Thermodynamics Relation with Materials Science ByeongJoo Lee POSTECH

Thermodynamics Relation with Materials Science Byeong-Joo Lee POSTECH - MSE calphad@postech. ac. kr Byeong-Joo Lee http: //cmse. postech. ac. kr
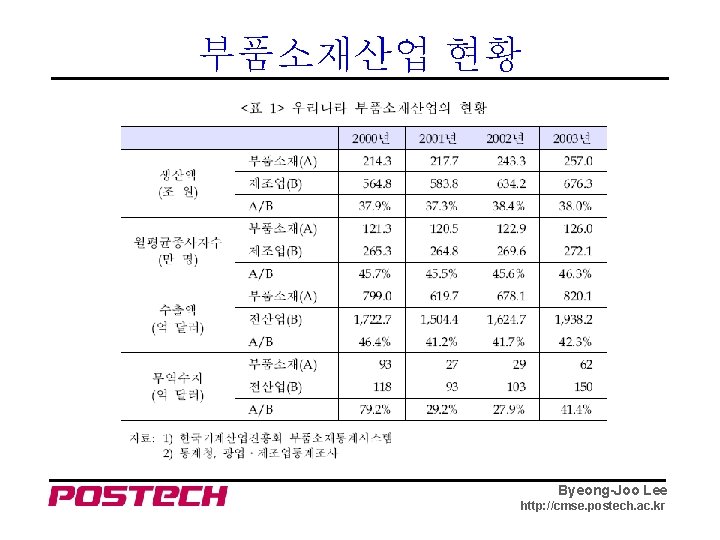
부품소재산업 현황 Byeong-Joo Lee http: //cmse. postech. ac. kr
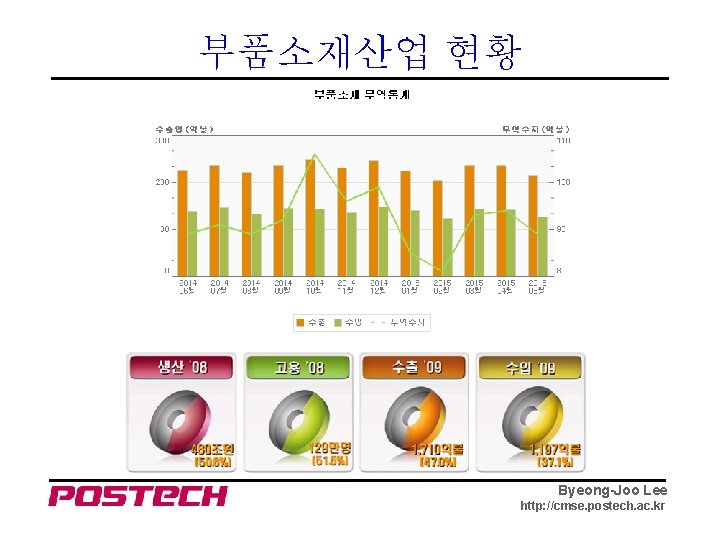
부품소재산업 현황 Byeong-Joo Lee http: //cmse. postech. ac. kr

부품소재산업 현황 Byeong-Joo Lee http: //cmse. postech. ac. kr
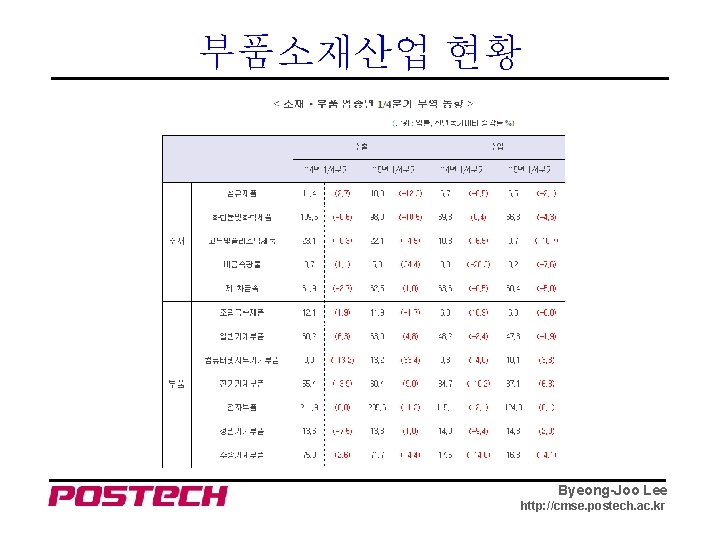
부품소재산업 현황 Byeong-Joo Lee http: //cmse. postech. ac. kr

