These particles have something in common Blood cells
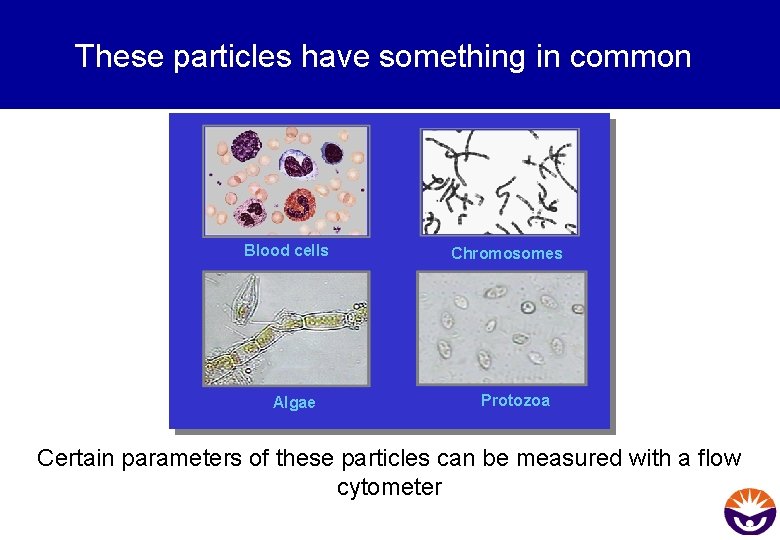
These particles have something in common Blood cells Algae Chromosomes Protozoa Certain parameters of these particles can be measured with a flow cytometer

Which parameters can be measured? § the relative size (Forward Scatter - FSC) § the granularity or complexity (Side Scatter - SSC) § the fluorescence intensity (FL 1, FL 2, up to FL X)
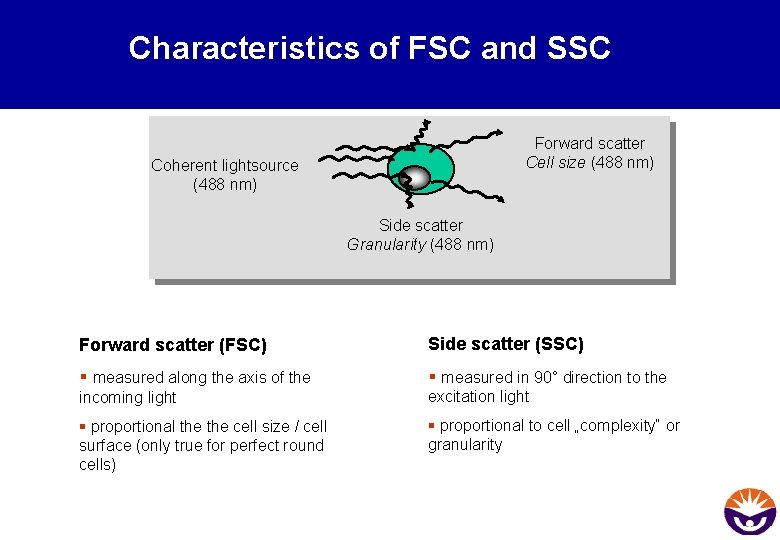
Characteristics of FSC and SSC Forward scatter Cell size (488 nm) Coherent lightsource (488 nm) Side scatter Granularity (488 nm) Forward scatter (FSC) Side scatter (SSC) § measured along the axis of the § measured in 90° direction to the incoming light excitation light § proportional the cell size / cell surface (only true for perfect round cells) § proportional to cell „complexity“ or granularity
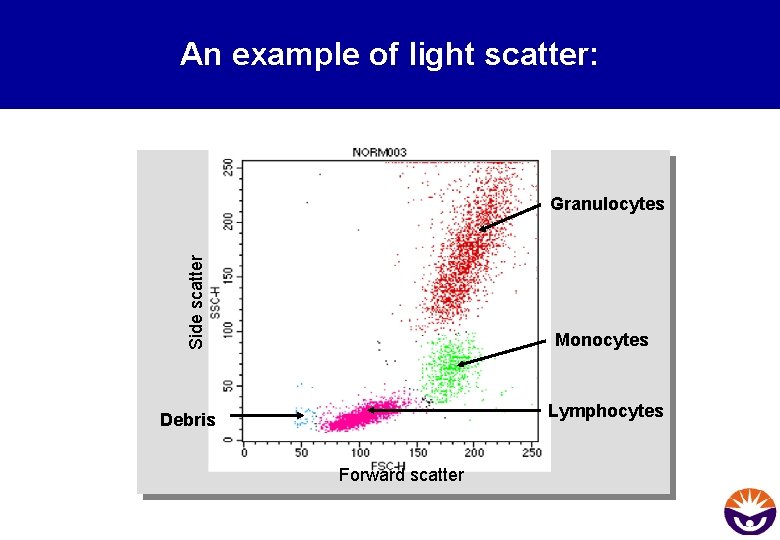
An example of light scatter: Side scatter Granulocytes Monocytes Lymphocytes Debris Forward scatter
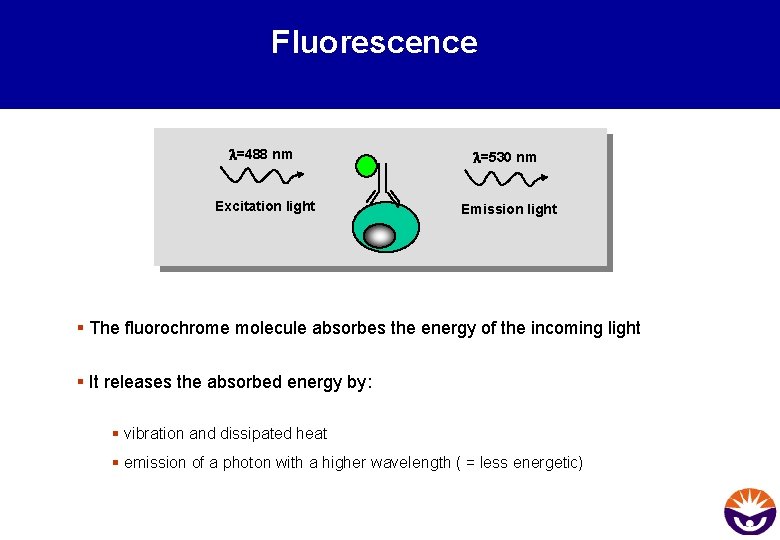
Fluorescence l=488 nm l=530 nm Excitation light Emission light § The fluorochrome molecule absorbes the energy of the incoming light § It releases the absorbed energy by: § vibration and dissipated heat § emission of a photon with a higher wavelength ( = less energetic)
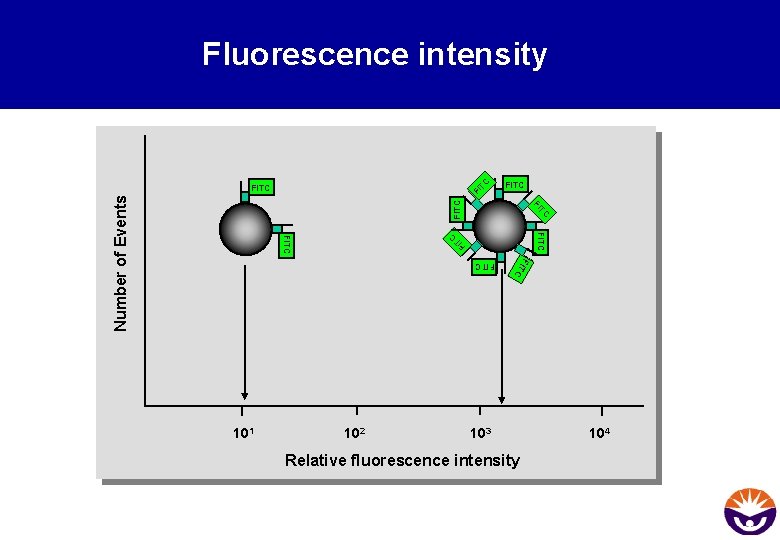
TC Fluorescence intensity FI FITC TC FI FITC Number of Events FITC TC FI FITC C FITC 102 FIT 101 103 Relative fluorescence intensity 104

