Thermodynamics Phase Equilibria in a Single Component ByeongJoo

Thermodynamics Phase Equilibria in a Single. Component Byeong-Joo Lee System POSTECH - MSE calphad@postech. ac. kr Byeong-Joo Lee http: //cmse. postech. ac. kr
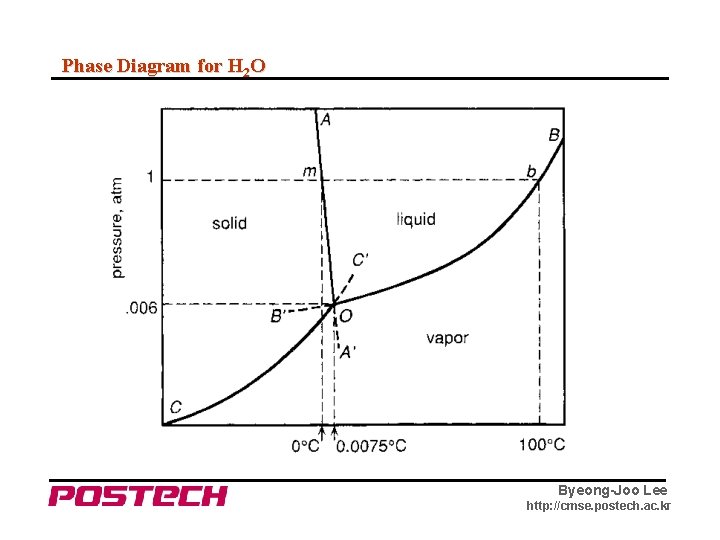
Phase Diagram for H 2 O Byeong-Joo Lee http: //cmse. postech. ac. kr
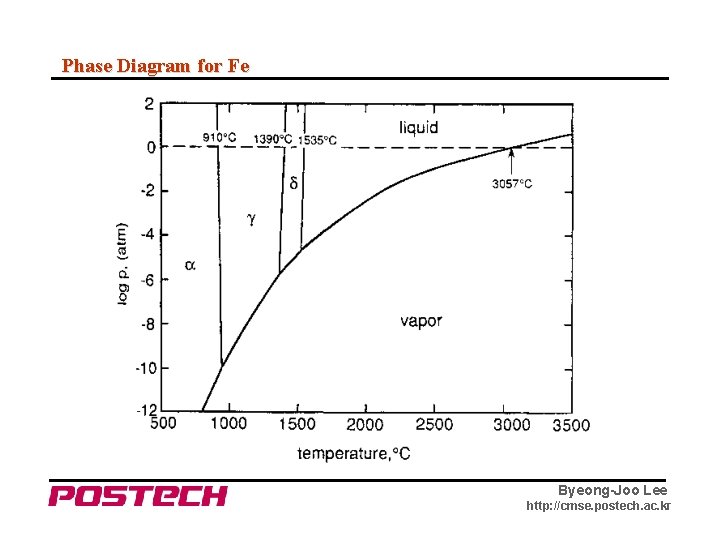
Phase Diagram for Fe Byeong-Joo Lee http: //cmse. postech. ac. kr
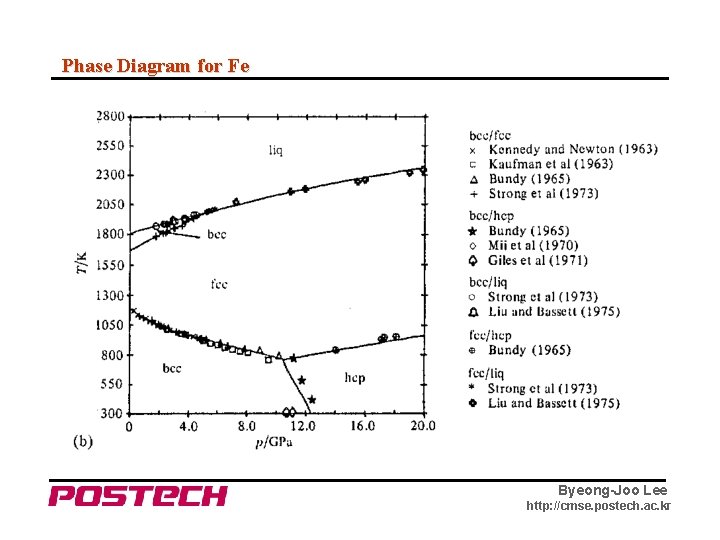
Phase Diagram for Fe Byeong-Joo Lee http: //cmse. postech. ac. kr

