Thermodynamics n Understanding the words Temperature Heat capacity






































- Slides: 47

Thermodynamics n Understanding the words Temperature ® Heat capacity ® The 0, 1, 2 laws of thermodynamics ® n (one of) Kelvin’s legacy’s William. Thompson (Lord Kelvin)

What is Heat? n n Perception as to hot and cold defined relative to our own body temperature, i. e. object is hotter or colder than oneself Objective measurement of temperature Macroscopic, display of temperature gauge ® Microscopic behaviour of atoms and molecules ® I feel hot

Measuring temperature n Properties of materials change with temperature ® Length ® Volume ® Resistance

Hotter things become longer n Solids get bigger when they get hot ®A 1 metre long bar heated by 1 degree gets bigger by • Steel ≈0. 01 mm • Glass ≈ 0. 001 mm Rails expand may buckle on a hot summer day

Hotter things take up more volume -1 n Most materials get bigger when they get hot (but not water 0°C > 4°C gets smaller!) ® Thermometer relies on a thermal expansion of a liquid (e. g. mercury) Thin tube (Gives big length change for small increase in volume) Large volume of reservoir

Hotter things change their resistance n All hotter metals have a higher electrical resistance or conductivity ® Digital n thermometer All hotter semiconductors have a lower electrical resistance ® key definition between to distinguish metals and insulators!

Example You have a (glass) jar and you can’t get the metal lid off. What should you do: a) ask your girlfriend b) run the jar & lid under cold water c) run the jar & lid under hot water

Solution: a) ask your girlfriend b) run the jar & lid under cold water c) run the jar & lid under hot water Because the metal has a substantially higher coefficient of thermal expansion than the glass, heating them will make both of them bigger, but the metal will be more bigger.

Insulation n Example of good (thermal) insulators ® ® n A vacuum, polystyrene, fibreglass, plastic, wood, brick (low density/foam structure, poor electrical conductors) Examples of poor insulators, i. e. good conductors ® ® ® Most metals (but stainless steel better than copper) e. g. gold contact used within IC chips to prevent heating Gases, liquids (high density, “mobile”, good electrical conductors)

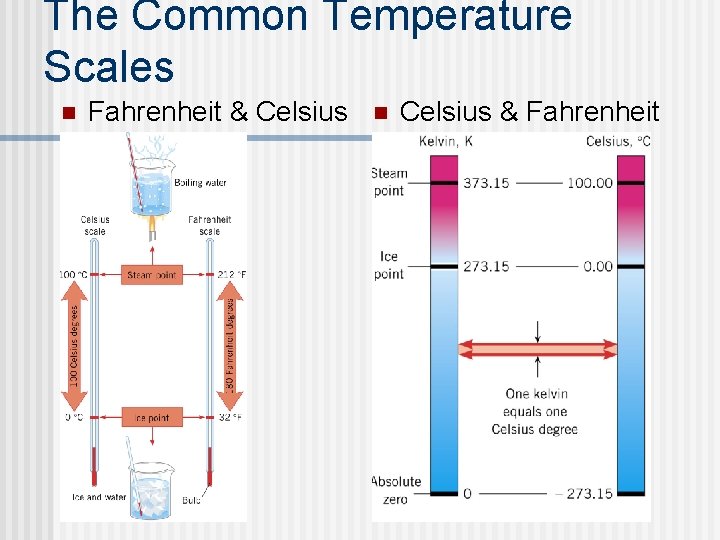
Temperature and scales n Temperature scales (melting & boiling of water) ® Degrees Celsius (MP 0°C 100°C) ® Degrees Kelvin (MP 273. 15 K BP 373. 15 K) ® Degree Fahrenheit (MP 32° F BP 212°F) n Explain how a temperature scale is constructed.