Parts of a flow cytometer • Fluidics – Provide a constant stream of sheath – Transport the sample to the interrogation point – Arrange and focus the cells to the laser intercept • Optics – Focus the excitation light – Collect the emitted light • Electronics – Convert the optical signals into electronic signals – Send the signals to the analysis computer • Computer – Display data graphically – Control instrument settings

What a flowcytometer is Very basically, a flow cytometer is an automated fluorescence microscope (in fact, that is how the first prototype instruments looked like). Like a microscope, some adjustments have to be made to optimally illuminate and collect the light.

The basic microscope In a standard microscope, the operator uses the XYstage to screen the sample and detect cells of interest.
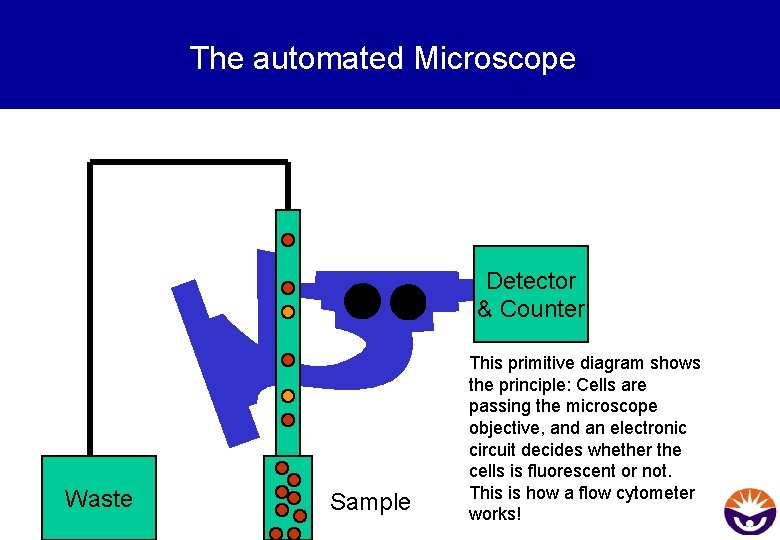
The automated Microscope Detector & Counter Waste Sample This primitive diagram shows the principle: Cells are passing the microscope objective, and an electronic circuit decides whether the cells is fluorescent or not. This is how a flow cytometer works!
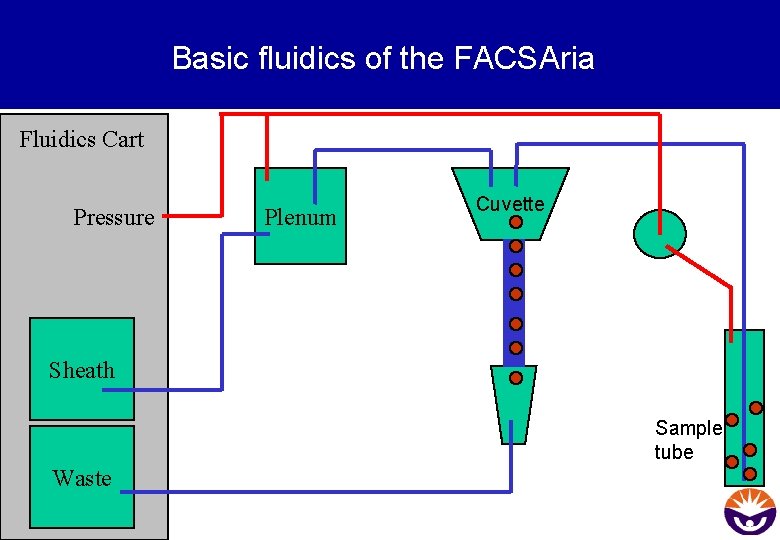
Basic fluidics of the FACSAria Fluidics Cart Pressure Plenum Cuvette Sheath Sample tube Waste
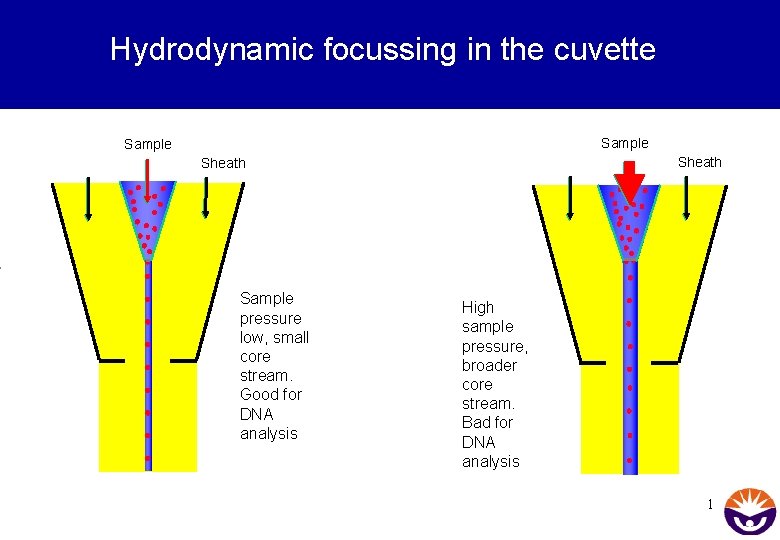
Hydrodynamic focussing in the cuvette Sample Sheath Sample pressure low, small core stream. Good for DNA analysis High sample pressure, broader core stream. Bad for DNA analysis 1

Summary • Pressure (= Sheath Pressure) drives the sheath buffer through the cuvette, and the higher pressure in the sample tube (= Sample Differential) delivers the sample to the cuvette. • In the cuvette the principle of hydrodynamic focussing arranges the cells like pearls on a string before they arrive at the laser interception point for analysis • Hydrodynamic focussing cannot separate cell aggregates! Flow cytrometry is a technique that requires single cell suspensions

Basic optics • Somehow the light from the laser(s) must be directed to the cuvette to illuminate the cells. • At the same time, the emitted light must be collected to analyse the signals from the cells. • The alignment of the system is performed during installation.

Basic optics A system of prisms and lenses directs the laser light to the interrogation point in the cuvette

