Thiol Redox Systems Petra Bergstrom Xu Zhang Aja

Thiol Redox Systems Petra Bergstrom, Xu Zhang, Aja Harris and Ben Arentson

Outline • Glutaredoxin • Thioredoxin Reductase and Glutathione Reductase • Glutathione

Glutaredoxin (Grx) (thioltransferase) 1. Reduction of protein disulfides 2. Reduction of glutathionylated

Dithiol mechanisms 1. R-S 2 + Grx-(SH)2 → R-(SH)2 + Grx-S 2 2. Grx-S 2 + 2 GSH → Grx-(SH)2 + GSSH Monothiol mechanisms 3. R-S-SG + Grx-(SH)2 → R-(SH) + Grx-S-SG 4. Grx-S-SG + GSH → Grx-(SH)2 + GSSG
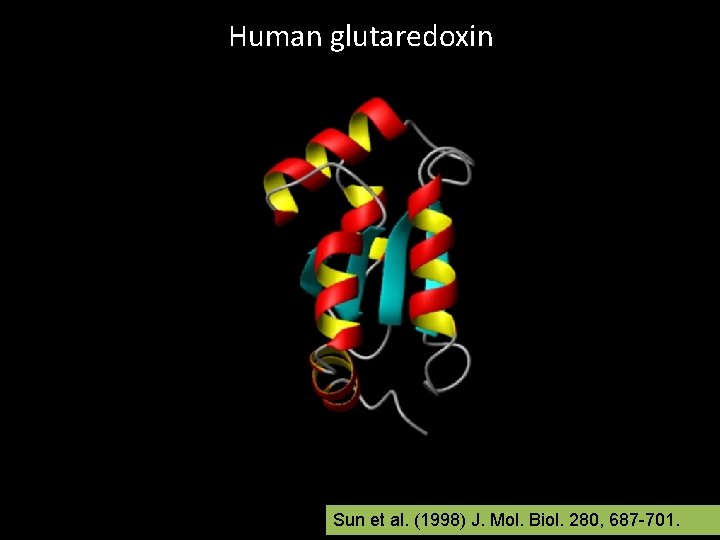
Human glutaredoxin Sun et al. (1998) J. Mol. Biol. 280, 687 -701.
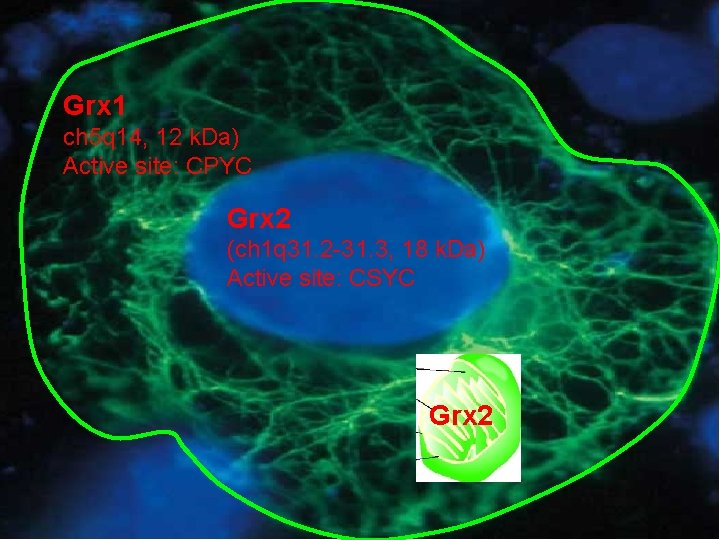
Grx 1 ch 5 q 14, 12 k. Da) Active site: CPYC Grx 2 (ch 1 q 31. 2 -31. 3, 18 k. Da) Active site: CSYC Grx 2