대일 부품소재산업 현황 Byeong-Joo Lee http: //cmse. postech. ac. kr

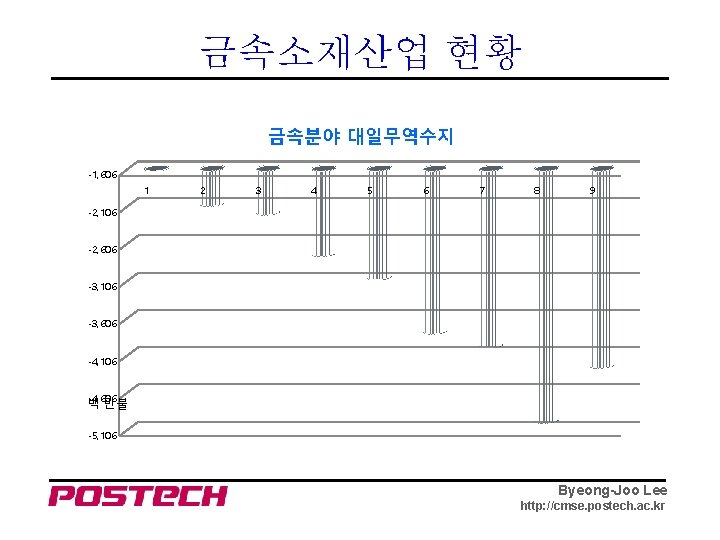
금속소재산업 현황 금속분야 대일무역수지 -1, 606 1 2 3 4 5 6 7 8 9 -2, 106 -2, 606 -3, 106 -3, 606 -4, 106 -4, 606 백 만불 -5, 106 Byeong-Joo Lee http: //cmse. postech. ac. kr

Byeong-Joo Lee http: //cmse. postech. ac. kr



Prologue (1) Materials Science and Engineering (2) 대학교 2학년 학생들이 해야 하는 일 (3) 소재 열역학 Byeong-Joo Lee http: //cmse. postech. ac. kr

Materials Science and Engineering What makes a Material Scientist a Genuine Materials Scientist 인문사회학자 vs. Medical Doctor Thinking & Simulation based on Scientific Knowledge Byeong-Joo Lee http: //cmse. postech. ac. kr

Materials Science and Engineering 연구를 어떻게 할 것인가? How to DO RESEARCH Byeong-Joo Lee http: //cmse. postech. ac. kr
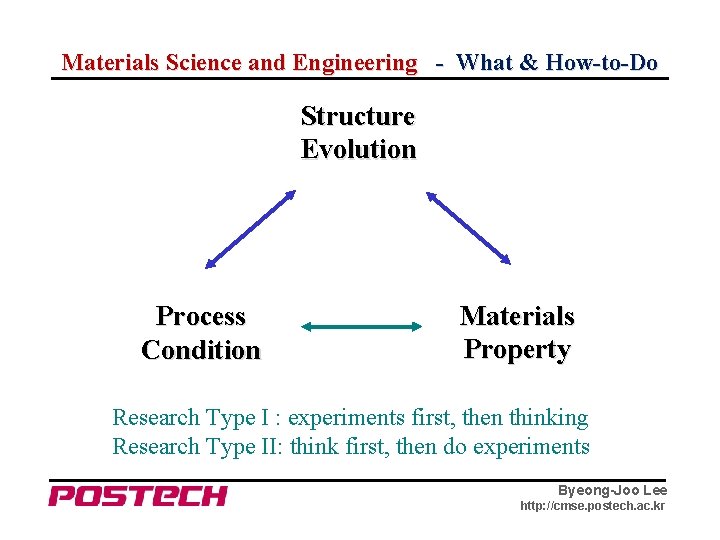
Materials Science and Engineering - What & How-to-Do Structure Evolution Process Condition Materials Property Research Type I : experiments first, then thinking Research Type II: think first, then do experiments Byeong-Joo Lee http: //cmse. postech. ac. kr

Materials Science and Engineering Thinking is important But, thinking without scientific background is nothing (空想) Byeong-Joo Lee http: //cmse. postech. ac. kr


General Fabrication of Monodisperse Nanoparticles Injection of Organometallic Precursor Surfactant Hot Surfactant Solution Aging Exhaustive Size Selection T. Hyeon, Chem. Comm. (Feature Article), 2003, 927 -934. Byeong-Joo Lee http: //cmse. postech. ac. kr
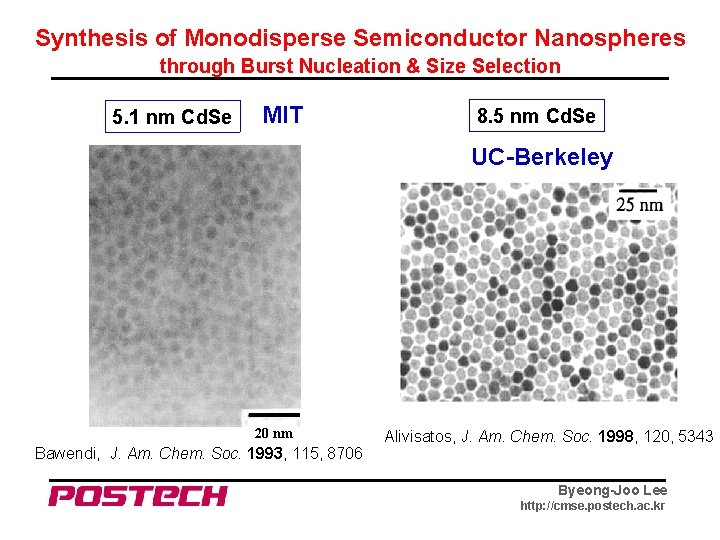
Synthesis of Monodisperse Semiconductor Nanospheres through Burst Nucleation & Size Selection 5. 1 nm Cd. Se MIT 8. 5 nm Cd. Se UC-Berkeley 20 nm Bawendi, J. Am. Chem. Soc. 1993, 115, 8706 Alivisatos, J. Am. Chem. Soc. 1998, 120, 5343 Byeong-Joo Lee http: //cmse. postech. ac. kr

