Thin film technology early stage growth sami franssilaaalto











- Slides: 37

Thin film technology, early stage growth sami. franssila@aalto. fi

This lecture Vapor from source to vicinity of substrate Vapor condensation on substrate, adsorption and reevaporation Nucleation and coalescence Epitaxy Strained layer epitaxy

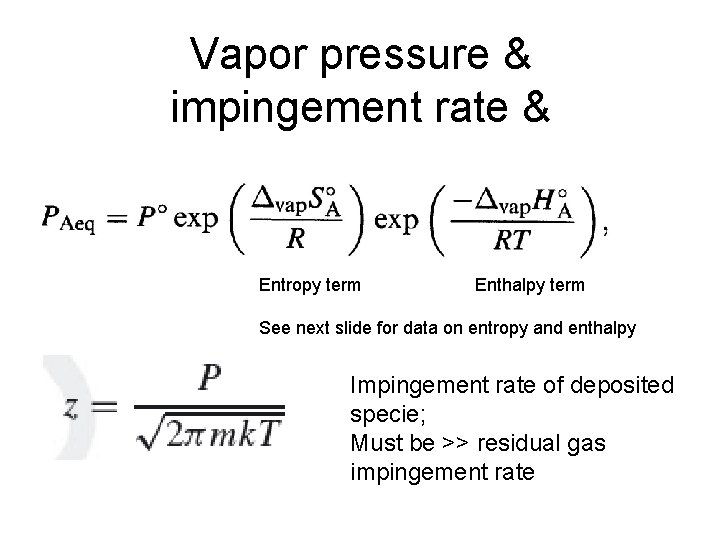
Vapor pressure & impingement rate & Entropy term Enthalpy term See next slide for data on entropy and enthalpy Impingement rate of deposited specie; Must be >> residual gas impingement rate


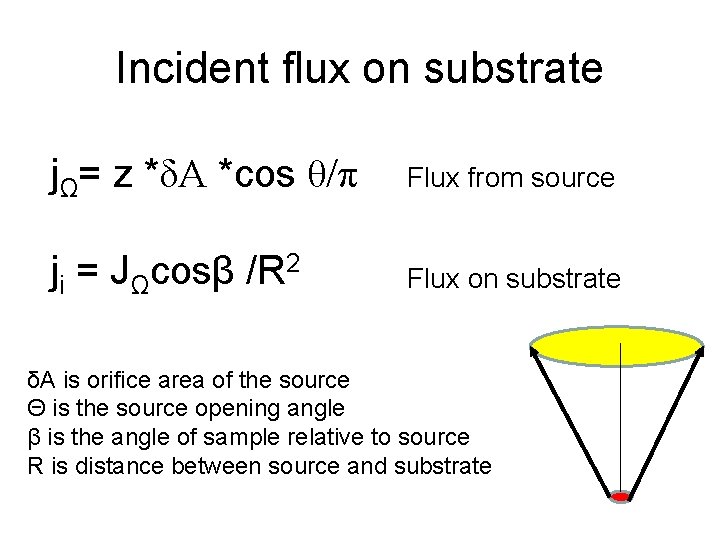
Incident flux on substrate jΩ= z *δA *cos θ/π Flux from source ji = JΩcosβ /R 2 Flux on substrate δA is orifice area of the source Θ is the source opening angle β is the angle of sample relative to source R is distance between source and substrate

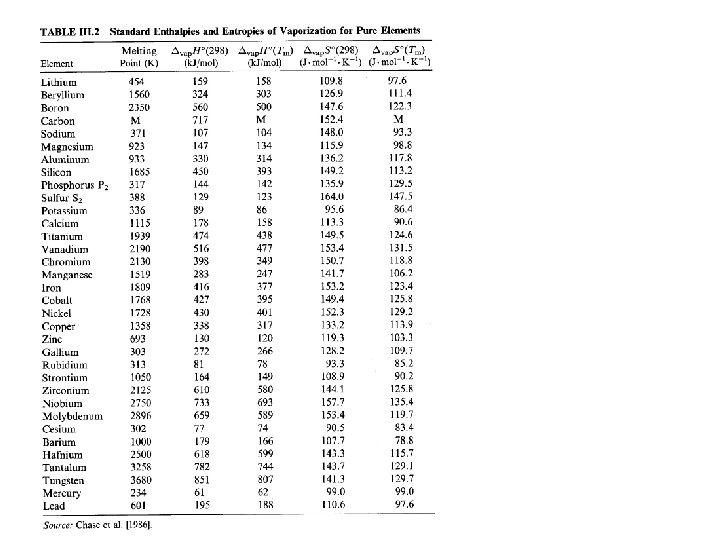
Magnesium deposition rate Magnesium, 900 K, P=2. 47 Pa; Impingement rate z = 440 Å-2/s jΩ= 440 Å-2/s * cos 0 o * 1 cm 2 = 1. 4*1018 s-1 At 10 cm distance from evaporation source: (β=0 gives maximum flux) ji = JΩcosβ /R 2 = 1. 4*1018 s-1 * cos 0 o / (10 cm)2 ji =1. 4 Å-2/s

