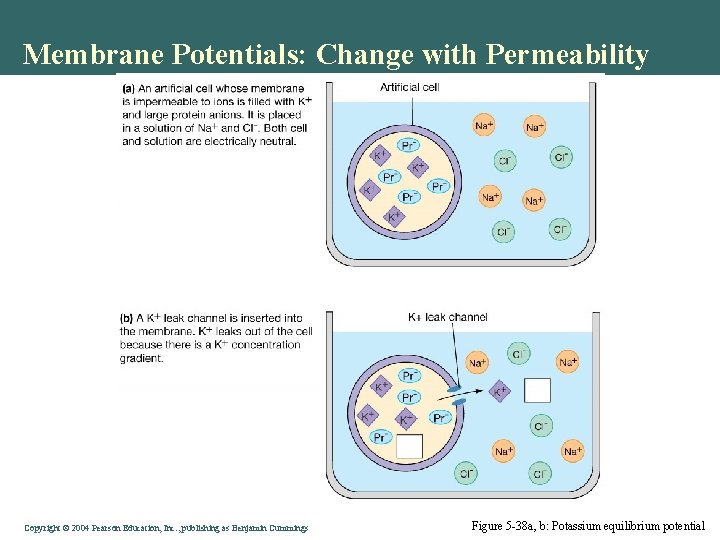
Membrane Potentials: Change with Permeability • Resting • Equilibrium • Channel opening • Voltage gated • ATP gated (leak) Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Membrane Potentials: Change with Permeability Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -38 a, b: Potassium equilibrium potential

Membrane Potentials: Change with Permeability Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings Figure 5 -38 c: Potassium equilibrium potential

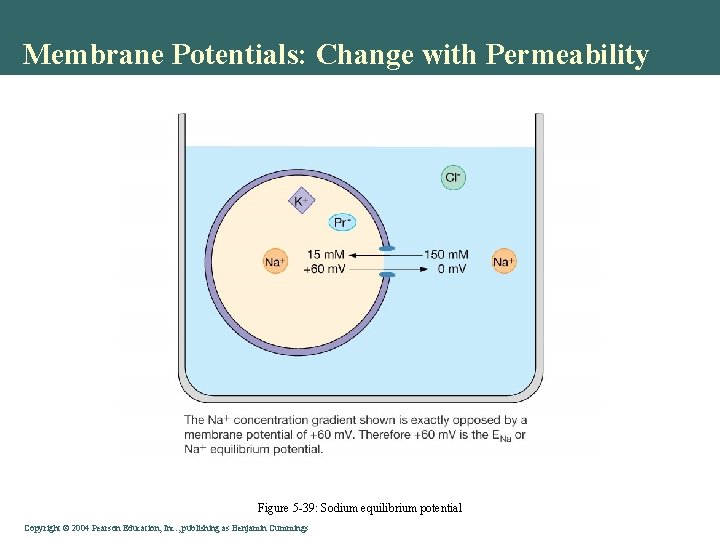
Membrane Potentials: Change with Permeability Figure 5 -39: Sodium equilibrium potential Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

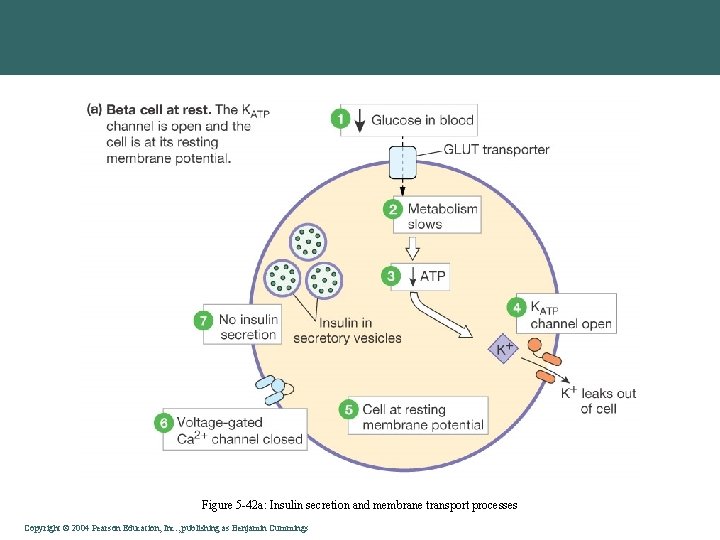
Figure 5 -42 a: Insulin secretion and membrane transport processes Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings

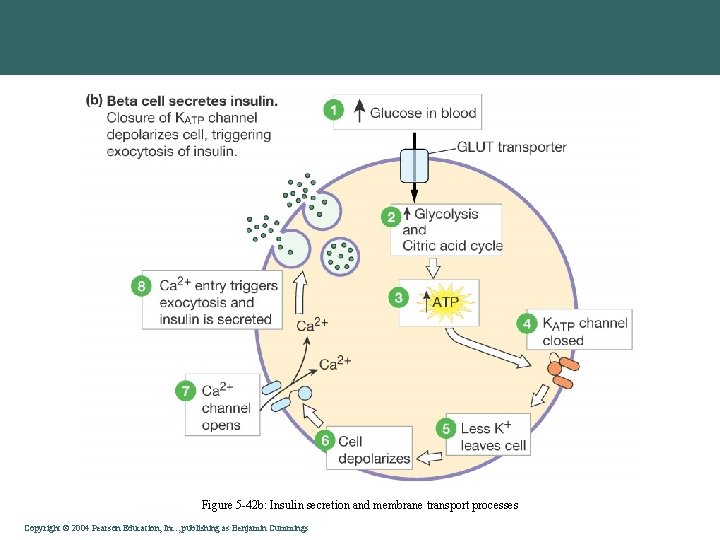
Figure 5 -42 b: Insulin secretion and membrane transport processes Copyright©© 2004 Pearson. Education, Inc. , publishingas as. Benjamin. Cummings