Direct Synthesis of Monodisperse Iron Nanoparticles without a Size Selection Process!!! 20 nm 7 nm Fe Nanoparticles 20 nm 18 nm Fe Nanoparticles T. Hyeon, Chem. Comm. (Feature Article) 2003, 927 -934. 11 nm Fe Nanoparticles Byeong-Joo Lee http: //cmse. postech. ac. kr

Byeong-Joo Lee http: //cmse. postech. ac. kr

Monodisperse 3. 5 nm Pd nanoparticles 1 nm 20 nm Why Monosize Distribution of Nanoparticles? Byeong-Joo Lee http: //cmse. postech. ac. kr
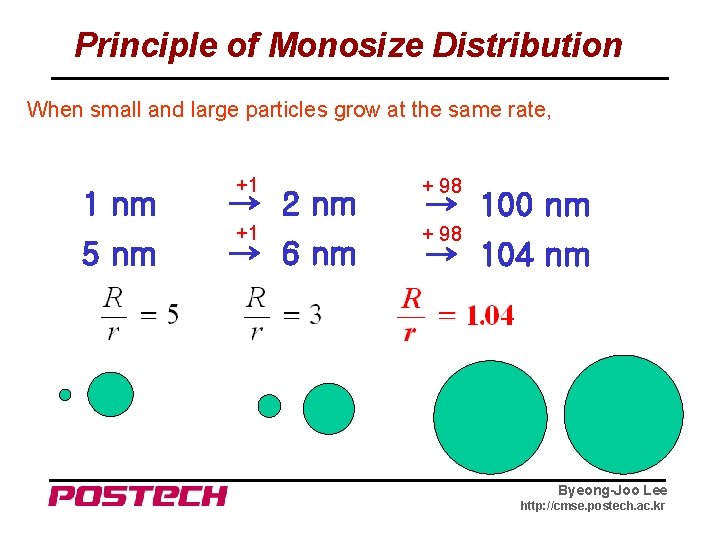
Principle of Monosize Distribution When small and large particles grow at the same rate, 1 nm 5 nm +1 + 98 → 2 nm → 6 nm → 100 nm → 104 nm Byeong-Joo Lee http: //cmse. postech. ac. kr
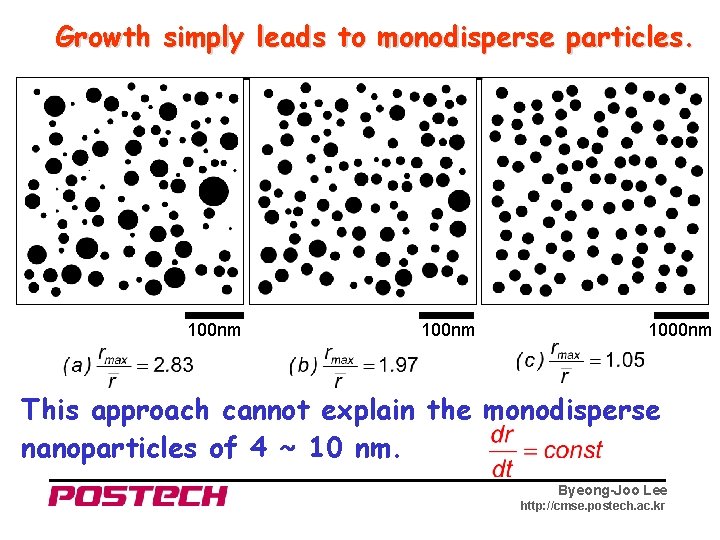
Growth simply leads to monodisperse particles. 100 nm 1000 nm This approach cannot explain the monodisperse nanoparticles of 4 ~ 10 nm. Byeong-Joo Lee http: //cmse. postech. ac. kr
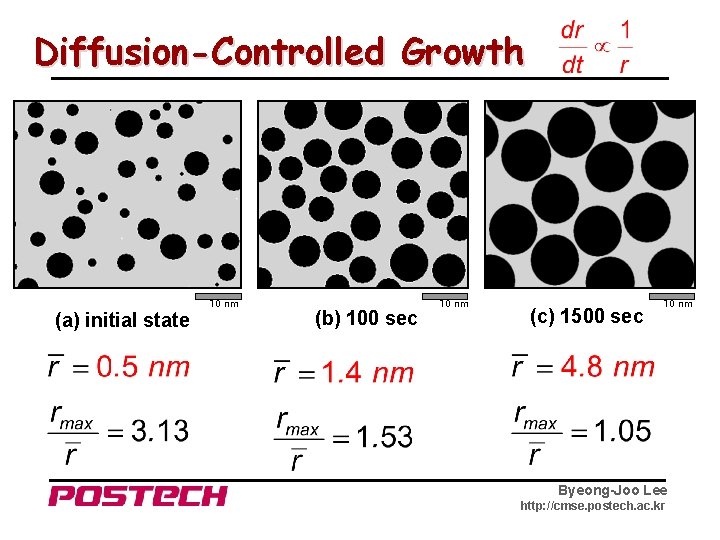
Diffusion-Controlled Growth 10 nm (a) initial state (b) 100 sec 10 nm (c) 1500 sec 10 nm Byeong-Joo Lee http: //cmse. postech. ac. kr