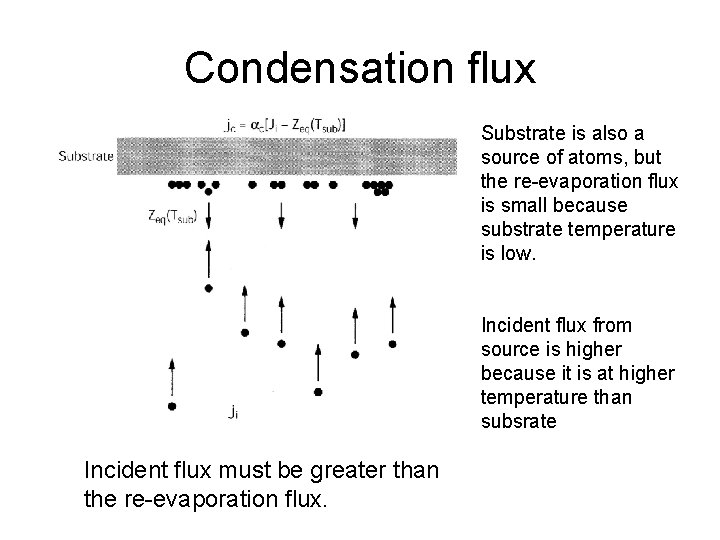
Condensation flux Substrate is also a source of atoms, but the re-evaporation flux is small because substrate temperature is low. Incident flux from source is higher because it is at higher temperature than subsrate Incident flux must be greater than the re-evaporation flux.

Bathroom analogy You start showering Water vapor is generated, but the bathroom air can accomodate some water vapor After some time, saturation is reached and water phase is formed Water starts condensing on cool surfaces, leading to mirror fogging But if you keep mirror warm, that will prevent fogging A clean mirror stays free of condensate longer, dirt acts as nucleation site

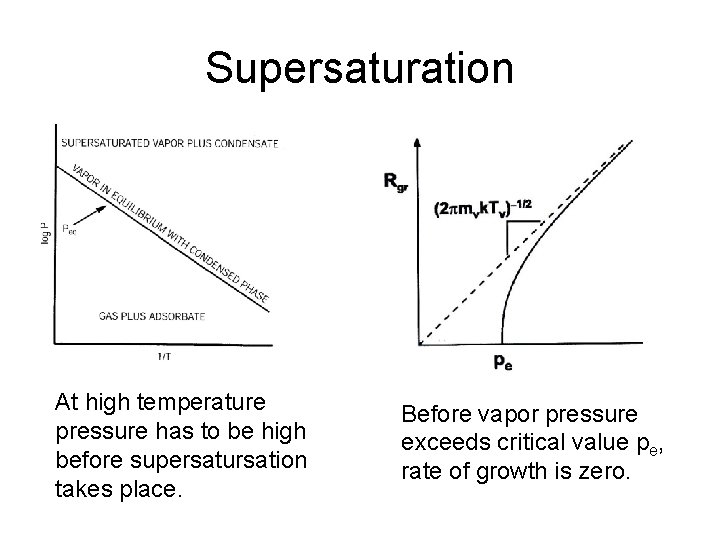
Supersaturation At high temperature pressure has to be high before supersatursation takes place. Before vapor pressure exceeds critical value pe, rate of growth is zero.

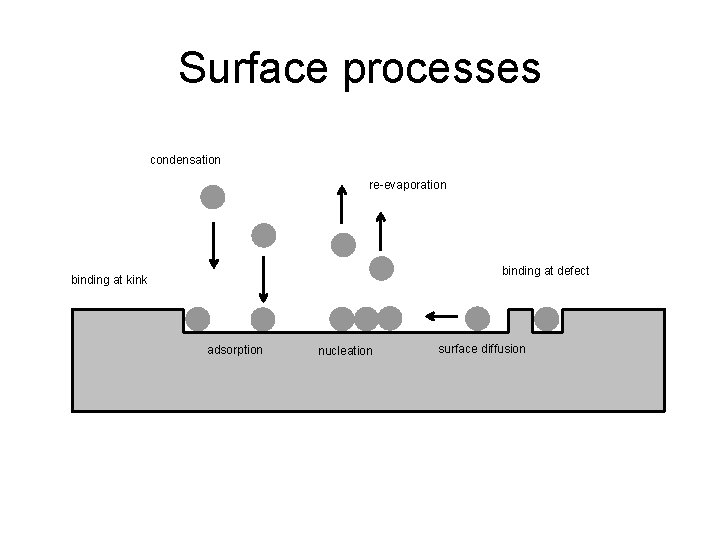
Surface processes condensation re-evaporation binding at defect binding at kink adsorption nucleation surface diffusion