Three protein targets of glutaredoxin. • De-glutathionylation of Actin-SSG • De-glutathionylation of NF 1 • De-glutathionylation of ASK-1 and Akt Shelton et al. 2005
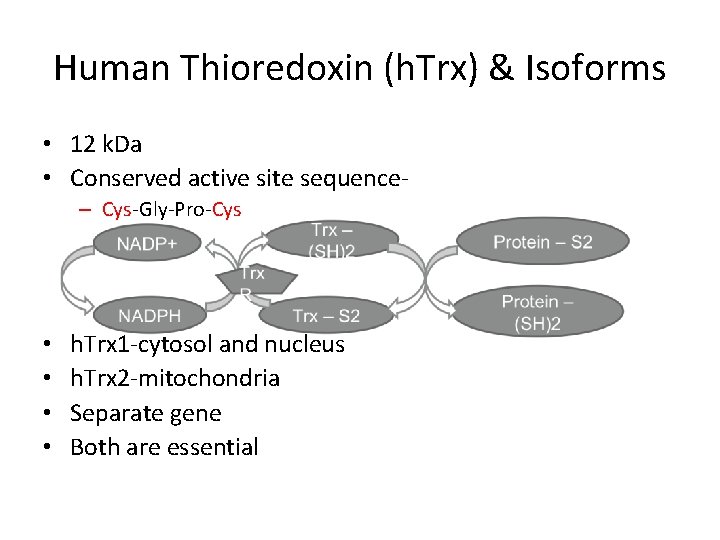
Human Thioredoxin (h. Trx) & Isoforms • 12 k. Da • Conserved active site sequence– Cys-Gly-Pro-Cys • • h. Trx 1 -cytosol and nucleus h. Trx 2 -mitochondria Separate gene Both are essential
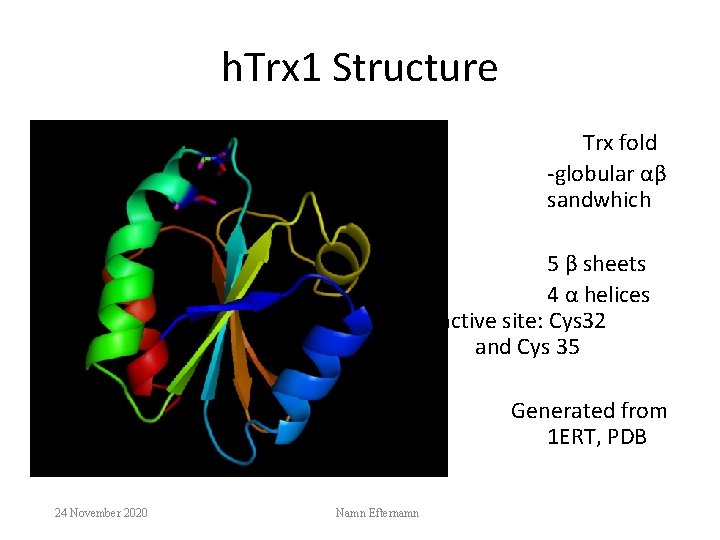
h. Trx 1 Structure Trx fold -globular αβ sandwhich 5 β sheets 4 α helices active site: Cys 32 and Cys 35 Generated from 1 ERT, PDB 24 November 2020 Namn Efternamn

h. Trxs Protein Targets & Protects Cells Against Stress A. Holmgren&J. Lu, Biochemical and Biophysical Research Communication 396(2010) 120 -124
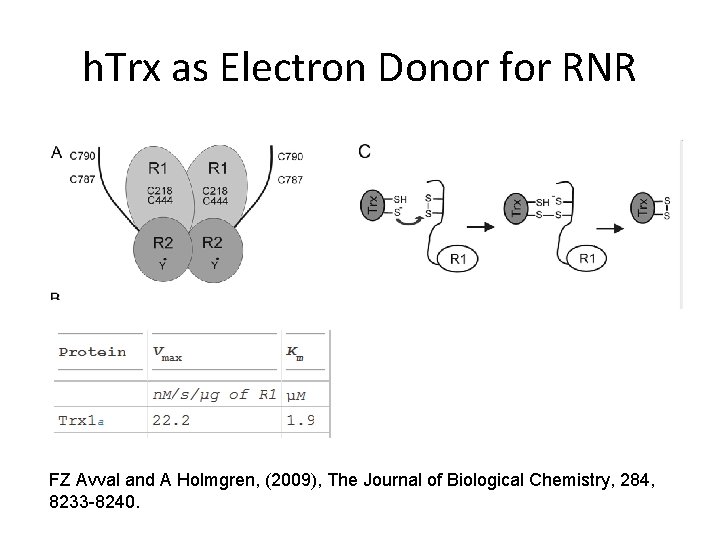
h. Trx as Electron Donor for RNR FZ Avval and A Holmgren, (2009), The Journal of Biological Chemistry, 284, 8233 -8240.