Necessary Conditions for Monosize Distribution 1. No Coagulation (Surfactant) 2. No Ostwald Ripening 3. No Nucleation during Growth Byeong-Joo Lee http: //cmse. postech. ac. kr

Synthesis of Monodisperse Nanoparticles without Size Section Requires that Growth Source should be different from Nucleation source. Identification of Nucleation and Growth Sources Nucleation Source Fe(CO)5 Growth Source Fe-oleic complex Byeong-Joo Lee http: //cmse. postech. ac. kr

6 nm 10 nm 7 nm 8 nm 11 nm 12 nm 9 nm 13 nm 1 nm size control of monodisperse nanoparticles Byeong-Joo Lee http: //cmse. postech. ac. kr

Computational Materials & Process Design A Success Story Byeong-Joo Lee POSTECH - MSE calphad@postech. ac. kr Byeong-Joo Lee http: //cmse. postech. ac. kr
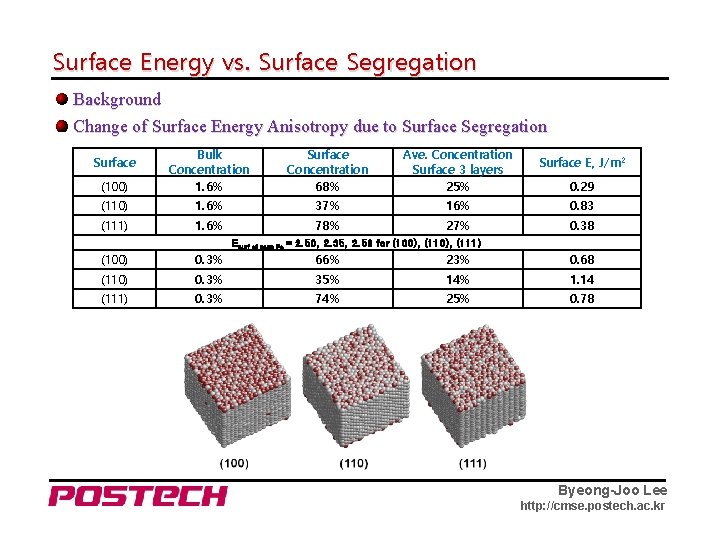
Surface Energy vs. Surface Segregation Background Change of Surface Energy Anisotropy due to Surface Segregation (100) Bulk Concentration 1. 6% Surface Concentration 68% Ave. Concentration Surface 3 layers 25% (110) 1. 6% 37% 16% 0. 83 (111) 1. 6% 78% 27% 0. 38 Surface E, J/m 2 0. 29 Esurf of pure Fe = 2. 50, 2. 35, 2. 56 for (100), (111) (100) 0. 3% 66% 23% 0. 68 (110) 0. 3% 35% 14% 1. 14 (111) 0. 3% 74% 25% 0. 78 Byeong-Joo Lee http: //cmse. postech. ac. kr
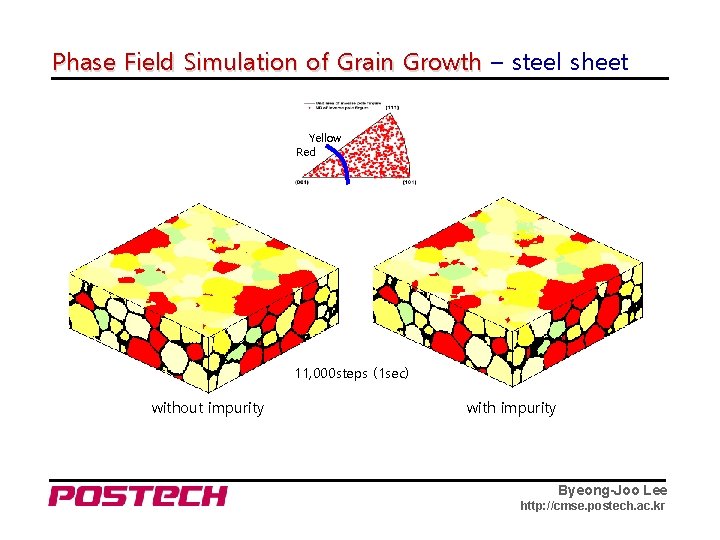
Phase Field Simulation of Grain Growth – steel sheet Yellow Red 11, 000 steps (1 sec) without impurity with impurity Byeong-Joo Lee http: //cmse. postech. ac. kr