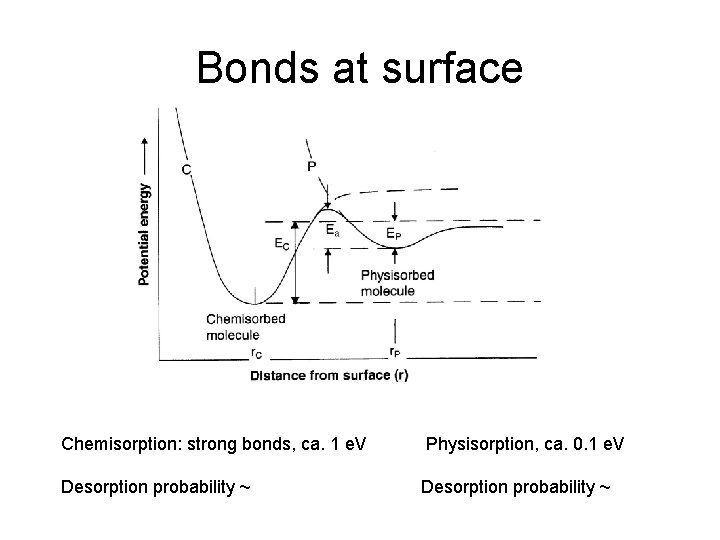
Bonds at surface Chemisorption: strong bonds, ca. 1 e. V Physisorption, ca. 0. 1 e. V Desorption probability ~

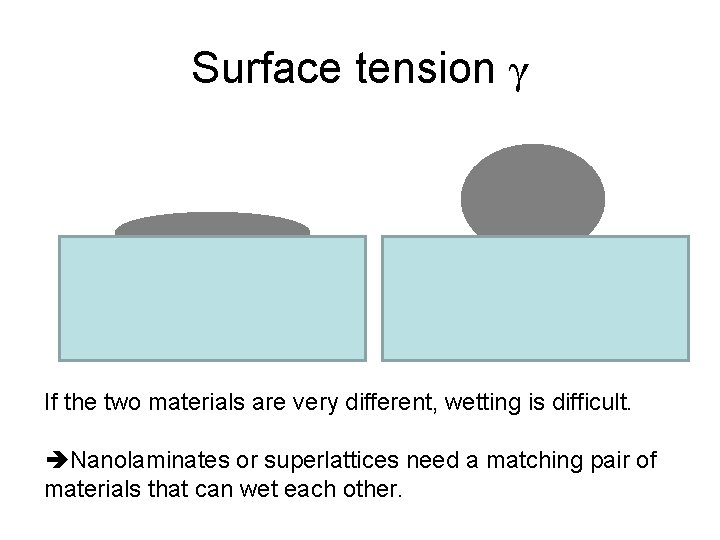
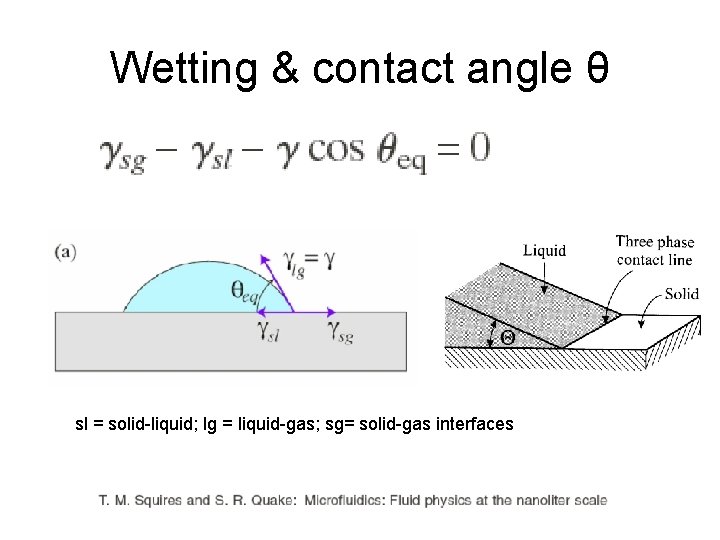
Surface tension γ If the two materials are very different, wetting is difficult. Nanolaminates or superlattices need a matching pair of materials that can wet each other.

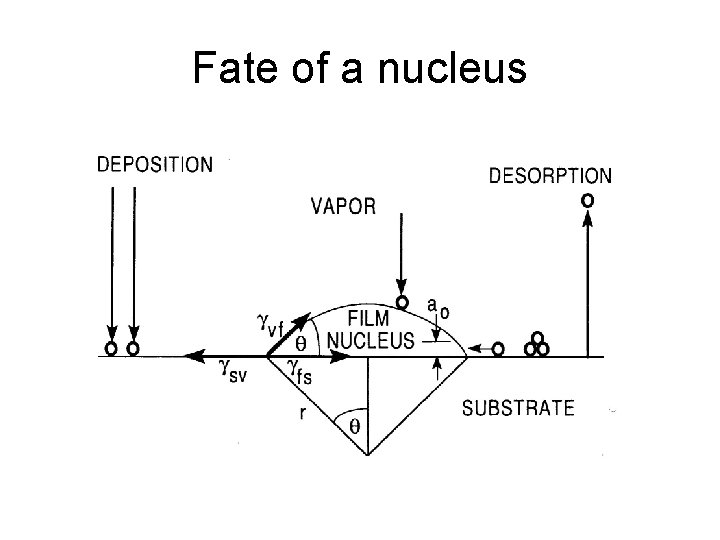
Fate of a nucleus

Wetting & contact angle θ sl = solid-liquid; lg = liquid-gas; sg= solid-gas interfaces