Thioredoxin • Maintains a reduced environment in cytosol of cells with a low redox potential • Regenerate reduced forms of Msrs and Prxs • Stress inducible antioxidant factor

Thioredoxin Reductase • Catalyzes the reduction of oxidized Trx to its reduced form by NADPH • Active site - Cys-Sec-Gly-OH Biterova et al (2005) PNAS. 102: 15018 -15023. Sandalova et al (2001) PNAS. 98: 9533 -9538.
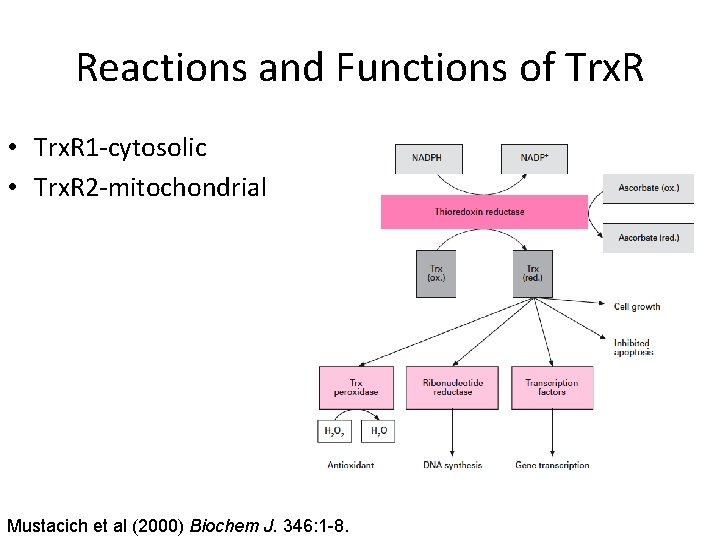
Reactions and Functions of Trx. R • Trx. R 1 -cytosolic • Trx. R 2 -mitochondrial Mustacich et al (2000) Biochem J. 346: 1 -8.