Basic Optics The emitted light induced from each laser is focussed onto separate glass fibers.
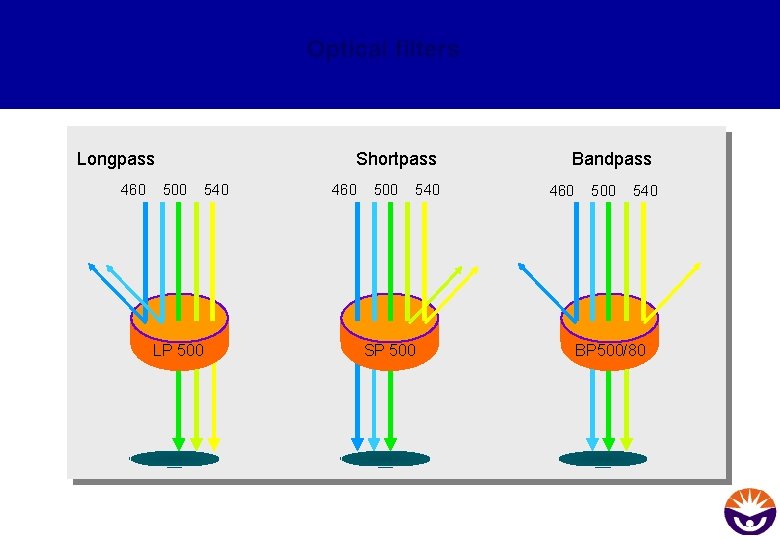
Optical filters Longpass 460 Shortpass 500 540 LP 500 460 500 540 SP 500 Bandpass 460 500 540 BP 500/80
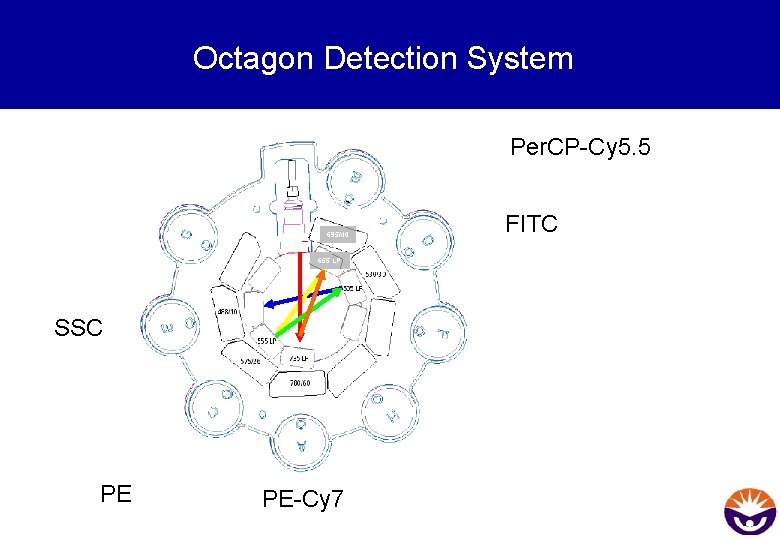
Octagon Detection System Per. CP-Cy 5. 5 695/40 655 LP SSC PE PE-Cy 7 FITC

Summary • Excitation light is steered with prisms and lenses to the interception point • Emitted light is collected using lenses and is split up with dichroic mirrors and filters

Tasks for the electronical system § Convert the optical signals into electonic signals (voltage pulses) § Digitise the data § Analyse Height (H), Width (W) and Area (A) of the pulse § Send the data to the analysis computer
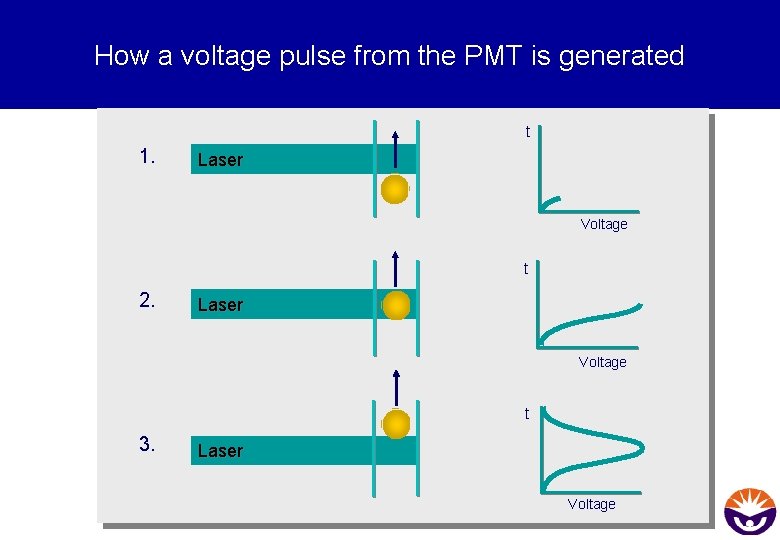
How a voltage pulse from the PMT is generated t 1. Laser Voltage t 2. Laser Voltage t 3. Laser Voltage
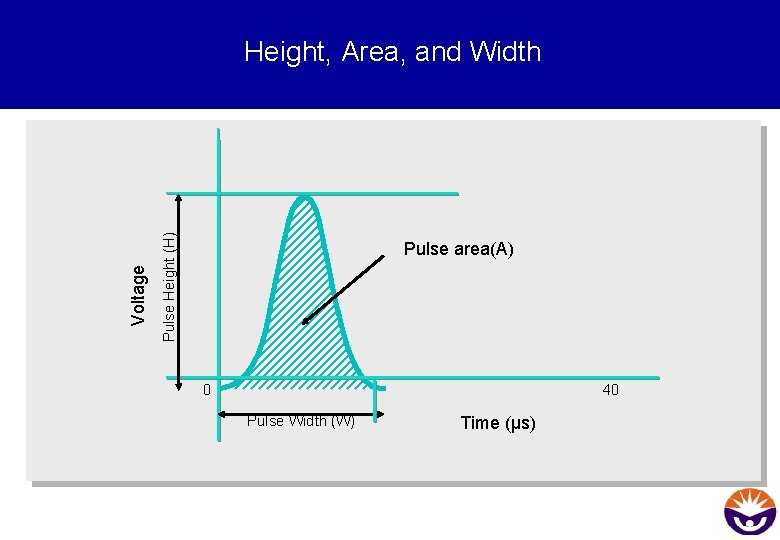
Pulse Height (H) Voltage Height, Area, and Width Pulse area(A) 0 40 Pulse Width (W) Time (µs)
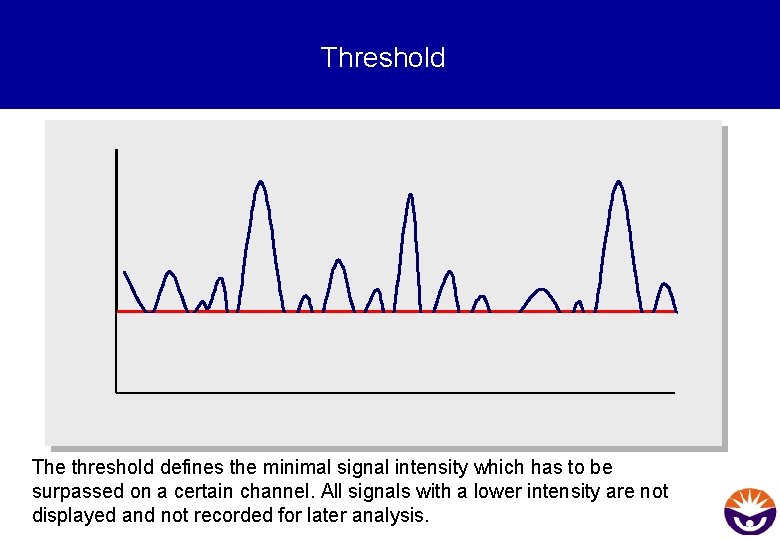
Threshold The threshold defines the minimal signal intensity which has to be surpassed on a certain channel. All signals with a lower intensity are not displayed and not recorded for later analysis.