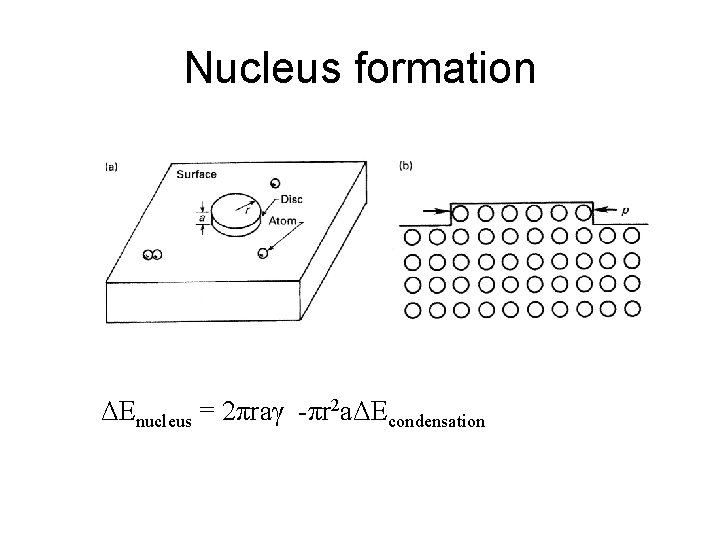
Nucleus formation ΔEnucleus = 2πraγ -πr 2 aΔEcondensation

Kettle analogy Homogenous nucleation is rare, because it is energetically unfvourable, and because perfectly flat surfaces are rare. In a kettle, water does not boil uniformly thru the volume of water. Boiling starts at scratches at kettle bottom. Condensation is a phase transition just like boiling.

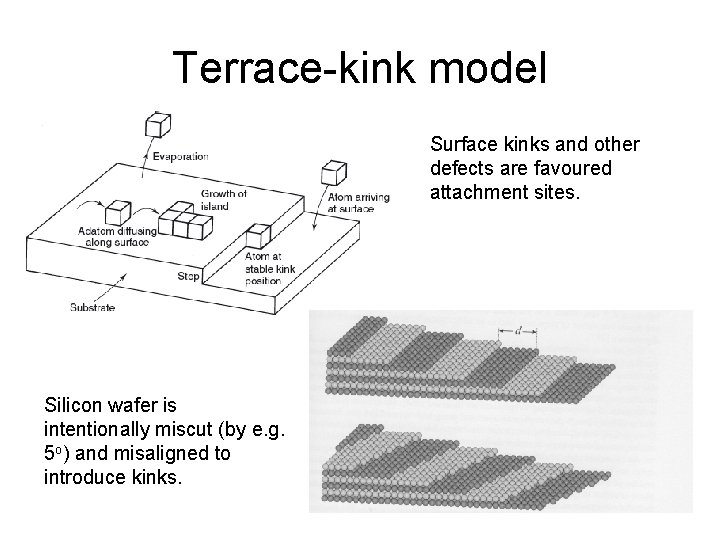
Terrace-kink model Surface kinks and other defects are favoured attachment sites. Silicon wafer is intentionally miscut (by e. g. 5 o) and misaligned to introduce kinks.