Glutaredoxin • Catalyzes reduction of proteins that are thiolated by GSH • Recycled to GSH via recycling system of NADPH and GR • 2 isoforms in mammals- Grx 1 (cytosolic) and Grx 2 (mitochondrial/nuclear) • GR reduces GSSG to GSH at the expense of NADPH
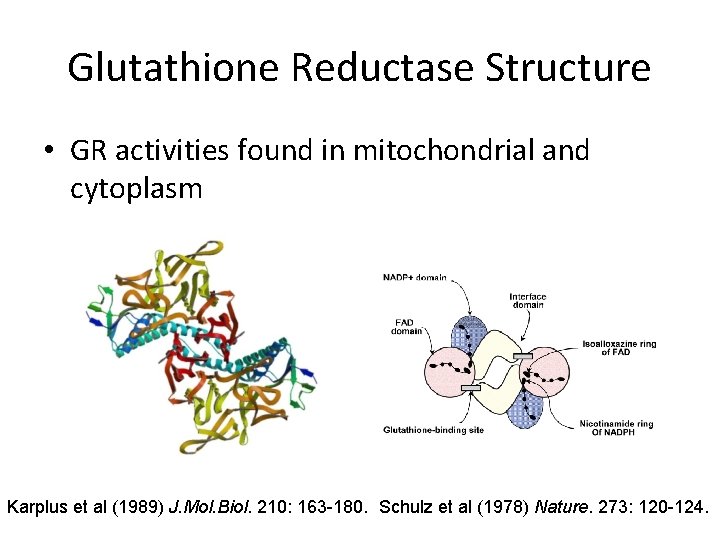
Glutathione Reductase Structure • GR activities found in mitochondrial and cytoplasm Karplus et al (1989) J. Mol. Biol. 210: 163 -180. Schulz et al (1978) Nature. 273: 120 -124.
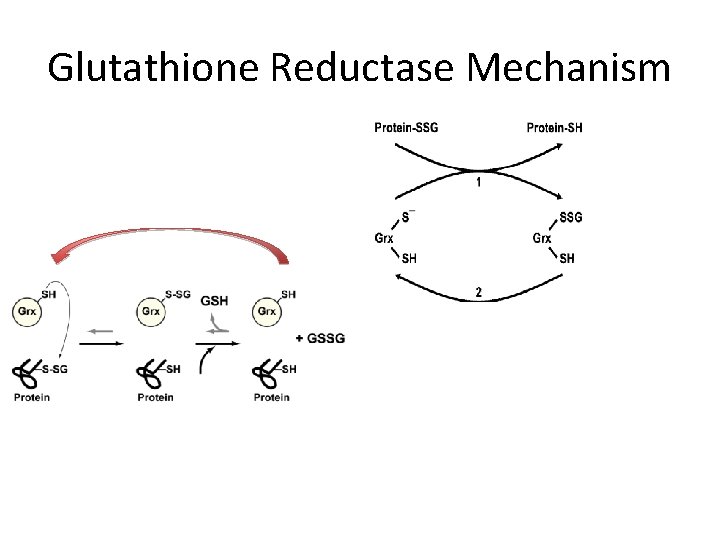
Glutathione Reductase Mechanism
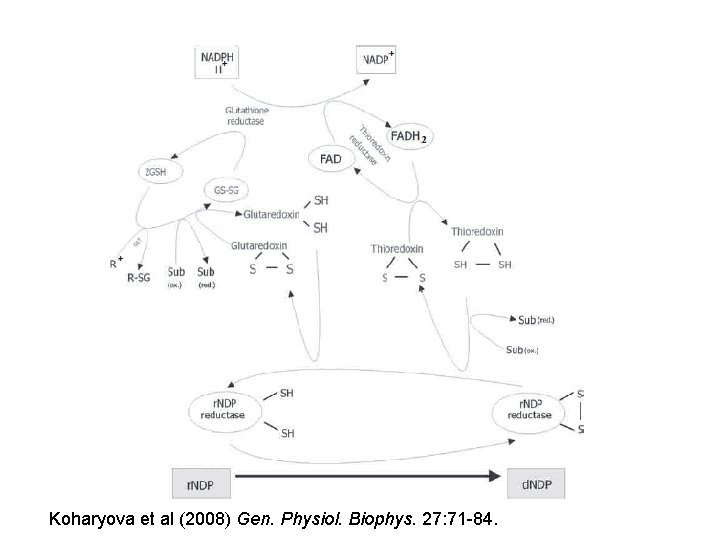
Koharyova et al (2008) Gen. Physiol. Biophys. 27: 71 -84.
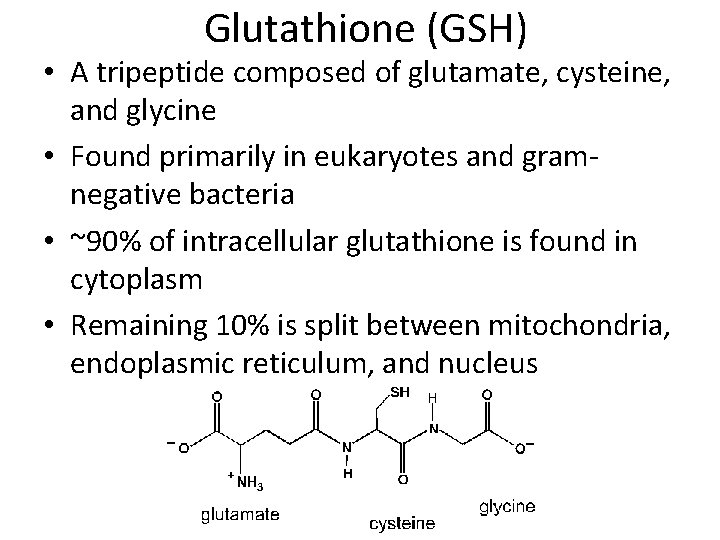
Glutathione (GSH) • A tripeptide composed of glutamate, cysteine, and glycine • Found primarily in eukaryotes and gramnegative bacteria • ~90% of intracellular glutathione is found in cytoplasm • Remaining 10% is split between mitochondria, endoplasmic reticulum, and nucleus
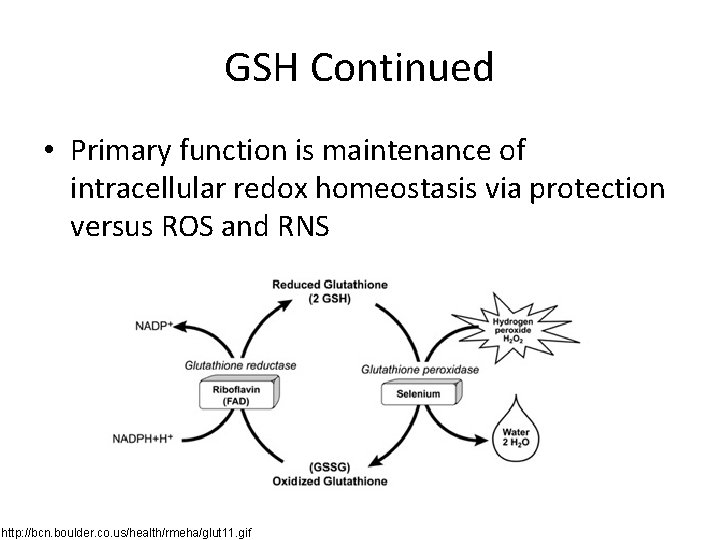