Phase Field Simulation of Grain Growth – modified assuming that impurity atoms were segregated before the grain growth Initial sample Yellow Red 8, 000 steps (0. 75 sec) Byeong-Joo Lee http: //cmse. postech. ac. kr

Experimental Verification – (100) texture on Steel Sheet Byeong-Joo Lee http: //cmse. postech. ac. kr
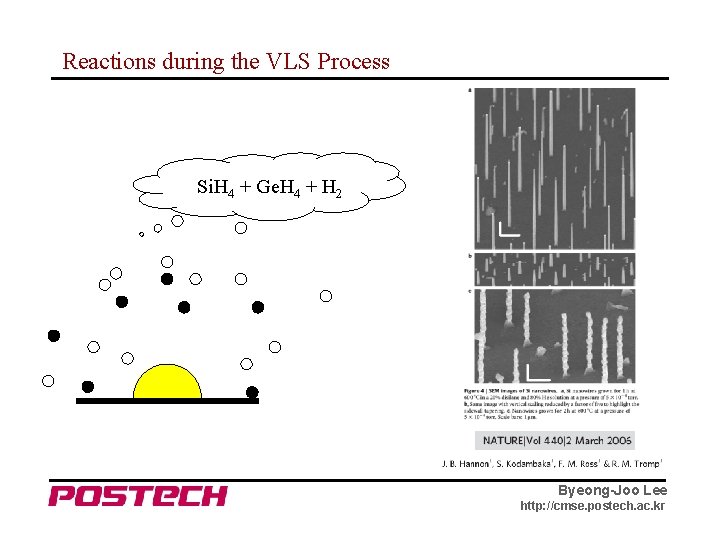
Reactions during the VLS Process Si. H 4 + Ge. H 4 + H 2 Byeong-Joo Lee http: //cmse. postech. ac. kr
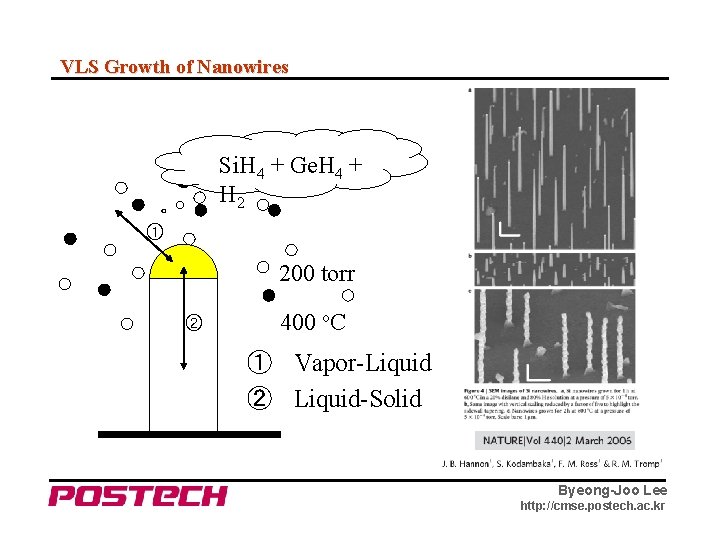
VLS Growth of Nanowires Si. H 4 + Ge. H 4 + H 2 ① 200 torr ② 400 o. C ① Vapor-Liquid ② Liquid-Solid Byeong-Joo Lee http: //cmse. postech. ac. kr
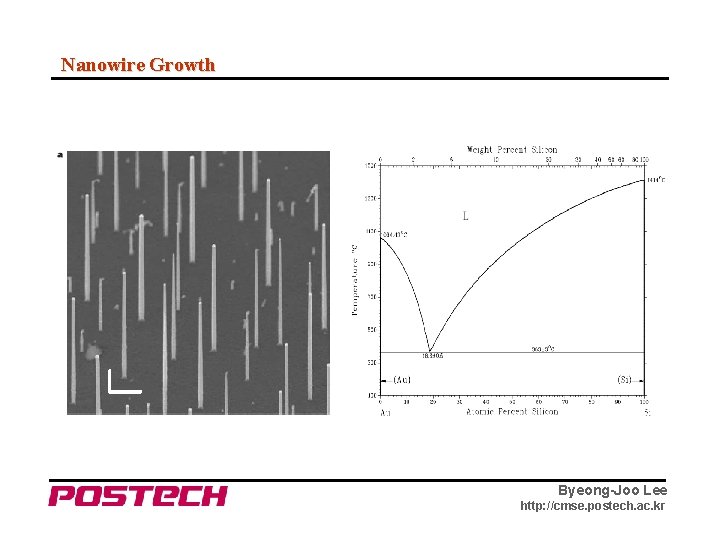
Nanowire Growth Byeong-Joo Lee http: //cmse. postech. ac. kr
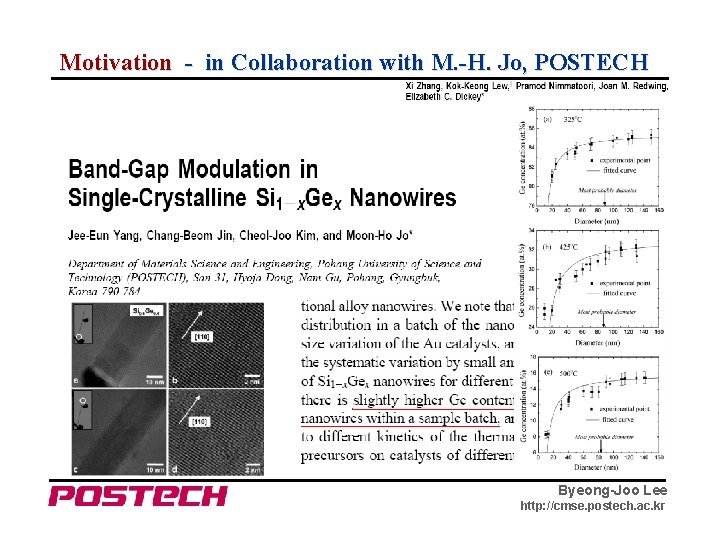
Motivation - in Collaboration with M. -H. Jo, POSTECH Byeong-Joo Lee http: //cmse. postech. ac. kr
Melting point depression of Nano particles - Capillarity : Thermodynamics Byeong-Joo Lee http: //cmse. postech. ac. kr
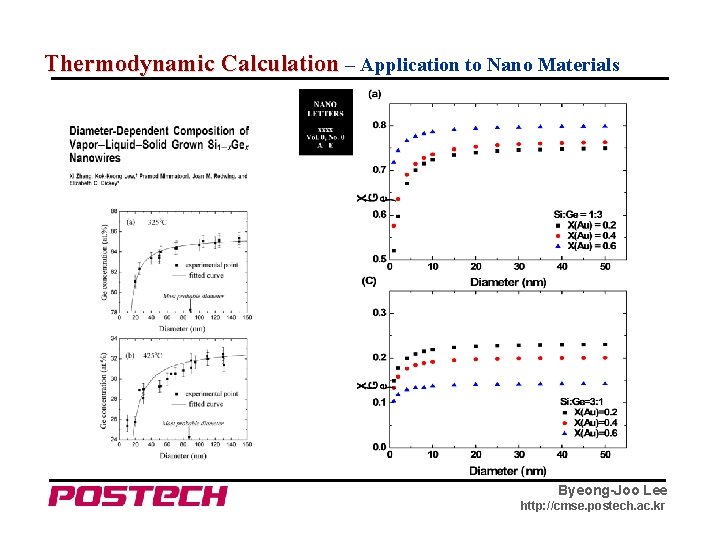
Thermodynamic Calculation – Application to Nano Materials Byeong-Joo Lee http: //cmse. postech. ac. kr