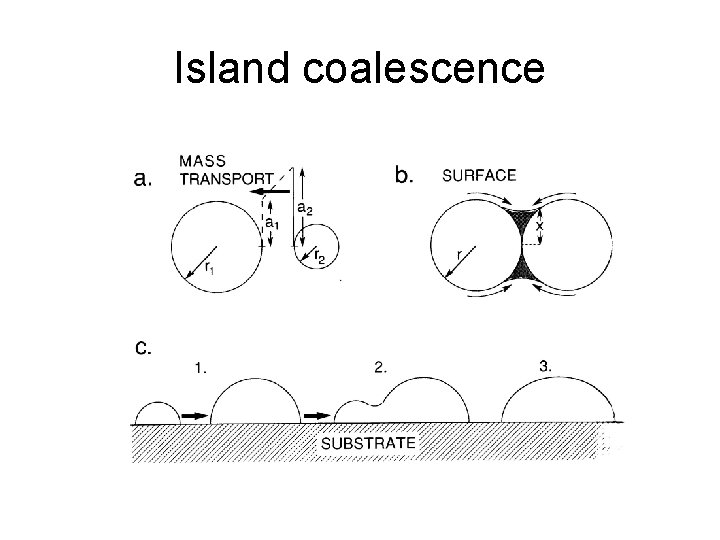
Island coalescence

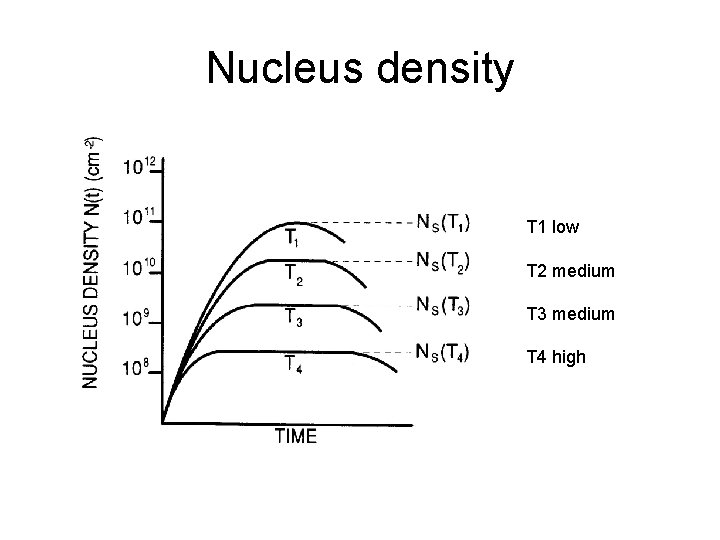
Nucleus density T 1 low T 2 medium T 3 medium T 4 high

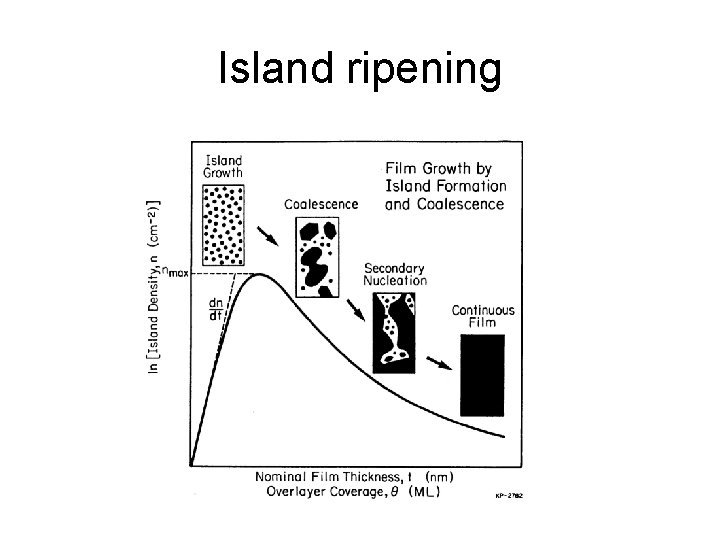
Island ripening

Island growth and coalescence Cu on Na. Cl

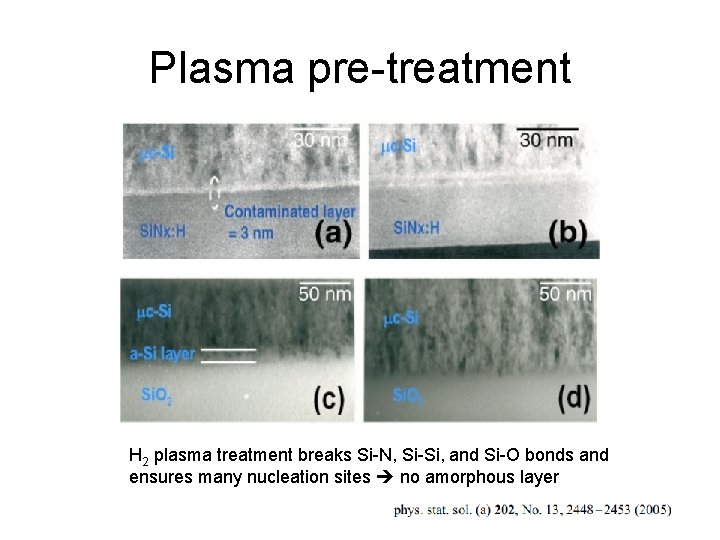
Plasma pre-treatment H 2 plasma treatment breaks Si-N, Si-Si, and Si-O bonds and ensures many nucleation sites no amorphous layer

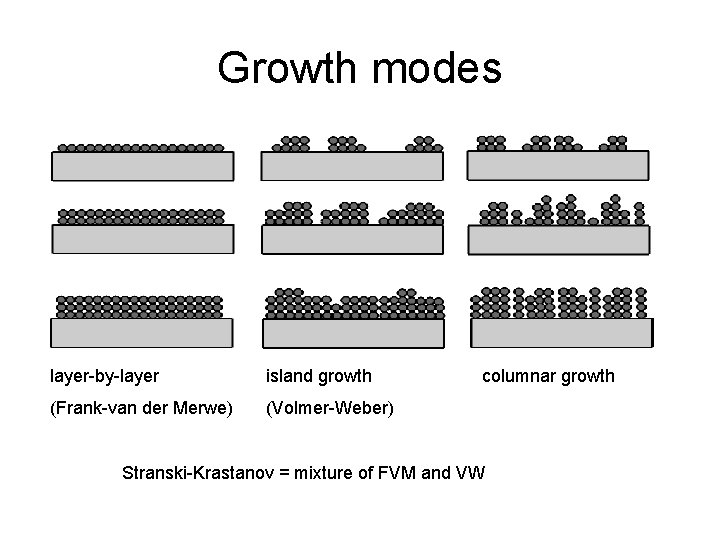
Growth modes layer-by-layer island growth (Frank-van der Merwe) (Volmer-Weber) columnar growth Stranski-Krastanov = mixture of FVM and VW

