Think back to last big teach Remember Last

Think back to last big teach. . .

Remember. . • Last time we look at some of the properties of waves. Used terms such as wave length and frequency. • Now we will apply our understanding. • Topics in physics are often not stand alone, they link with other topics!
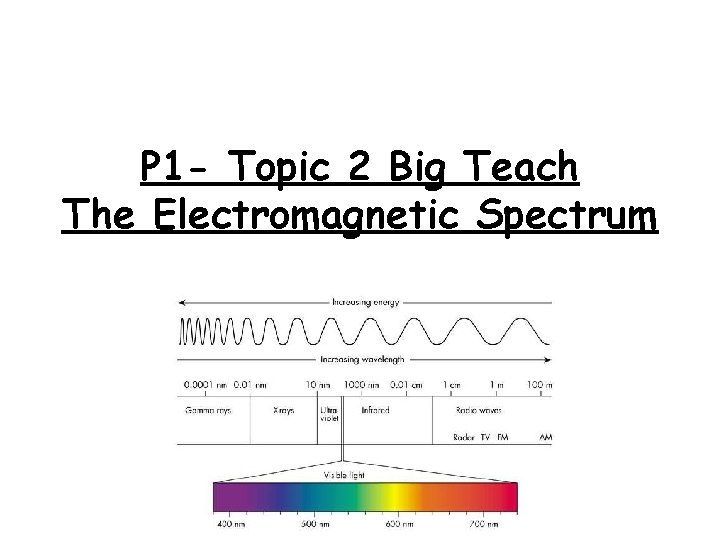
P 1 - Topic 2 Big Teach The Electromagnetic Spectrum
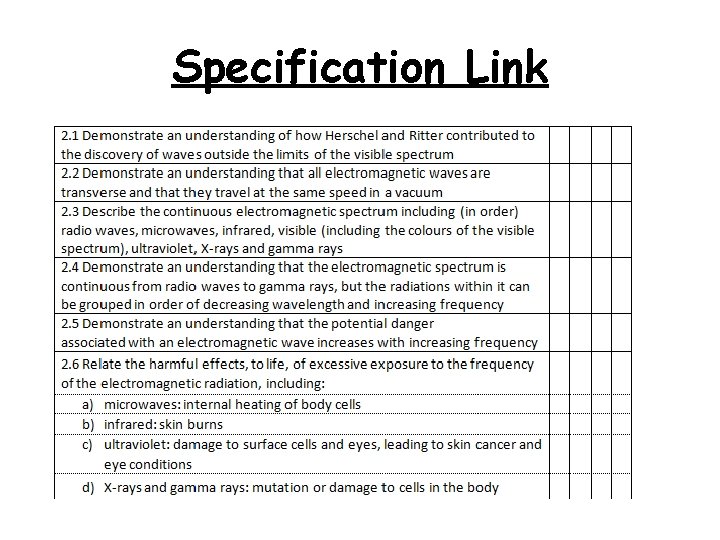
Specification Link
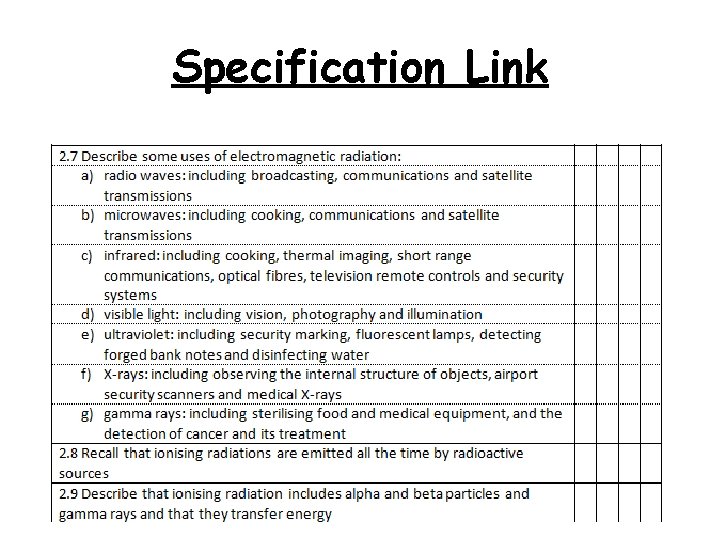
Specification Link