Surface Transition and Alloying Effect Pure W – N. M. Hwang et al. , 2000. W + 0. 4 wt% Ni Vaccum Annealing Byeong-Joo Lee http: //cmse. postech. ac. kr
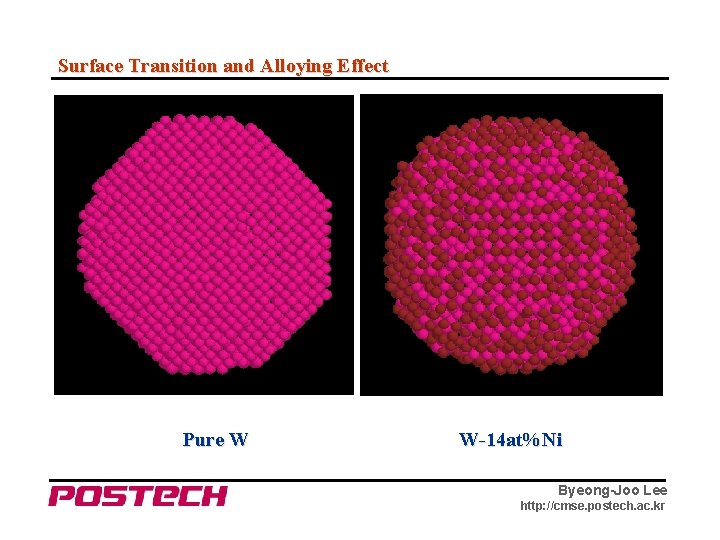
Surface Transition and Alloying Effect Pure W W-14 at%Ni Byeong-Joo Lee http: //cmse. postech. ac. kr