The Common Temperature Scales n Fahrenheit & Celsius n Celsius & Fahrenheit

Converting between scales State the relation between Kelvin & Celsius Scales. n Kelvin to Celsius ®K = C + 273. 15 ® C = K - 273. 15 n Fahrenheit to Celsius (Not IB) ®F = C x (9/5) + 32 ® C = (F - 32) x (5/9)

Example n n Convert the following temperatures into °F and K Boiling water, 100°C Freezing water, 0°C Absolute zero, -273. 15°C 212°F, 373. 15 K 32°F, 273. 15 K -460°F, 0 K

Stop here

Type of thermometer Change in electrical resistance (convenient but not very linear) n Change in length of a bar (bimetallic strip) n Change in volume of a liquid n Change in volume of gas (very accurate but slow and bulky) n

Heat and internal energy n Can you describe the difference between the terms. ® Temperature ® Heat ® Internal Energy

Temperature & Absolute Temperature n Temperature is a property that determines the direction of thermal energy transfer between two bodies in thermal contact. n Absolute temperature is a measure of the average kinetic energy of the molecules of a substance. ® Average kinetic energy is proportional to absolute temperature in Kelvin.

Example n What is the kinetic energy of an oxygen molecule at room temperature ( 21˚C)? KE = 3/2 k. T = 3/2(1. 38 x 10 -23 x 294) = 6. 09 x 10 -21 Joules (k = 1. 38 x 10 -23 J/K) We could equate n Since we know the kinetic energy, how is it travelling? This is called the root mean squared speed or rms speed. KE = 1/2 mv 2 = KE = 3/2 k. T and get v 2 = 3 k. T/m mass must be in kg!!!! Not u.

Heat (Energy) Is the flow of energy in or out of a system. n Heat (energy) flows because of temperature difference n ® Bigger temperature difference bigger heat flow ® Less insulation give more heat flow for the same temperature difference n Heat will not flow between two bodies of the same temperature

Equilibrium n Two objects of different temperature when placed in contact will reach the same temperature + = Cold milk Light brown coffee Hot black coffee

Heat transfer = energy transfer n Energy measured in Joules but heat often measured in Calories One cal raises one gram of water from 14. 5°C to 15. 5°C ® 1 cal = 4. 186 J ® n Doing work on something usually makes it hot ® n Joules Experiment! 1 st law of thermodynamics heat and work are both forms of energy

Sir James Joule n n James Prescott Joule 1818 -1889 Stirring water made it warm Change in temperature proportional to work done ® Showing equivalence of heat and mechanical energy ® n Also that electrical current flow through a resistor causes heating

Joules Experiment

Internal Energy n Is the total potential and kinetic energy of the molecules in a substance. ® Potential energy is associated with intermolecular forces. ® Kinetic energy includes both translational and rotational motion.

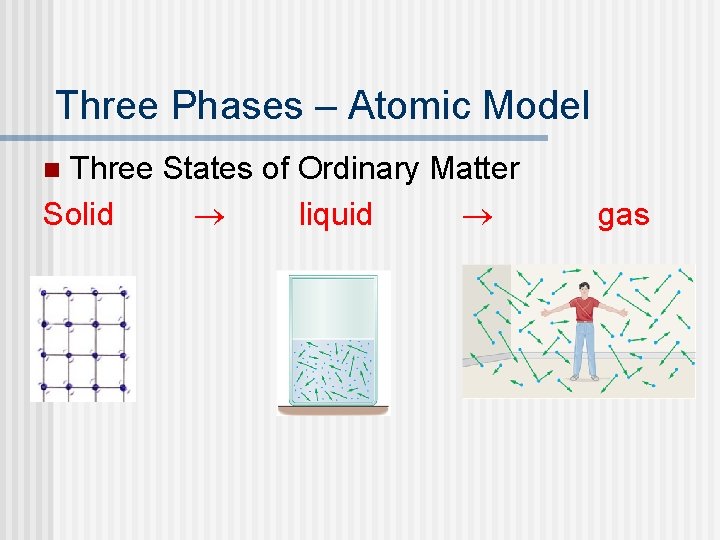
Three Phases – Atomic Model Three States of Ordinary Matter Solid ® liquid ® n gas