Equilibrium • Thermal, Mechanical and Chemical Equilibrium • Concept of Chemical Potential In a one component system, • Temperature and Pressure dependence of Gibbs free energy Byeong-Joo Lee http: //cmse. postech. ac. kr
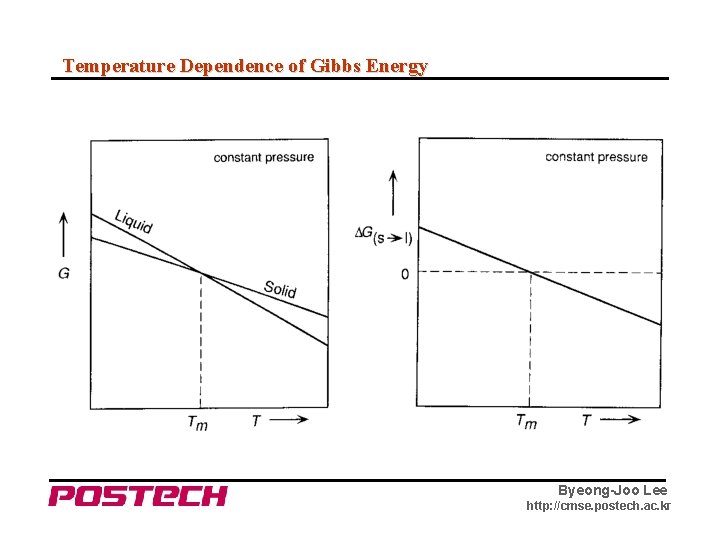
Temperature Dependence of Gibbs Energy Byeong-Joo Lee http: //cmse. postech. ac. kr
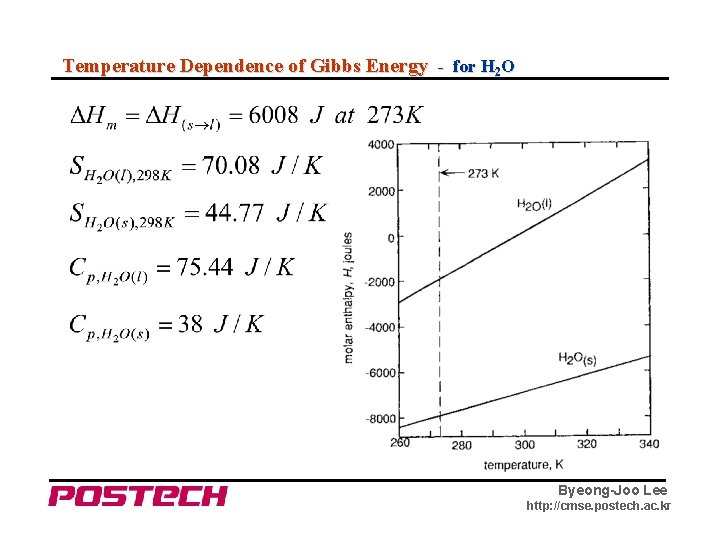
Temperature Dependence of Gibbs Energy - for H 2 O Byeong-Joo Lee http: //cmse. postech. ac. kr