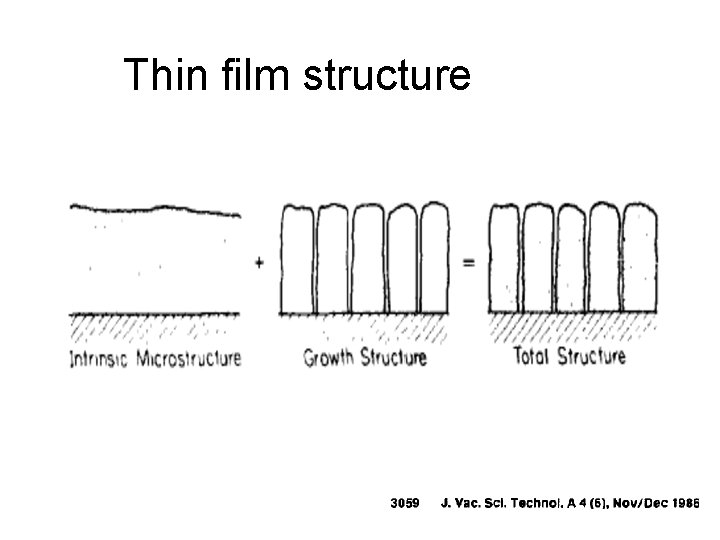
Thin film structure

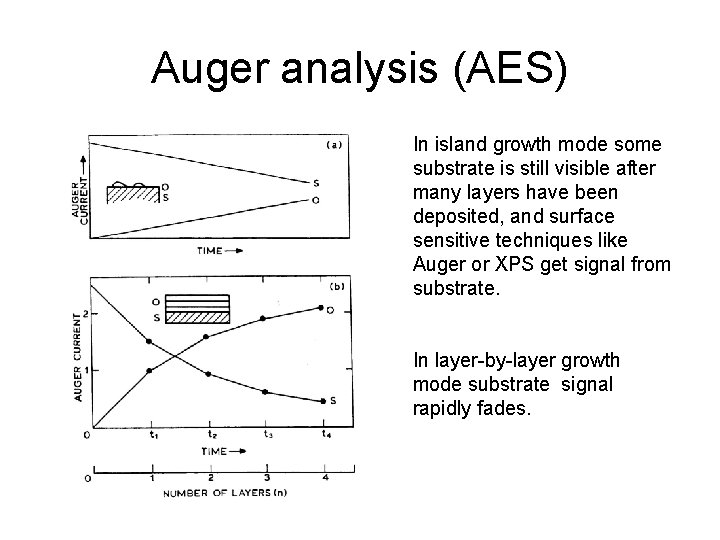
Auger analysis (AES) In island growth mode some substrate is still visible after many layers have been deposited, and surface sensitive techniques like Auger or XPS get signal from substrate. In layer-by-layer growth mode substrate signal rapidly fades.

Which growth mode ? Film substrate potential W = Ufs/Uff is a ratio of filmsubstrate and film-film atom interaction potentials. W >>1 layer-by-layer growth W>1 Stranski-Krastanov W~1 island growth

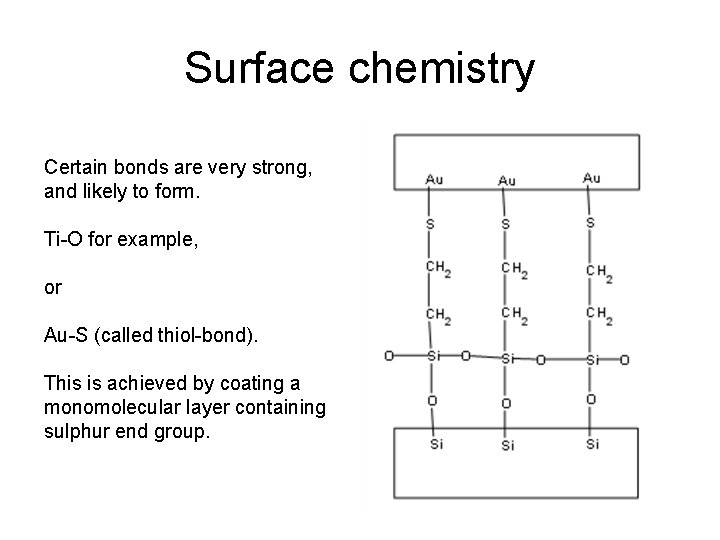
Surface chemistry Certain bonds are very strong, and likely to form. Ti-O for example, or Au-S (called thiol-bond). This is achieved by coating a monomolecular layer containing sulphur end group.