Atomic Model of Matter n Comparing Molecular Forces ® Solid – Largest molecular forces ® Liquid ® Gas – Weakest molecular forces • When the kinetic energy of the molecules become comparable to the energy required for separation the molecules change there position and separate (PE increase). This is a phase transition • Melting or vaporizing

Avogadro constant n One mole a any substance is that quantity of the substance whose mass in grams is numerically equal to the substances molar mass, μ. ® EX: The moloar mass of O 2 is 32 g mol-1 ® NA = 6. 02 x 1023 molecules mol-1

Example n How many grams are there in a quantity of oxygen gas containing 1. 2 x 1025 molecules? The number of moles is (1. 20 x 1025)/6. 02 x 1023 = 19. 93 mol Since the molar mass is 32 g mol-1 The mass is 19. 93 x 32 = 638 g or 0. 638 kg

Example n So, how fast is that O 2 molecule traveling? ® O 2 = 32 g/mole v 2 = 3 k. T/m (rms speed of a molecule) m = 0. 032/(6. 023 x 1023) = 5. 3 x 10 -26 kg v 2 = 3(1. 38 x 10 -23 J/K)(294)/(5. 3 x 10 -26 kg) v = 479 m/sec

Transferring heat energy n 3 mechanisms ® Conduction • Heat transfer through material ® Convection • Heat transfer by movement of hot material ® Radiation • Heat transfer by light

Conduction of heat n Conduction in solids ® n Heat energy causes atoms to vibrate, a vibrating atom passes this vibration to the next Conduction in metal ® Heat energy causes electrons to gain energy, electrons travel through metal (conduction) and carry heat energy with them • Metals are good conductors of both heat and electricity

Conduction of heat n The atoms at the bottom are at a higher temperature and will oscillating more strongly than those at the top.

Rate of heat flow n Heat flow (H) is energy transfer per unit time, depends on ® ® Temperature difference Thermal conductivity (k) • k (copper) = 385 W/(m K) • k (glass) = 0. 8 W/(m K) • k (air) =0. 02 W/(m K) A TH L TC

Example n Two rods of the same cross-sectional area are joined together. The right rod is a better conductor of heat than the rod at left. Heat entering the joint must equal the The ends are kept at heat leaving the joint. (Conservation of Energy). Hence, the rate of heat fixed temperatures. ® ® In which rod is the rate of heat transfer the largest? Is the temperature at the joining point lower are higher than 54 ˚C? transfer is the same. Since the seconductor is poor a much larger temperature gradient can be maintained. Thus, the temperature at the junction will be larger.

Thermal conduction vs thermal resistance n Also can use thermal resistance, cf n Can make equation of heat flow more general

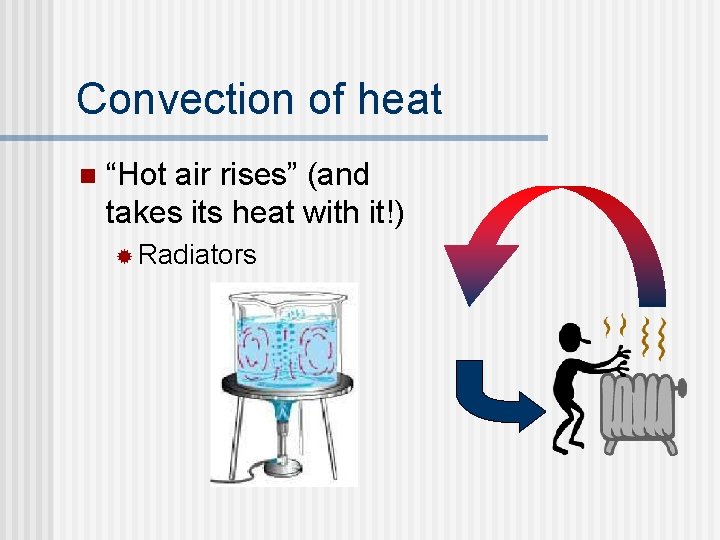
Convection of heat n “Hot air rises” (and takes its heat with it!) ® Radiators