Summary § During passing the laser voltage pulses are generated at the PMT § Amplifiers enhance the signals § The electronics digitizes the pulse using 10 MHz sampling § Only signals passing the desired threshold(s) are analysed and recorded § The data are finally passed to the analysis computer connected to the cytometer

Instrument settings n the exact values for PMT voltages and thresholds are depending on the applications (type of cells, staining methods) and the specific instrument. n Displaying the data in a linear fashion or using the logarithmic form is also depending on the application.

Workstation • The connected workstation is designed for instrument control, data acquisition, -storage and -analysis. • OS is Windows 2000 Professional running on a IBM-compatible computer platform. • Software • Di. Va application: Instrument connectivity, Data-acquisition and analysis system • Di. Va Data Manager: Backup and Restore the database.

Data saving All data are saved directly into a special database. Every plot is connected with its corresponding datafile. All tubes carry a copy of the instrument setting that was active during acquisition. Due to this, there are no special save commands in the software. Every action is recorded in the database. When you quit and re-start the software, it will open the last experiment exactly at the position you left it.
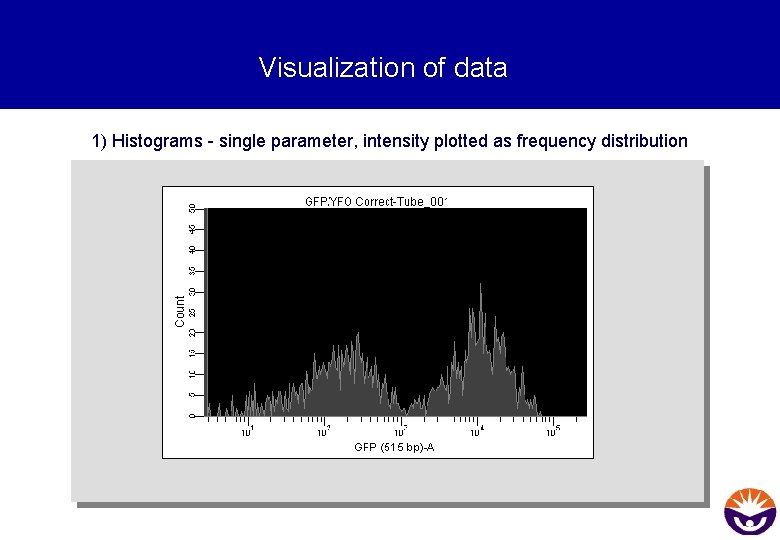
Visualization of data 1) Histograms - single parameter, intensity plotted as frequency distribution
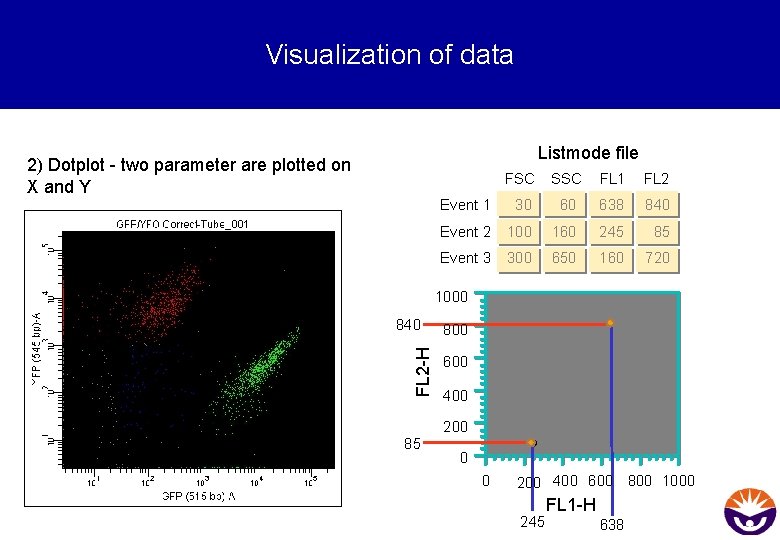
Visualization of data Listmode file 2) Dotplot - two parameter are plotted on X and Y FSC SSC FL 1 FL 2 Event 1 30 60 638 840 Event 2 100 160 245 85 Event 3 300 650 160 720 1000 FL 2 -H 840 800 600 400 200 85 0 0 200 400 600 800 1000 245 FL 1 -H 638