Herschel and Ritter. . . • https: //www. youtube. com/watch? time_conti nue=112&v=-Jdhy. SH 2 PMc
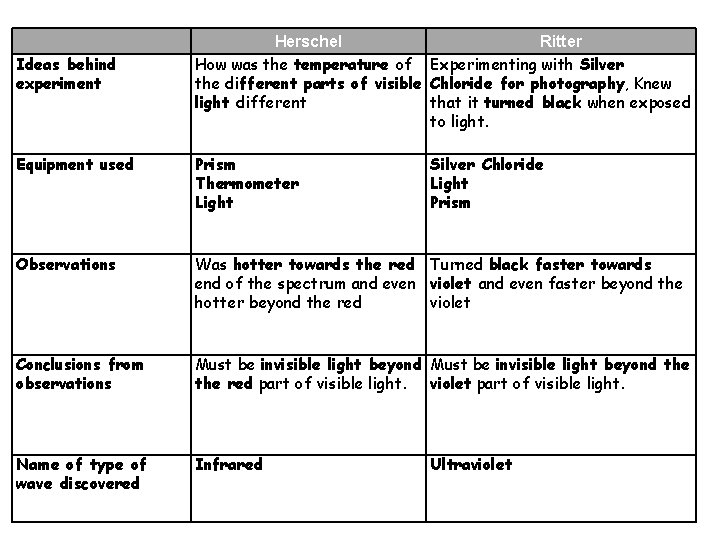
Ideas behind experiment Herschel Ritter How was the temperature of Experimenting with Silver the different parts of visible Chloride for photography, Knew light different that it turned black when exposed to light. Equipment used Prism Thermometer Light Silver Chloride Light Prism Observations Was hotter towards the red Turned black faster towards end of the spectrum and even violet and even faster beyond the hotter beyond the red violet Conclusions from observations Must be invisible light beyond the red part of visible light. violet part of visible light. Name of type of wave discovered Infrared Ultraviolet

Things to remember… • You can bullet point 6 mark questions, just make sure they are full sentences using correct scientific language • They have asked you to discuss two experiments, do one at a time!

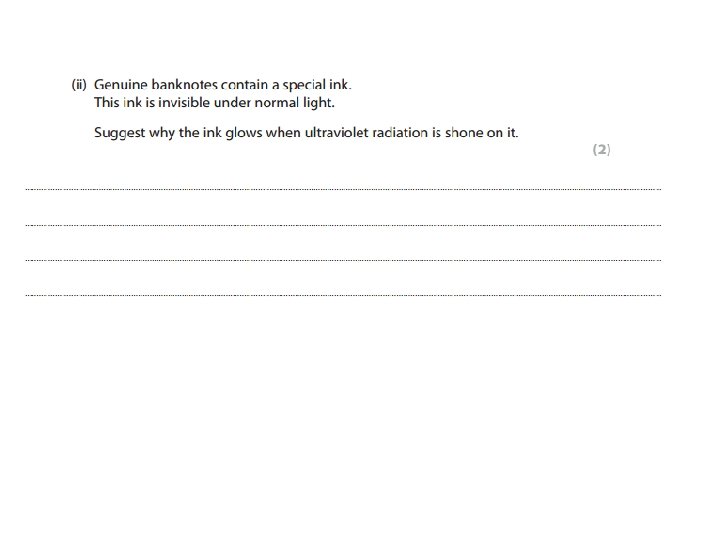
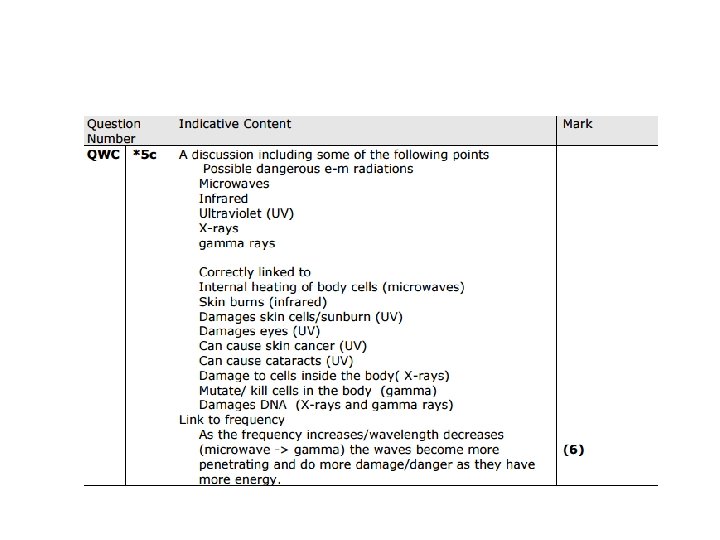
• An explanation including some of the following points Results obtained: • Herschel: temperature on thermometer • Ritter: speed of darkening of silver chloride paper Trend of results: • Herschel: hotter towards red end • Ritter: quicker towards blue/violet end Extension of experiment to get more results: • Herschel: measure below red; found it even hotter • Ritter: measure above blue/violet; paper darkened quicker • Conclusion: · Herschel: Must be radiation below red ( Infra Red) · Ritter: Must be radiation above blue/violet ( UV)
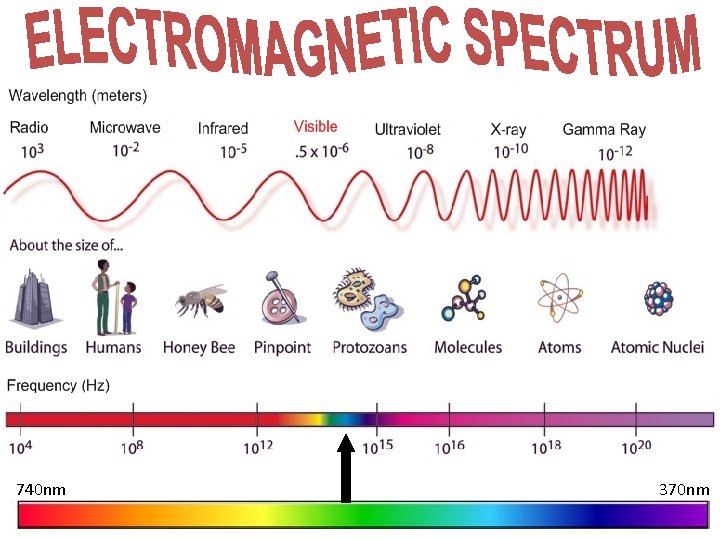
740 nm 370 nm