Convection of heat n “Hot air rises” (and takes its heat with it!) ® Cumulus clouds

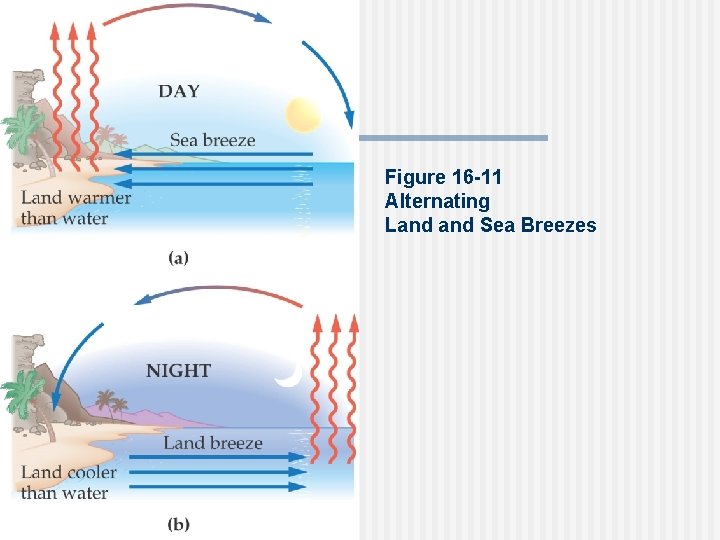
Figure 16 -11 Alternating Land Sea Breezes

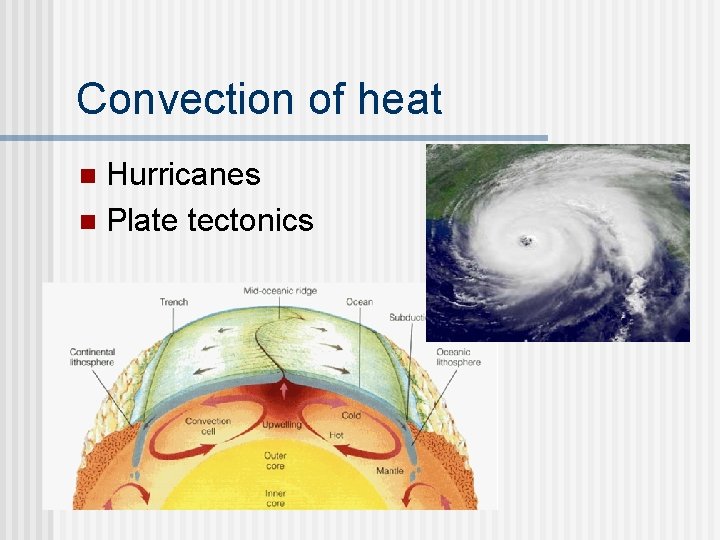
Convection of heat Hurricanes n Plate tectonics n

Radiation of heat n n n Don’t confuse with radioactivity Instead realize that light carries heat (e. g. the sun heats the earth) Anything above absolute zero radiates heat ® P a AT 4 Stefan-Boltzmann law.