Enough theory of flow! Let`s have a look at an example from real life

Example: Determine the percentage of CD 3, CD 4, and CD 8 populations from whole blood Material • Mouse splenocytes Method • Three-colour immunofluorescence Preparation • Staining of freshly isolated splenocytes Stainings • Isotype controls • Single-colour stainings for CD 3 -FITC, CD 3 -PE, CD 3 -Per. CP und CD 3 APC to determine suitable instrument settings

Prepare the instrument

Proper adjustment of FSC and SSC voltage • FSC und SSC are optimally adjusted when the population of interest (i. e. Lymphocytes) can be resolved from all other populations • The threshold on FSC is adjusted so that most of the debris is excluded from the data acquisition.

Parameters (I) • FSC and SSC n are depending on cell type and cell state (activated, resting) n depend on the preparation method (Ficoll, LW, LNW, fixation method etc. ) n are normally used to define the population of interest for further analysis

Parameters • Fluorescence channels (FL 1, FL 2, FL 3, FLX) n depending on the specific staining (conjugate) antibodies, propidium iodide for DNA-labelling, etc. ) n most of the time fluorescence serves as marker for statistical analysis the
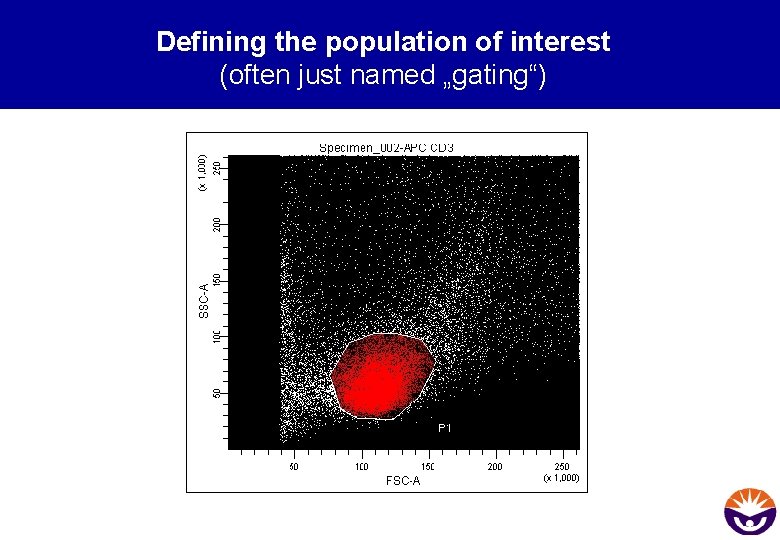
Defining the population of interest (often just named „gating“)