The Electromagnetic Spectrum Key points you need to know. . . • Electromagnetic waves that have long wavelengths have lower frequencies and less energy. • And electromagnetic waves that have short wavelengths have high frequencies and high energy. • LEARN THE ORDER!! • Rich Men In Vegas Use e. Xpensive Gadgets (learn the order!) Think of your own pneumonic.
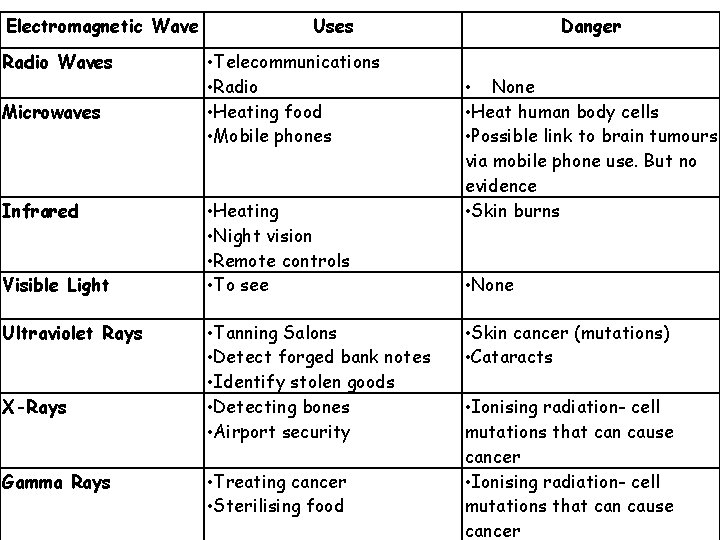
Electromagnetic Wave Radio Waves Microwaves Infrared Visible Light Ultraviolet Rays X-Rays Gamma Rays Uses • Telecommunications • Radio • Heating food • Mobile phones • Heating • Night vision • Remote controls • To see • Tanning Salons • Detect forged bank notes • Identify stolen goods • Detecting bones • Airport security • Treating cancer • Sterilising food Danger • None • Heat human body cells • Possible link to brain tumours via mobile phone use. But no evidence • Skin burns • None • Skin cancer (mutations) • Cataracts • Ionising radiation- cell mutations that can cause cancer
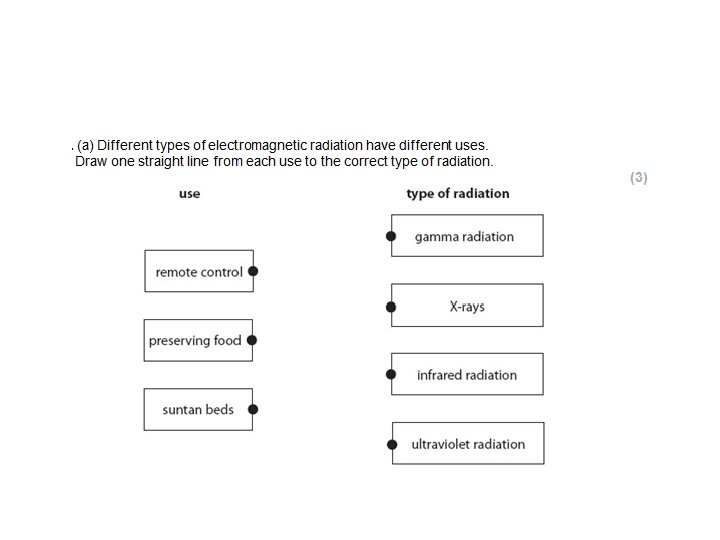