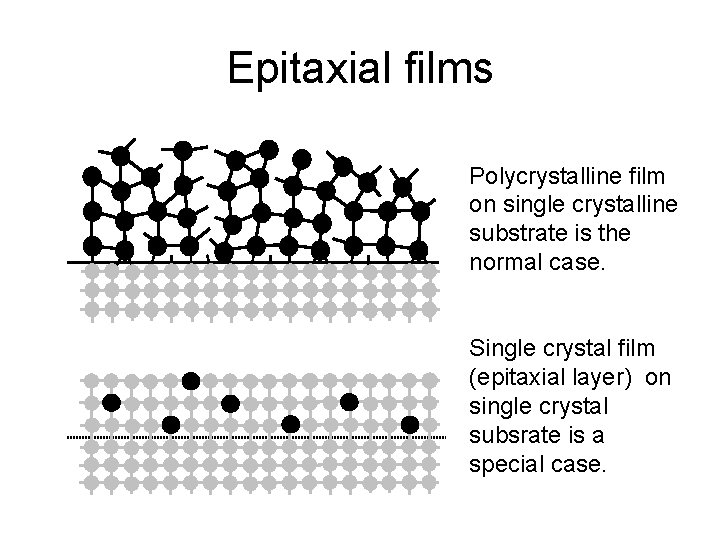
Epitaxial films Polycrystalline film on single crystalline substrate is the normal case. Single crystal film (epitaxial layer) on single crystal subsrate is a special case.

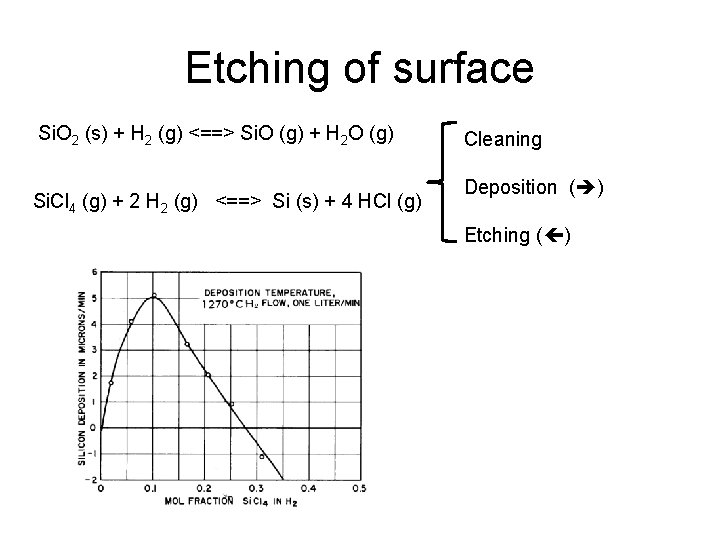
Etching of surface Si. O 2 (s) + H 2 (g) <==> Si. O (g) + H 2 O (g) Si. Cl 4 (g) + 2 H 2 (g) <==> Si (s) + 4 HCl (g) Cleaning Deposition ( ) Etching ( )

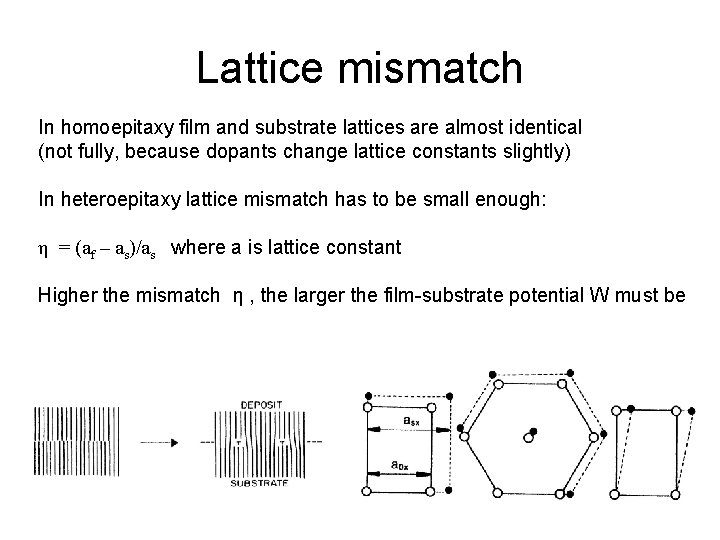
Lattice mismatch In homoepitaxy film and substrate lattices are almost identical (not fully, because dopants change lattice constants slightly) In heteroepitaxy lattice mismatch has to be small enough: η = (af – as)/as where a is lattice constant Higher the mismatch η , the larger the film-substrate potential W must be

Epitaxy Heteroepitaxy: Aluminum on spinel Heteroepitaxy: Cd. Te on Ga. As. Stacking faults are arranged at regular intervals at the interface and epilayer is perfect