GSH Continued • Primary function is maintenance of intracellular redox homeostasis via protection versus ROS and RNS http: //bcn. boulder. co. us/health/rmeha/glut 11. gif
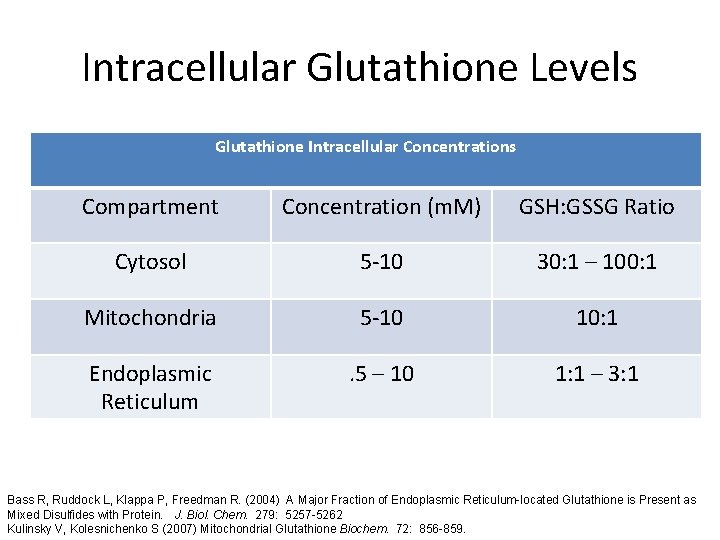
Intracellular Glutathione Levels Glutathione Intracellular Concentrations Compartment Concentration (m. M) GSH: GSSG Ratio Cytosol 5 -10 30: 1 – 100: 1 Mitochondria 5 -10 10: 1 Endoplasmic Reticulum . 5 – 10 1: 1 – 3: 1 Bass R, Ruddock L, Klappa P, Freedman R. (2004) A Major Fraction of Endoplasmic Reticulum-located Glutathione is Present as Mixed Disulfides with Protein. J. Biol. Chem. 279: 5257 -5262 Kulinsky V, Kolesnichenko S (2007) Mitochondrial Glutathione Biochem. 72: 856 -859.
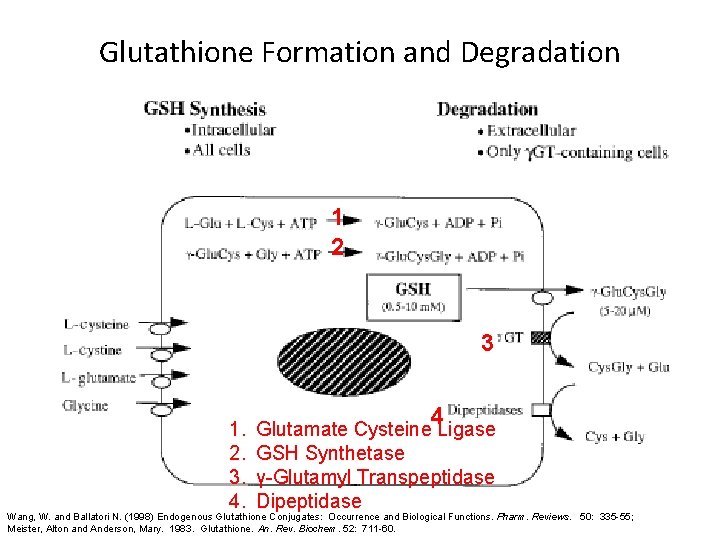
Glutathione Formation and Degradation 1 2 3 1. 2. 3. 4. 4 Glutamate Cysteine Ligase GSH Synthetase γ-Glutamyl Transpeptidase Dipeptidase Wang, W. and Ballatori N. (1998) Endogenous Glutathione Conjugates: Occurrence and Biological Functions. Pharm. Reviews. 50: 335 -55; Meister, Alton and Anderson, Mary. 1983. Glutathione. An. Rev. Biochem. 52: 711 -60.

