Thermodynamics X Unit 9 Energy Basic Principles Thermodynamics

Thermodynamics X Unit 9

Energy: Basic Principles Thermodynamics – the study of energy changes p Energy – the ability to do work or produce heat p n Kinetic Energy – energy of motion n Potential Energy – energy due to position or composition

Law of Conservation of Energy p A. k. a. First Law of Thermodynamics p Energy can be converted from one form to another but can’t be created or destroyed n This means the total energy of the universe is CONSTANT!

Heat vs. Temperature p Temperature – measure of the random motion of a substance n p Temperature is proportional to kinetic energy (it is a measure of the average kinetic energy in a substance) Heat (q) – flow of energy due to a temperature difference

System vs. Surroundings p A system is the part of the universe we are studying. The surroundings are everything else outside of the system. p

Direction of Heat Flow p Heat transfer occurs when two objects are at two different temperatures. p Eventually the two objects reach the same temperature n At this point, we say that the system has reached equilibrium.
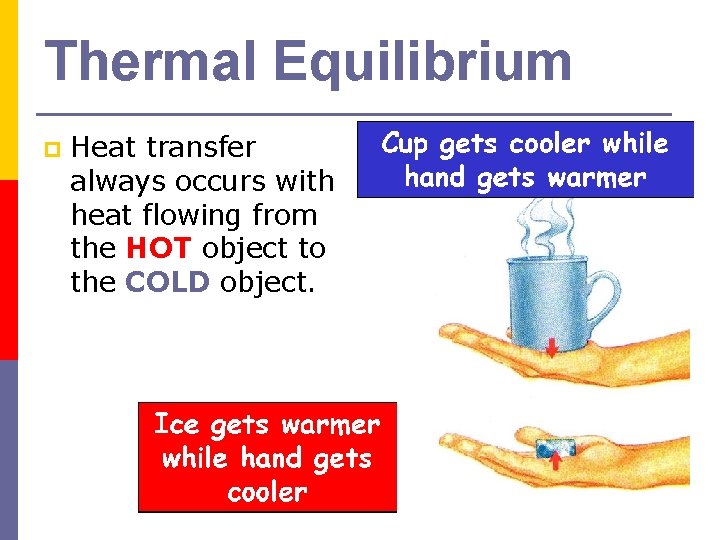
Thermal Equilibrium p Heat transfer always occurs with heat flowing from the HOT object to the COLD object.

Exothermic vs. Endothermic p Exothermic process heat is transferred from the system to the surroundings n Heat is lost from the system (temperature in system decreases) p Endothermic process heat transferred from the surroundings to the system n Heat is added to the system (temperature in system increases)

Units of Energy p Joule (J) is the SI unit of energy & heat One kilojoule (k. J) = 1000 joules (J) p calorie (cal) = heat required to raise the temperature of 1. 00 g of water by 1 °C 1 calorie = 4. 184 J

Units of Energy p Food is measured in Calories (also known as kilocalories) instead of calories 1 Cal = 1 kcal = 1000 calories

Units of Energy 3800 cal = _____ Cal = _____ J

Units of Energy The label on a cereal box indicates that 1 serving provides 250 Cal. What is the energy in k. J?

Heat Transfer Direction and sign of heat flow – MEMORIZE! ENDOTHERMIC: heat is added to the system & the temperature increases (+q) EXOTHERMIC: heat is lost from the system (added to the surroundings) & the temperature in the system decreases (-q)

Specific Heat (Specific Heat Capacity) p Specific Heat (C) - The quantity of heat required to raise the temperature of one gram of a substance by 1 °C p Units: n J/(g·°C) or J/(g·K) n cal/(g·°C) or cal/(g·K)

Examples of Specific Heat At the beach, which gets hotter, the sand or the water? Higher specific heat means the substance takes longer to heat up & cool down!

Examples of Specific Heat p Specific heat (C)= the heat required to raise the temperature of 1 gram of a substance by 1 °C Cwater = 4. 184 J/(g °C) Csand = 0. 664 J/(g °C)

Calculating Changes in Thermal E q = m. CΔT q = heat (J) m = mass (g) C = specific heat capacity, J/(g °C) ΔT = change in temperature, Tfinal – Tinitial (°C or K) ***All units must match up!!!***

Example q = m. CΔt How much heat in J is given off by a 75. 0 g sample of pure aluminum when it cools from 84. 0°C to 46. 7°C? The specific heat of aluminum is 0. 899 J/(g°C).

Example q = m. CΔt What is the specific heat of benzene if 3450 J of heat are added to a 150. 0 g sample of benzene and its temperature increases from 22. 5 °C to 35. 8 °C?

Enthalpy p Enthalpy (H) n The heat content of a reaction (chemical energy) p ΔH = change in enthalpy n The amount of energy absorbed or lost by a system during a chemical process p ΔH = ΔHfinal - ΔHinitial
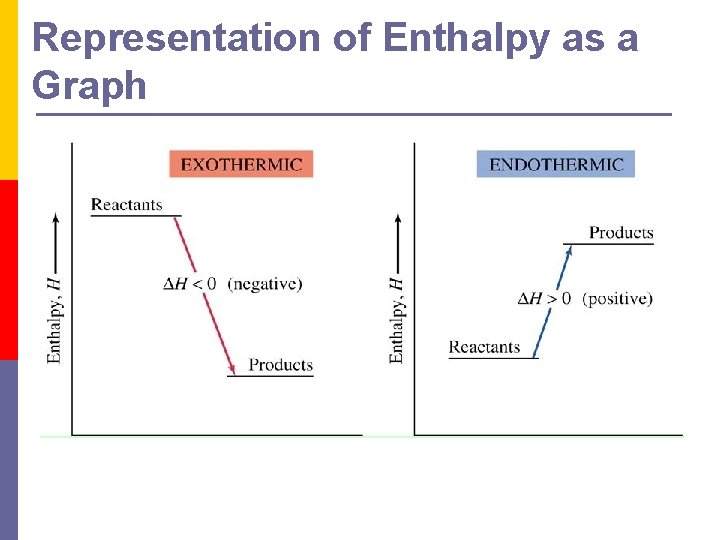
Representation of Enthalpy as a Graph

Two Ways to Designate Thermochemical Equations Endothermic: a) H 2 (g) + I 2 (s) 2 HI (g) b) H 2 (g) + I 2 (s) + 53. 0 k. J 2 HI (g) k. J ΔH = 53. 0

Two Ways to Designate Thermochemical Equations Exothermic: a) ½ CH 4 (g) + O 2 (g) ½ CO 2 (g) + H 2 O (l) ΔH = -445. 2 k. J b) ½ CH 4(g) + O 2(g) ½ CO 2(g) + H 2 O(l) + 445. 2 k. J

Two Ways to Designate Thermochemical Equations Note the meaning of the sign in ΔH in the equations above!! Endothermic: ΔH = + Exothermic: ΔH = -
- Slides: 24