Abnormal Grain Growth – A 60 years old unsolved problem Byeong-Joo Lee http: //cmse. postech. ac. kr

CVD Diamond - N. M. Hwang et al. , J. Crystal Growth 162, 55 (1996) Diamond deposited on silicon substrate Soot deposited on the iron substrate Byeong-Joo Lee http: //cmse. postech. ac. kr
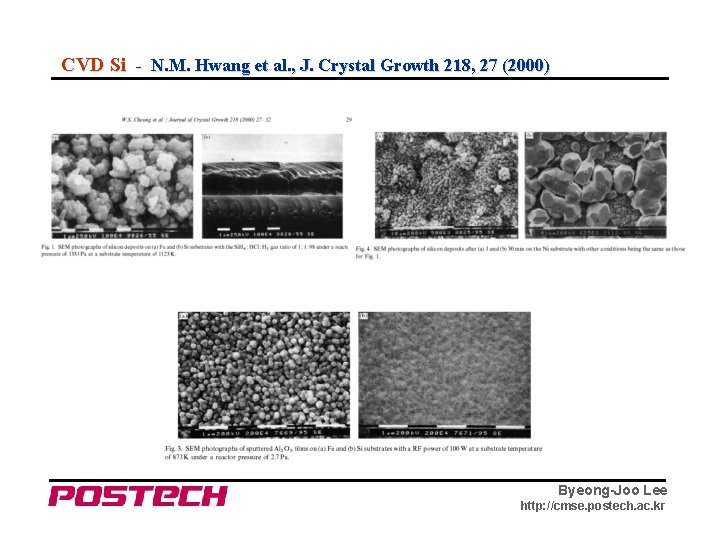
CVD Si - N. M. Hwang et al. , J. Crystal Growth 218, 27 (2000) Byeong-Joo Lee http: //cmse. postech. ac. kr

연구를 어떻게 할 것인가? How to DO RESEARCH Byeong-Joo Lee http: //cmse. postech. ac. kr

Thinking is important But, thinking without scientific background is nothing (空想) Byeong-Joo Lee http: //cmse. postech. ac. kr
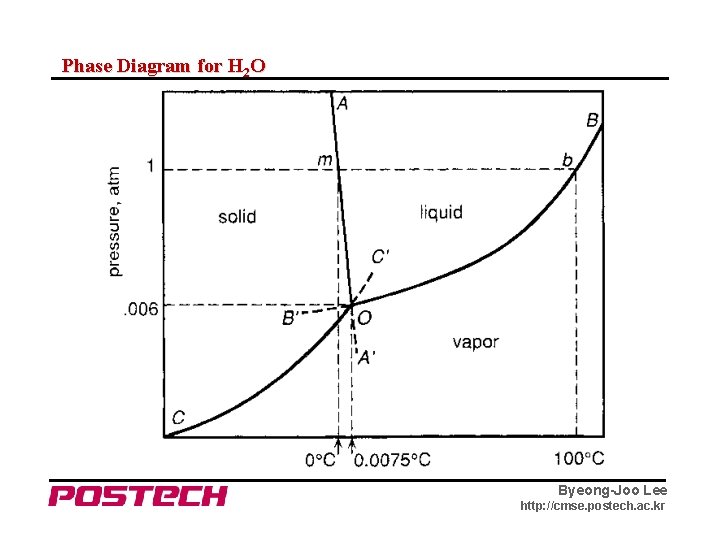
Phase Diagram for H 2 O Byeong-Joo Lee http: //cmse. postech. ac. kr

Course Outline (1) 열역학 기본법칙의 이해 및 응용 (2) 단일 및 복합계 상평형 및 상태도의 이해 (3) 주요 반응, 결함, 상변화 현상 열역학적 해석 능력 배양 시험 : 70% 숙제 : 20% 출석 : 10% 1. Introduction to the Thermodynamics of Materials, David R. Gaskell, Taylor & Francis, 3 rd Edition, 1995. 2. Thermodynamics in Materials Science, Robert T. De. Hoff, Mc. Graw-Hill International Editions, 1993. Byeong-Joo Lee http: //cmse. postech. ac. kr

