Thermodynamics of a Single Particle System W J












- Slides: 15

Thermodynamics of a Single Particle System W. J. Wilson Department of Engineering and Physics University of Central Oklahoma Edmond, OK 73034 2011 Oklahoma Academy of Sciences Annual. Technical Meeting Southeastern Oklahoma State University Durant, OK November 11, 2011 wwilson@uco. edu

Thermodynamics of a Particle? Single particle thermodynamics is a bit of an uncharted subject. Some, steeped in statistical basis of thermodynamics, conclude that the laws of thermodynamics fall apart at the single particle level. “The law of entropy increasing is only a statistical law; it is not ‘fundamental’ because it cannot describe the behavior of an individual atom or molecule; it deals with the average number of them. Entropy is not a concept that can be meaningfully applied to a single particle, or even to a small number of particles. ” – John Wheeler wwilson@uco. edu

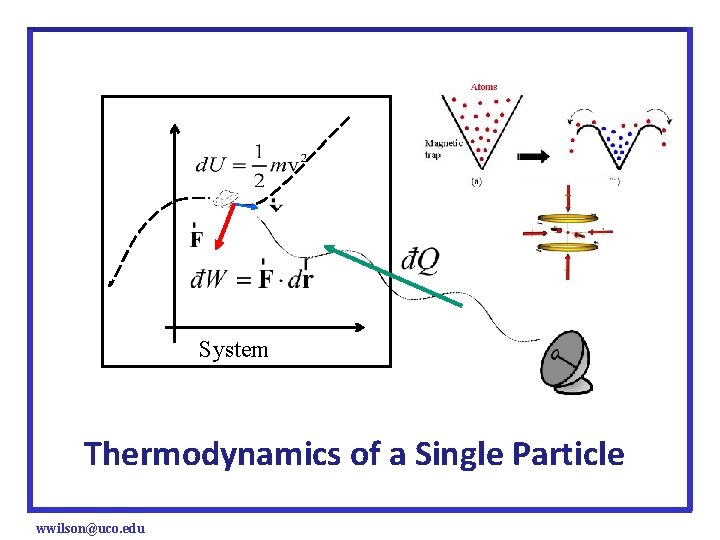
System Thermodynamics of a Single Particle wwilson@uco. edu

First Law – Sign Convention The positive work done by system sign convention (used in physics and engineering ) is wwilson@uco. edu

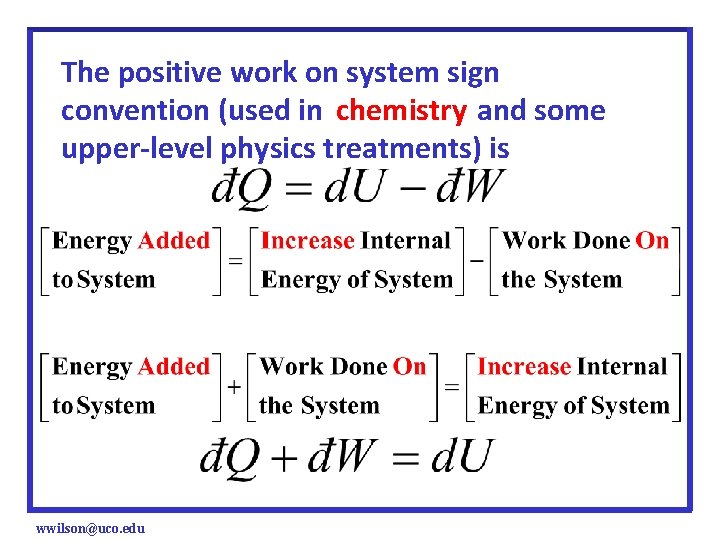
The positive work on system sign convention (used in chemistry and some upper-level physics treatments) is wwilson@uco. edu

For a single particle, U = KE+PE = U(x, v) and F = F(x, v) are functions of position and velocity only so if we use the work on system positive sign convention which becomes for a free(non-relativistic) particle, wwilson@uco. edu

Now if the change dx and dv occur in time dt So for a single particle system, wwilson@uco. edu

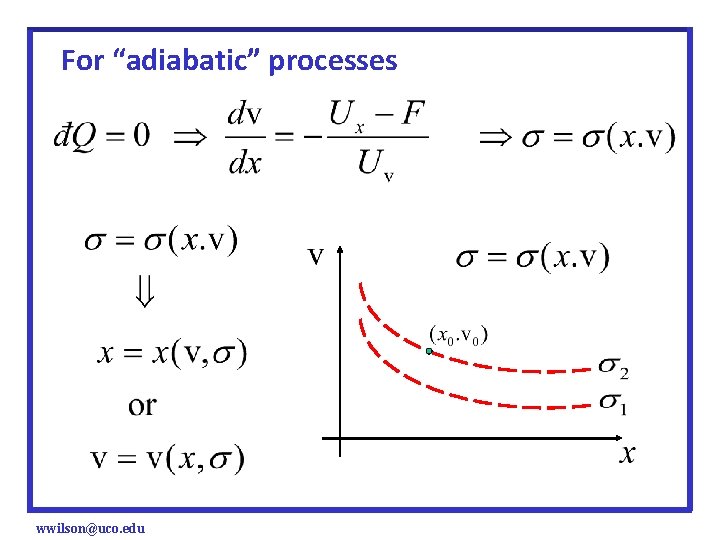
So for a single particle system, And for a closed system (no energy allowed to enter or escape) We find wwilson@uco. edu (i. e. , an “Adiabatic” Process)

So for a single particle system, using the work done on the system is positive sign convention, 1. Yields Newton’s 2 nd Law in the standard form 2. Particle motions governed by Newton’s 2 nd Law correspond to “adiabatic” processes wwilson@uco. edu

Integrating Factor For a single particle, U = KE+PE = U(x, v) and F = F(x, v) are functions of position and velocity only so wwilson@uco. edu

For “adiabatic” processes or The right-hand side is a function of x and v only, so we are guaranteed a unique solution through a given initial state (x 0, v 0) wwilson@uco. edu

For “adiabatic” processes wwilson@uco. edu

For “adiabatic” processes and So we must have a factor λ(x, v) such that or wwilson@uco. edu

Now since These yield an exact differential As So wwilson@uco. edu

Summary / Conclusions • The natural sign convention for physics • There is an exact differential (“entropy”) for the single particle References E. C. G. Stueckelberg and P. B. Scheurer, Thermocinéque Phénoménologique Galiléenne, (Birkhauser, Stuttgart, 1974) P. E. Williams, On a Possible Formulation of Particle Dynamics in Terms of Thermodynamics Conceptualizations and the Role of Entropy in It (M. S. Thesis, Naval Postgraduate School, Monterey, CA, 1976). wwilson@uco. edu 15