About „Gating“ • selectively analyse defined cell populations • Gates can be set manually or automatically by software • multidimensional gating with hierarchical gates • too narrow gates may lead to the loss of cell populations • too wide gates enhance the number of unwanted cells • during analysis of the desired cell population the cells in the gate are considered to be the 100%
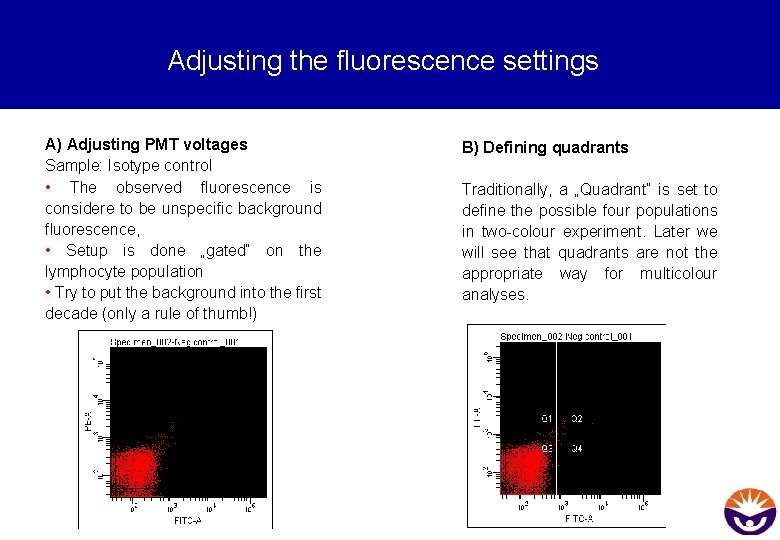
Adjusting the fluorescence settings A) Adjusting PMT voltages Sample: Isotype control • The observed fluorescence is considere to be unspecific background fluorescence, • Setup is done „gated“ on the lymphocyte population • Try to put the background into the first decade (only a rule of thumb!) B) Defining quadrants Traditionally, a „Quadrant“ is set to define the possible four populations in two-colour experiment. Later we will see that quadrants are not the appropriate way for multicolour analyses.
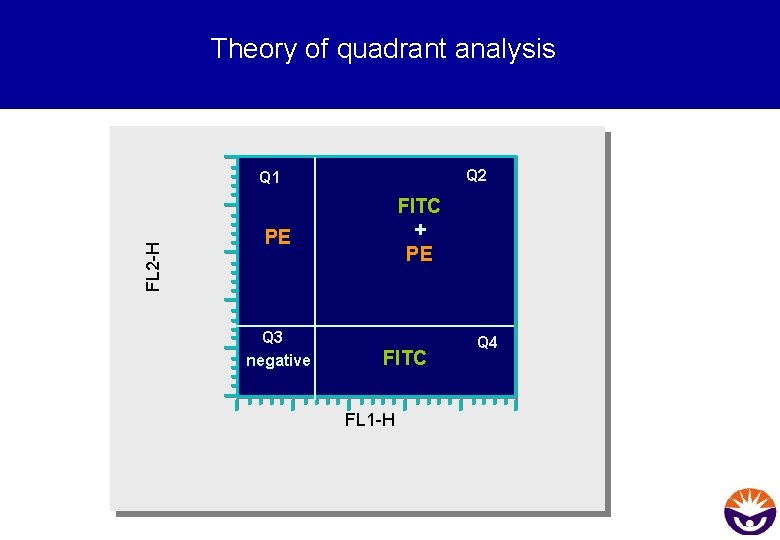
Theory of quadrant analysis Q 2 FL 2 -H Q 1 FITC + PE PE Q 3 negative FITC FL 1 -H Q 4
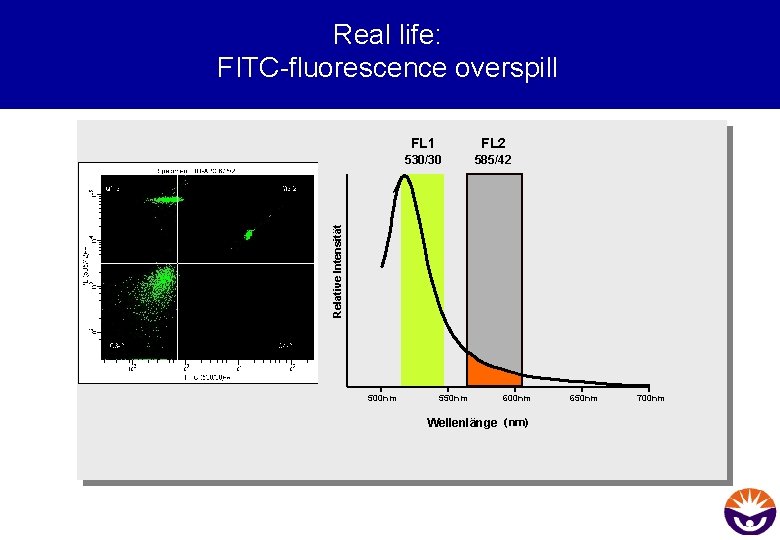
Real life: FITC-fluorescence overspill FL 2 530/30 585/42 Relative Intensität FL 1 500 nm 550 nm 600 nm Wellenlänge (nm) 650 nm 700 nm
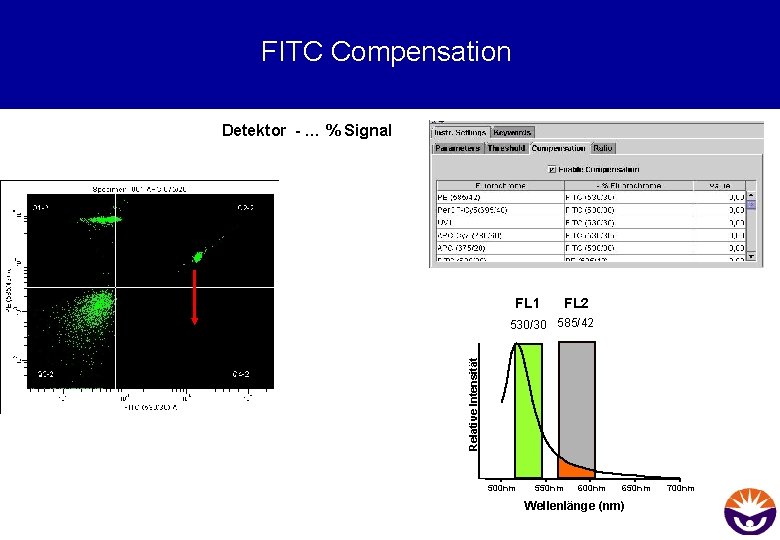
FITC Compensation Detektor - … % Signal FL 1 FL 2 Relative Intensität 530/30 585/42 500 nm 550 nm 600 nm 650 nm Wellenlänge (nm) 700 nm