Temperature & Pressure Dependence of Gibbs Energy Clausius-Clapeyron equation For equilibrium between the vapor phase and a condensed phase constant Byeong-Joo Lee http: //cmse. postech. ac. kr
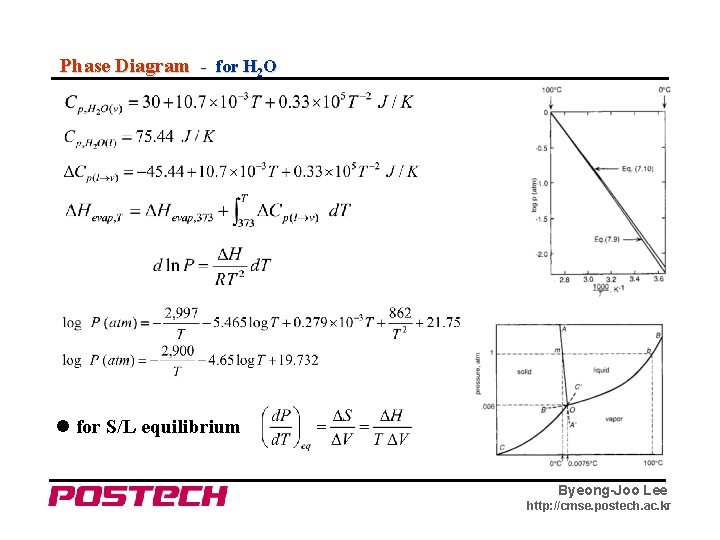
Phase Diagram - for H 2 O for S/L equilibrium Byeong-Joo Lee http: //cmse. postech. ac. kr
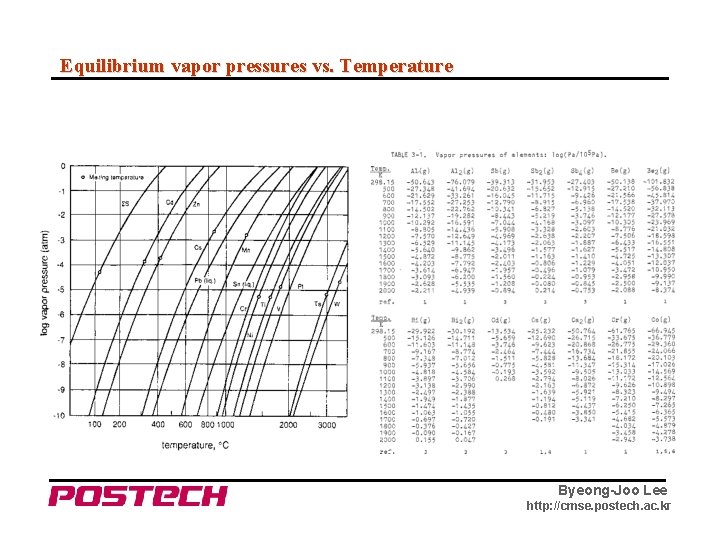
Equilibrium vapor pressures vs. Temperature Byeong-Joo Lee http: //cmse. postech. ac. kr
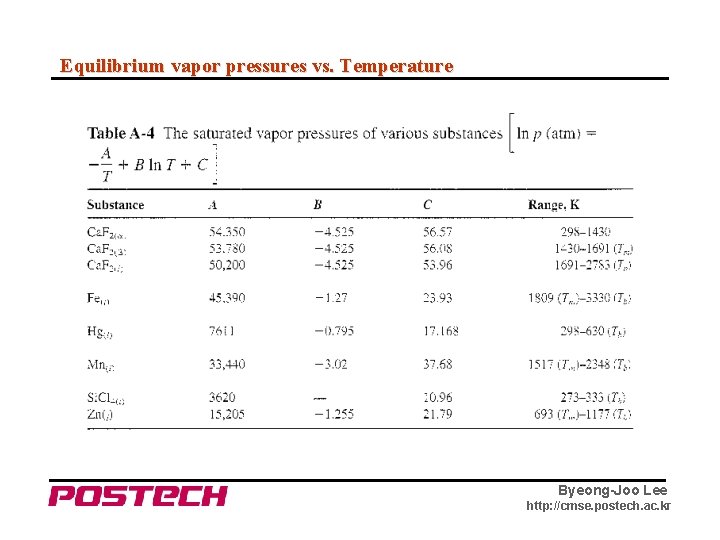
Equilibrium vapor pressures vs. Temperature Byeong-Joo Lee http: //cmse. postech. ac. kr

Gibbs Phase Rule • Degree of Freedom number of variables which can be independently varied without upsetting the equilibrium • F = p(1+c) – (p-1)(2+c) = c – p + 2 Byeong-Joo Lee http: //cmse. postech. ac. kr

Example - Phase Transformation of Graphite to Diamond • Calculate graphite→diamond transformation pressure at 298 K, given H 298, gra – H 298, dia = -1900 J S 298, gra = 5. 74 J/K S 298, dia = 2. 37 J/K density of graphite at 298 K = 2. 22 g/cm 3 density of diamond at 298 K = 3. 515 g/cm 3 Byeong-Joo Lee http: //cmse. postech. ac. kr
- Slides: 13