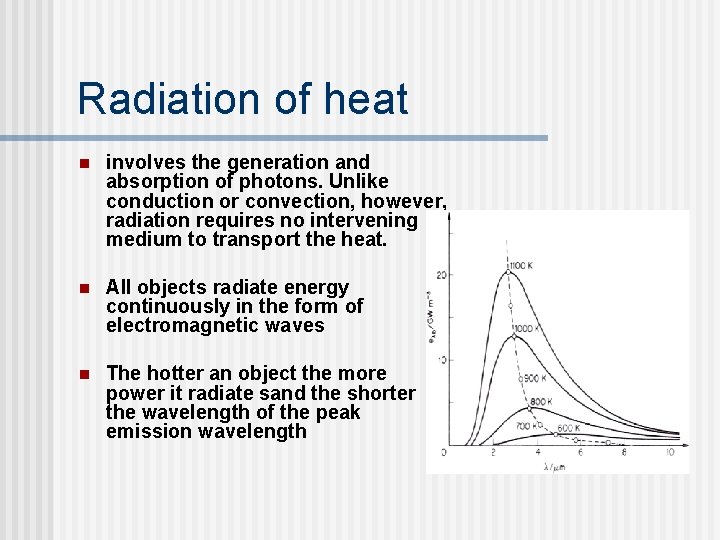
Radiation of heat n involves the generation and absorption of photons. Unlike conduction or convection, however, radiation requires no intervening medium to transport the heat. n All objects radiate energy continuously in the form of electromagnetic waves n The hotter an object the more power it radiate sand the shorter the wavelength of the peak emission wavelength

Not all things emit heat the same n Heat emission from an object area A ®P • • = Aes. T 4 s = Stafan’s constant = 5. 6 x 10 -8 W/(m 2 K 4) e = emissivity of a body, 0 -1 ecopper = 0. 3 ecarcoal ≈ 1

Example n Estimate the upper limit to the heat emission of the sun Suns temperature 7000 k ® Sun’s radius 7 x 108 m ® Emission, P = Aes. T 4 Area = 4 pr 2 = 6. 2 x 1018 m 2 Emissivity ≈ 1 H = 6. 2 x 1018 x 5. 6 x 10 -8 x 70004 Sun’s output = 8. 3 x 1026 W

Are heat emitter also good absorbers? n Black and dull on the surface ® ® ® Best emitter/absorber Charcoal Blackbody radiators • perfect absorber & emitter n White and polished/shiny ® ® Good Reflectors Stay cool in the summer

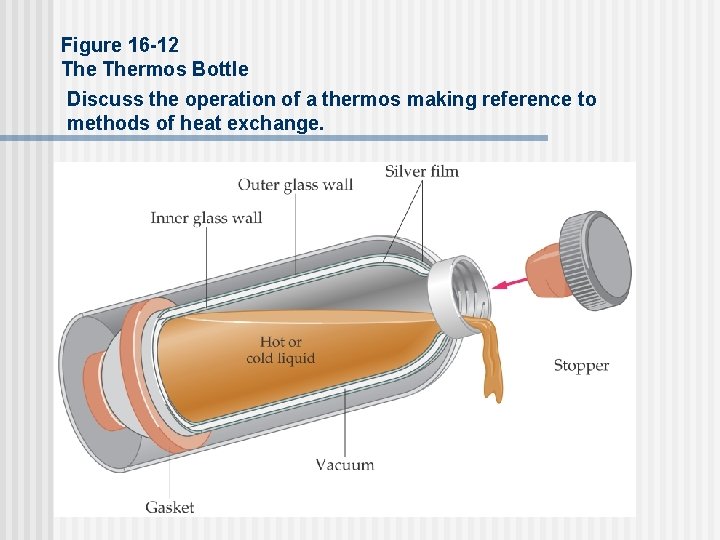
Figure 16 -12 Thermos Bottle Discuss the operation of a thermos making reference to methods of heat exchange.

Assignment n Questions from Packet. 1, 2, 3, 5, 7, 10, 11, 12, 14, 18, 19.

The “colour” of heat n n Peak wavelength of light emitted depends on temperature Spectrum includes all wavelength longer than the peak but not many above 20°C - peak in infrared (need thermal imaging camera to see body heat) ® 800°C - peak in red (electric coil, fire glows reds) ® 3000° - peak in blue (but includes green and red light hence appears white) ® 2. 7 K peak in micro-wave (background emission in the universe left over from the Big Bang) ®