Not always so simple!
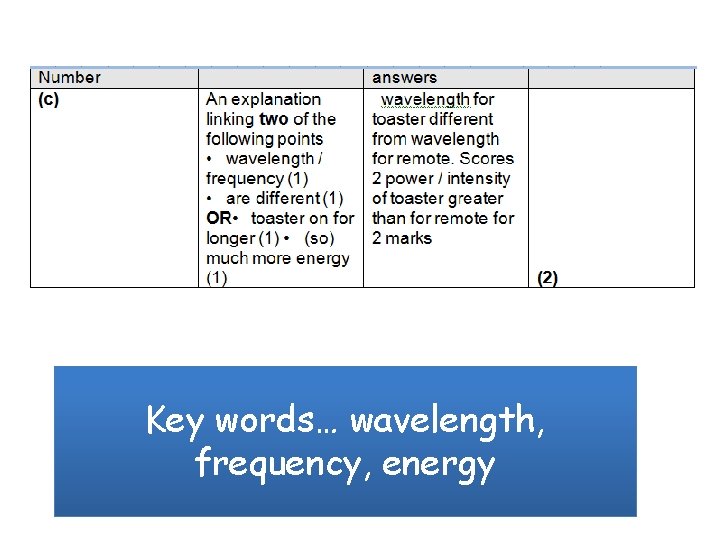
Key words… wavelength, frequency, energy


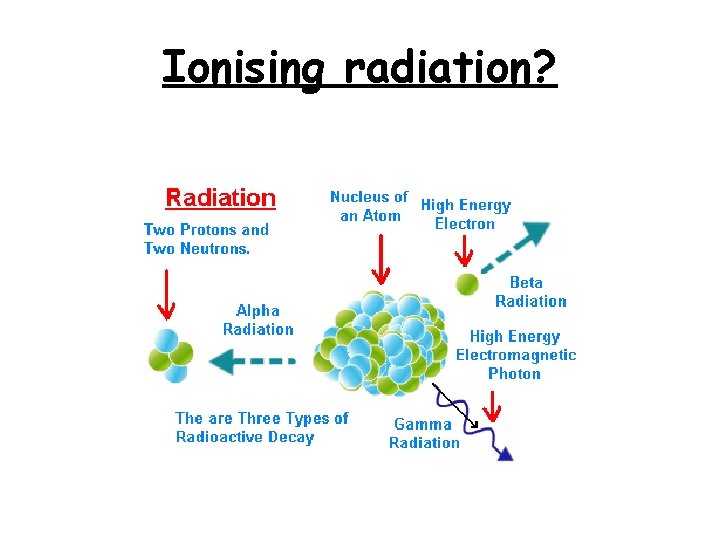
Ionising radiation?
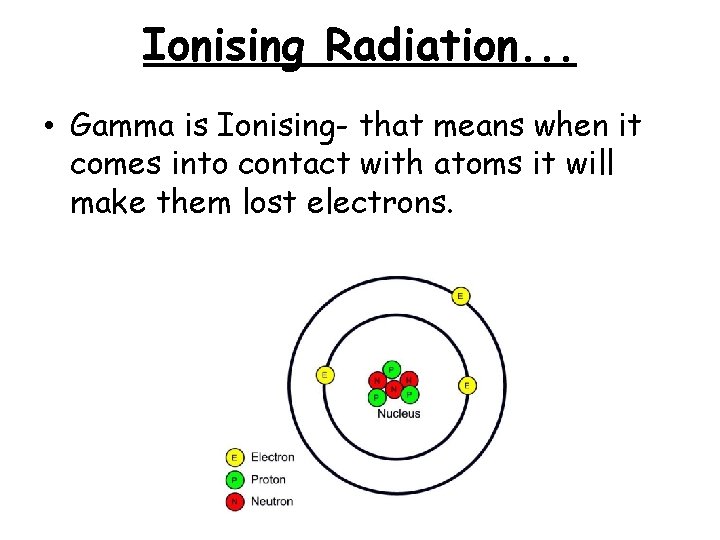
Ionising Radiation. . . • Gamma is Ionising- that means when it comes into contact with atoms it will make them lost electrons.

Why is this bad? • We are made up of atoms. When atoms lose electrons they become ions. Ions are reactive. • This causes the cells to mutate (because they are made up of atoms), which can lead to cancer!

Is Gamma the only type of ionising radiation?

• https: //www. youtube. com/watch? v=K_zc 1 W KT 0 CA

What you need to know. . . • Ionising radiation is emitted by radioactive sources. • Alpha, beta and gamma are all ionising. • Alpha and beta are particles and gamma is a wave. • Ionising radiation can knock of electrons in atoms which can lead to cell mutation and eventually cancer.


Want more questions? • Past papers on singhscience and an exam booklet which is split up into the 6 different topics in P 1.
- Slides: 28