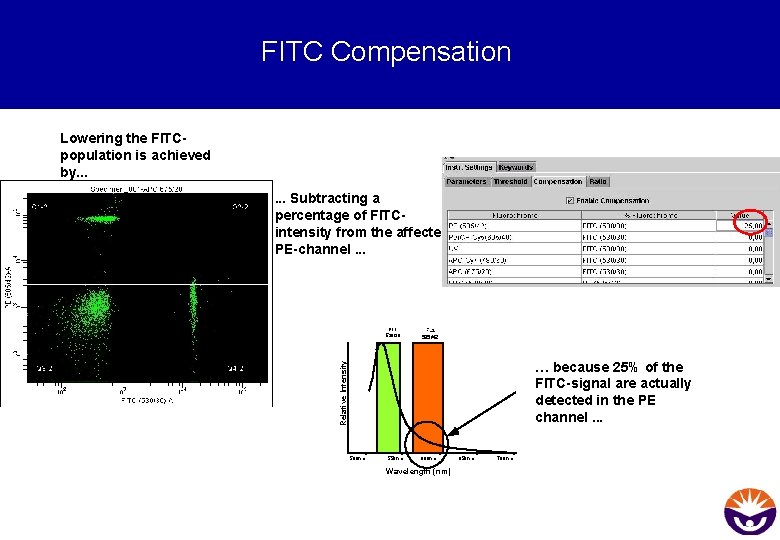
FITC Compensation Lowering the FITCpopulation is achieved by. . . Subtracting a percentage of FITCintensity from the affected PE-channel. . . FL 1 530/30 FL 2 585/42 Relative Intensity … because 25% of the FITC-signal are actually detected in the PE channel. . . 500 nm 550 nm 600 nm Wavelength (nm) 650 nm 700 nm
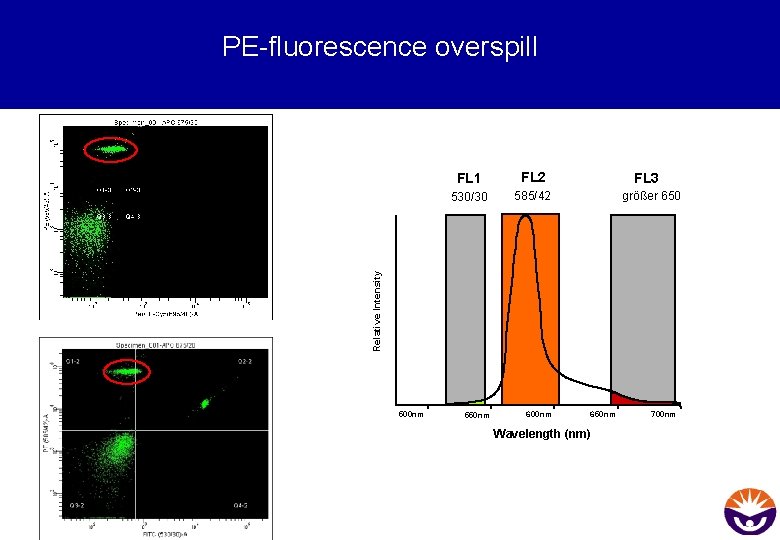
PE-fluorescence overspill FL 2 530/30 585/42 FL 3 größer 650 Relative Intensity FL 1 500 nm 550 nm 600 nm 650 nm Wavelength (nm) 700 nm

Automatic Multicolour Compensation • Multicolour compensation with more than three colours can become very time-consuming because each channel has to be compensated against each other. • Automatic compensation offers the possibility to run singlecolor controls and let the software calculate all overspills. • Mathematical calculation results in the correct spillover values for all channels. However, to the user the visual data may look undercompensated. This will be discussed in detail during the training course.

Summary What we have seen: • the emission spectra of common fluorochromes (FITC, PE) • the spectral overlap of fluorochromes into neighbouring channels depending on the emission spectra and filtersets • how spectral overlap can lead to misinterpretation of multicolour stainings • How compensation can correct the spectral overlap of fluorochromes
- Slides: 45