Epitaxy considerations Thermal mismatch: CTE differences cause strains Diffusion: high T produces surface diffusion which helps epitaxy, but too high T produces diffusion of film atoms into substrate Usable temperature range e. g. 0. 35 T – 0. 4 T Defects: Misfit dislocations can propagate into film Impurities (from the gas phase): Can act as faux nucleation sites, prevent surface diffusion, generate defects, cause oxidation (oxygen, water vapor) Surface impurites (not removed by cleaning): Native oxides completely prevent epi, and must be in situ removed

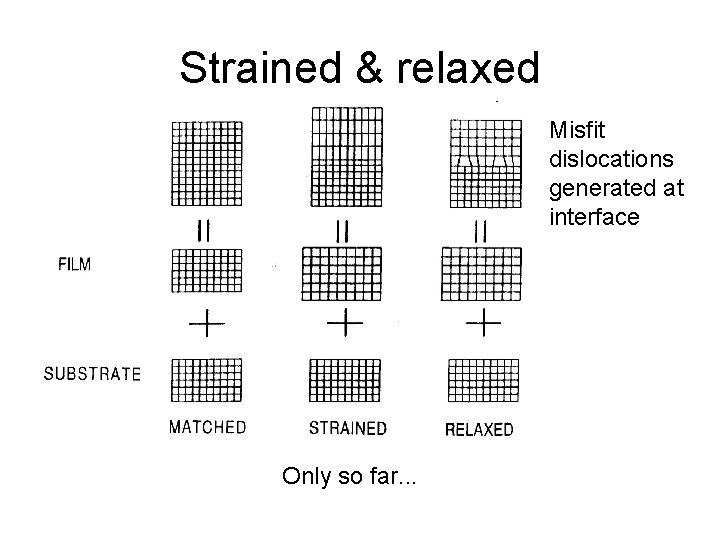
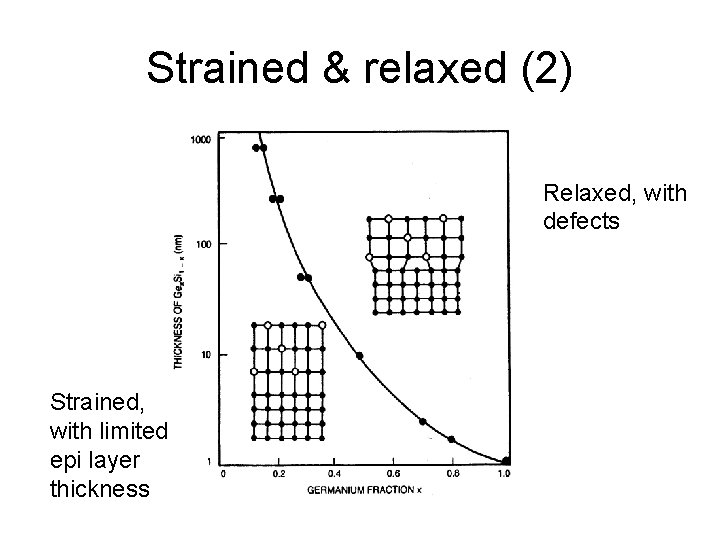
Strained & relaxed Misfit dislocations generated at interface Only so far. . .

Strained & relaxed (2) Relaxed, with defects Strained, with limited epi layer thickness

Epi on epi If Y 2 O 3 grows epitaxially on silicon, then silicon grows epitaxially on Y 2 O 3.

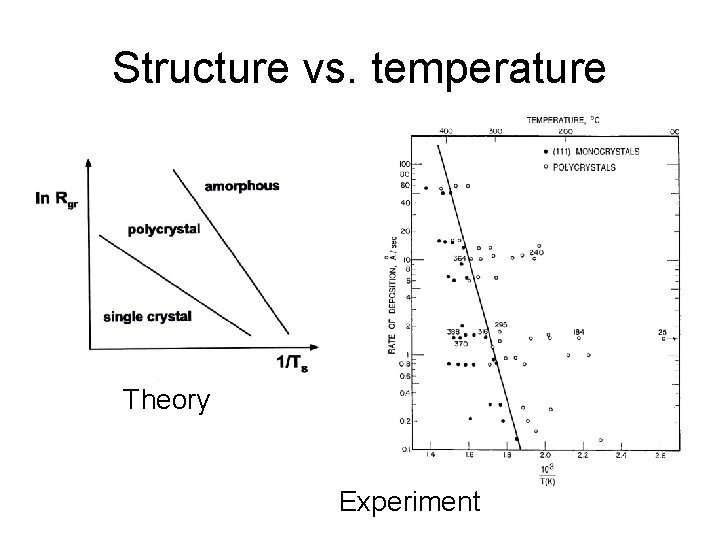
Structure vs. temperature Theory Experiment

Major factors affecting film structure 1. Deposition rate 2. Vacuum level 3. Substrate temperature 4. Surface structure and chemistry