Glutathionylation • Post-translational modification where GSH is attached to protein via disulfide bond • Involved in regulation of a variety of regulatory, structural, and metabolic proteins Dalle-Donne I, Rossi R, Giustarini D, Colombo R, Milzani A (2007) S-glutathionylation in Protein Redox Regulation. Free Radical Biology 43: 883 -898

Proteins Regulated by Glutathionylation • • α-ketoglutarate dehydrogenase Creatine kinase HIV-1 Protease Thioredoxin
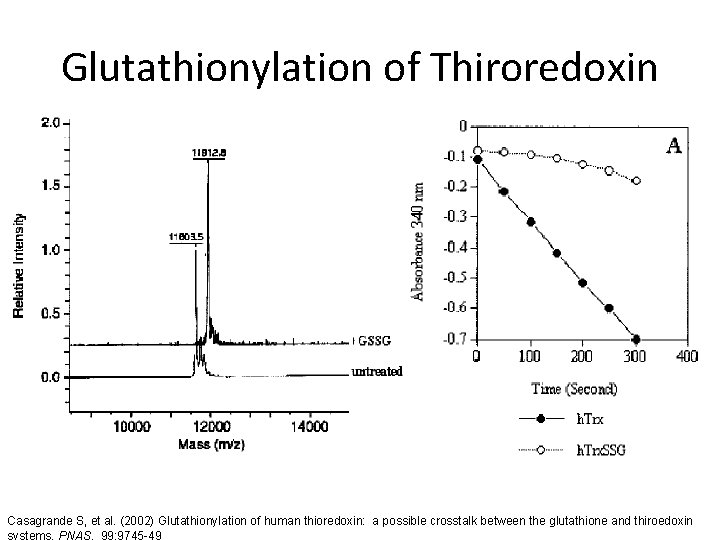
Glutathionylation of Thiroredoxin Casagrande S, et al. (2002) Glutathionylation of human thioredoxin: a possible crosstalk between the glutathione and thiroedoxin systems. PNAS. 99: 9745 -49

Questions?
- Slides: 26