Thermodynamics Introduction Byeong-Joo Lee POSTECH - MSE calphad@postech. ac. kr Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction – Historical Background Maxwell · Newton → Classical Mechanics → Electromagnetism → Quantum Mechanics · Lagrangian · Rationalism · Hamiltonian · Time Reversible · Experimental fact (산업혁명) · Heat → Concept of Temperature · PV work → Concept of Pressure → Thermodynamics · Empirical Rule · Time irreversible · Thompson → Rumford · Davy · Mayor · Carnot · Joule · Thomson → Kelvin · Clausius · Maxwell · Gibbs Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction - J. W. Gibbs ※ Expansion in scope has been made to embrace not only thermal, mechanical and chemical effects, but also the kinetic energy and the complete set of potentials energies: gravitational, electrical, magnetic and body forces, (with the exception of atomic energy). ※ J. Willard Gibbs, 1883 : On the Equilibrium of Heterogeneous Substances Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction - Microscopic vs. Macroscopic Point of View · macroscopic : no special assumption on the structure of matter, directly measured · microscopic : assumption on the structure of matter (molecules), unmeasurable. can only be justified by comparing some deduction with that from macroscopic point of view. · quantum mechanics Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction - Role of Thermodynamics · Foundation of materials science · Phase Diagrams, Chemical Reactions, Adsorption, Capillarity effects and Electrochemistry, …, etc. Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction - Basic Terminology · System / Surrounding (reservoir) · State (Properties: description of state), Change of state (properties) during a process · Measurable and unmeasurable properties : Unmeasurable property는 다른 측정 가능한 특성치로부터 결정 ⇔ Property 간의 관계식 (EOS) ※ Thermodynamic systems, thermodynamic properties, thermodynamic relationships Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction - Terminology for System · Unary / multicomponent · Homogeneous / Heterogeneous · Closed / Open · Non-reacting / reacting · Exchange of energies / No exchange (※ Isolated) Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction - State Function & Process Variable · Thermodynamics State or State of Thermodynamic Equilibrium Mechanical, Chemical and Thermal Equilibrium Uniform and well defined thermodynamic coordinates No tendency to change with time of thermodynamic coordinates · path independence → · Extensive and Intensive Properties · Process variable (only have meaning for changing systems) · work & heat Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction - Equation of State of an ideal gas · Boyle’s law (1660): · Charles’ law (1787): = constant = 1 atm× 22. 414 liters ∕ 273. 16 degree-mole = 0. 082057 liter-atm/degree-mole ∙ EOS of the ideal gas: PV = RT ▶ unit of Work 1 atm = 76 cm of mercury = 76. 0× 13. 595× 980. 665 = 1. 013× 10 6 dyne/cm 2 1 atm-liter = 1. 013 × 102 joule Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction - Ideal Gas Temperature Scale Byeong-Joo Lee http: //cmse. postech. ac. kr
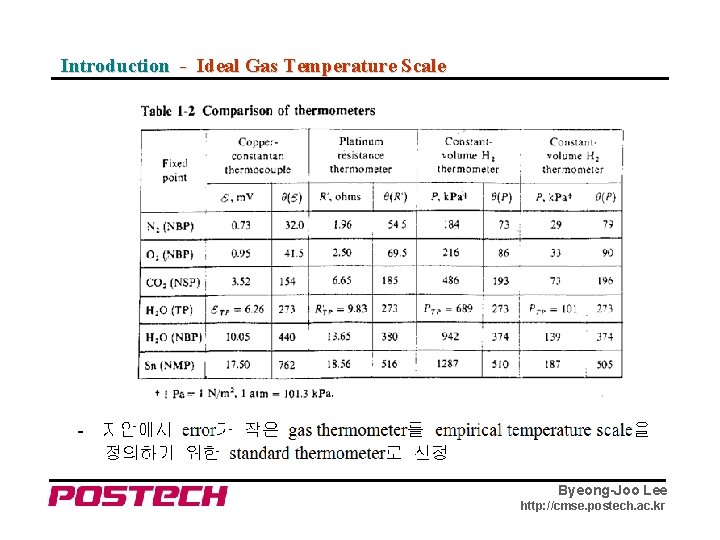
Introduction - Ideal Gas Temperature Scale Byeong-Joo Lee http: //cmse. postech. ac. kr
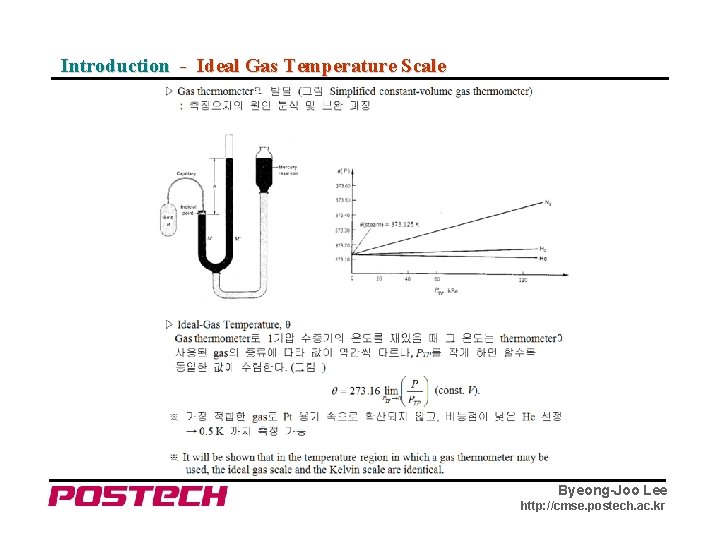
Introduction - Ideal Gas Temperature Scale Byeong-Joo Lee http: //cmse. postech. ac. kr

Introduction - Ideal Gas Temperature Scale Byeong-Joo Lee http: //cmse. postech. ac. kr

- Slides: 63