Thermodynamics Thermodynamic Criterions Functions and ByeongJoo Lee Relations

Thermodynamics Thermodynamic Criterions, Functions and Byeong-Joo Lee Relations POSTECH - MSE calphad@postech. ac. kr Byeong-Joo Lee http: //cmse. postech. ac. kr

New Thermodynamic Functions – Reason for the necessity ※ Further development of Classical Thermodynamics results from the fact that S and V are an inconvenient pair of independent variables. + need to include composition variables in any equation of state and in any criterion of equilibrium + need to deal with non P-V work (e. g. , electric work performed by a galvanic cell) d. U = Td. S - Pd. V S, V 는 control이 쉽지 않은 변수. 따라서 control이 쉬운 변수를 택하고 그때 평형을 가늠할 수 있는 척도 (새로운 state function)를 개발할 필요. → F, G Byeong-Joo Lee http: //cmse. postech. ac. kr

Helmholtz Free Energy - Work Function, F ≡ U – ST d. F ≡ d. U – Td. S – Sd. T For a reversible process d. F = [Td. S – Pd. V – δw’] – Td. S – Sd. T = – Sd. T – Pd. V – δw’ d. FT = – Pd. V – δw’ = – δw. T. Total ▷ Constant T 조건에서 system이 상태를 변화해 가면서 할 수 있는 최대 work은 -ΔF. For an irreversible isothermal process △FT = [q – w] – T△S = q + T△Sirr w = P△V + w’ = w’ For constant V △FT, V + w’ + T△Sirr = 0 ▷ 최대의 일을 하지 못한다면 이는 ΔSirr의 발생 때문. Constant T, V 조건일 때 평형은 system의 maximum (w’+ ΔSirr) 또는 system의 minimum F에서 얻어짐. Byeong-Joo Lee http: //cmse. postech. ac. kr
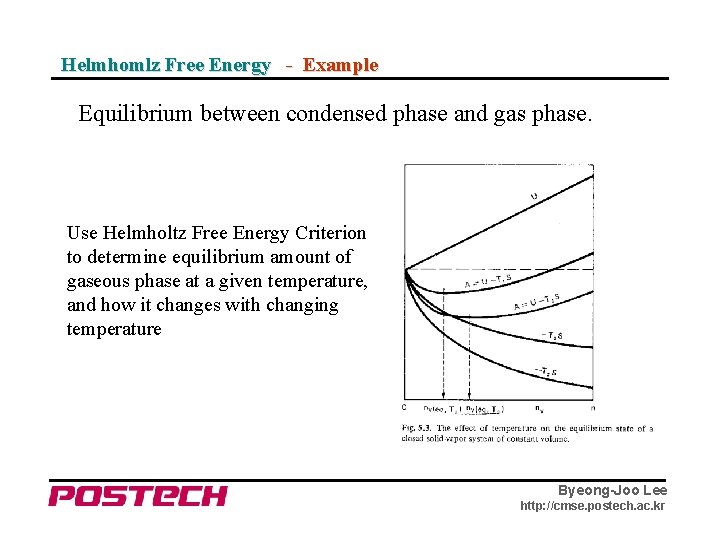
Helmhomlz Free Energy - Example Equilibrium between condensed phase and gas phase. Use Helmholtz Free Energy Criterion to determine equilibrium amount of gaseous phase at a given temperature, and how it changes with changing temperature Byeong-Joo Lee http: //cmse. postech. ac. kr

Gibbs Free Energy - Gibbs Function, G ≡ U + PV – ST d. G ≡ d. U + Pd. V + Vd. P – Td. S – Sd. T For a reversible process d. G = [Td. S – Pd. V – δw’] + Pd. V + Vd. P – Td. S – Sd. T = – Sd. T + Vd. P – δw’ d. GT, P = – δw’ ▷ Constant T, P 조건에서 system이 상태를 변화해 가면서 할 수 있는 최대 non-PV work은 -ΔG. For an irreversible isothermal process △GT, P = [q – w] + P△V – T△S = q + T△Sirr w = P△V + w’ △GT, P + w’ + T△Sirr = 0 ▷ 최대의 일을 하지 못한다면 이는 ΔSirr의 발생 때문. Constant T, P 조건일 때 평형은 system의 maximum (w’+ΔSirr) 또는 system의 minimum G에서 얻어짐. Byeong-Joo Lee http: //cmse. postech. ac. kr

Thermodynamic Relations - For a closed system d. U = Td. S – Pd. V d. H = Td. S + Vd. P d. F = – Sd. T – Pd. V d. G = – Sd. T + Vd. P Byeong-Joo Lee http: //cmse. postech. ac. kr

Thermodynamic Relations - For a multicomponent system ▷ ▷ Chemical Potential is the chemical work done by the system Byeong-Joo Lee http: //cmse. postech. ac. kr

Thermodynamic Relations Byeong-Joo Lee http: //cmse. postech. ac. kr

Thermodynamic Relations - Maxwell Relation Byeong-Joo Lee http: //cmse. postech. ac. kr

Thermodynamic Relations - Transformation Formula For an incremental change of state at constant x Byeong-Joo Lee http: //cmse. postech. ac. kr

Thermodynamic Relations - Application ▷ Heat capacity equation ▷ Gibbs-Helmholtz equation Byeong-Joo Lee http: //cmse. postech. ac. kr

Thermodynamic Relations 1. Gibbs Free Energy, Helmholtz Free Energy에서 “Free” 라는 단어의 의미는 무엇일까? 2. 수 많은 thermodynamic relation들이 존재한다. 그 relation들의 중요성은 무엇일까? 3. Gibbs-Helmholtz equation을 어떻게 활용할 수 있는지 용도를 조사하시오. Byeong-Joo Lee http: //cmse. postech. ac. kr
- Slides: 12