Thermodynamics Lecture Series Pure substances Property tables and

Thermodynamics Lecture Series Pure substances – Property tables and Property Diagrams & Ideal Gases Applied Sciences Education Research Group (ASERG) Faculty of Applied Sciences Universiti Teknologi MARA email: drjjlanita@hotmail. com http: //www 5. uitm. edu. my/faculties/fsg/drjj 1. html
Submitted-Post-assessment
Unreadable Self-assessment

CHAPTER 2 Pure substance Properties of Pure Substances- Part 2 Send self-assessments to: Thermopre@salam. uitm. edu. my Thermopost@salam. uitm. edu. my

Quotes "Education is an admirable thing, but it is well to remember from time to time that nothing that is worth knowing can be taught. " taught - Oscar Wilde "What we have to learn to do, we learn by doing. " doing -Aristotle

Introduction Objectives: 1. Choose the right property table to read and to determine phase and other properties. 2. Derive and use the mathematical relation to determine values of properties in the wet-mix phase 3. Sketch property diagrams with respect to the saturation lines, representing phases, processes and properties of pure substances.

Introduction Objectives: 4. Use an interpolation technique to determine unknown values of properties in the superheated vapor region 5. State conditions for ideal gas behaviour 6. Write the equation of state for an ideal gas in many different ways depending on the units. 7. Use all mathematical relations and skills of reading the property table in problem-solving.
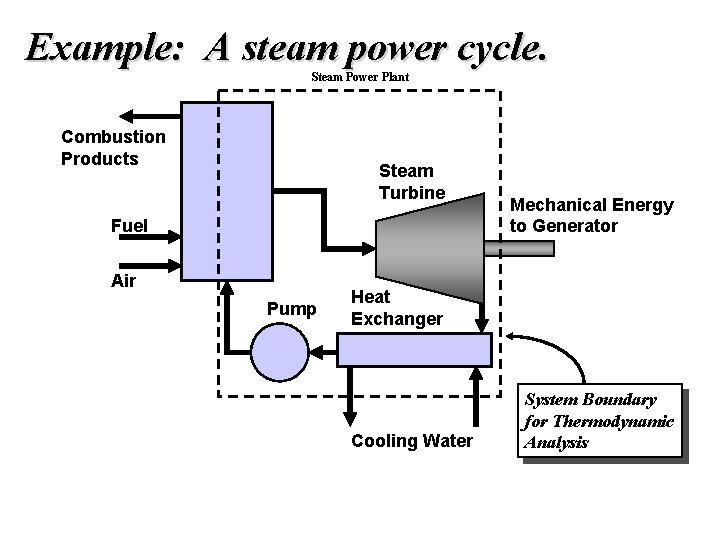
Example: A steam power cycle. Steam Power Plant Combustion Products Steam Turbine Fuel Air Pump Mechanical Energy to Generator Heat Exchanger Cooling Water System Boundary for Thermodynamic Analysis
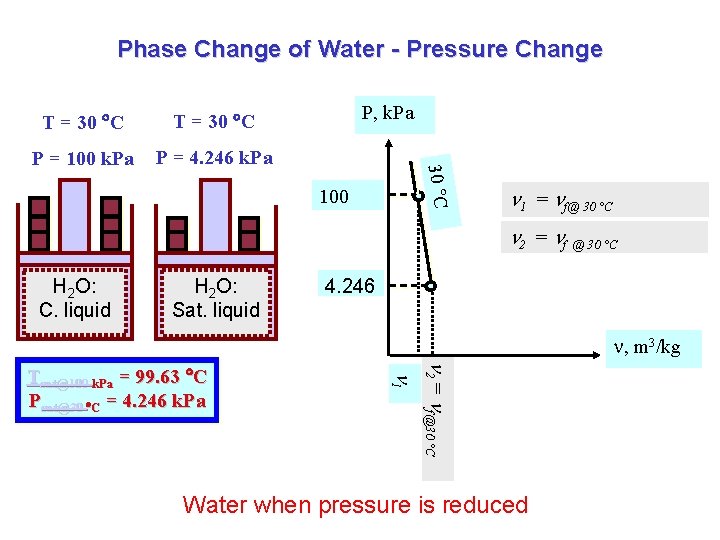
Phase Change of Water - Pressure Change T = 30 C P = 100 k. Pa P = 4. 246 k. Pa P, k. Pa 30 C T = 30 C 100 1 = f@ 30 °C 2 = f @ 30 °C H 2 O: C. liquid H 2 O: Sat. liquid 4. 246 , m 3/kg 2 = f@30 °C 1 Tsat@100 k. Pa = 99. 63 C Psat@30 C = 4. 246 k. Pa Water when pressure is reduced
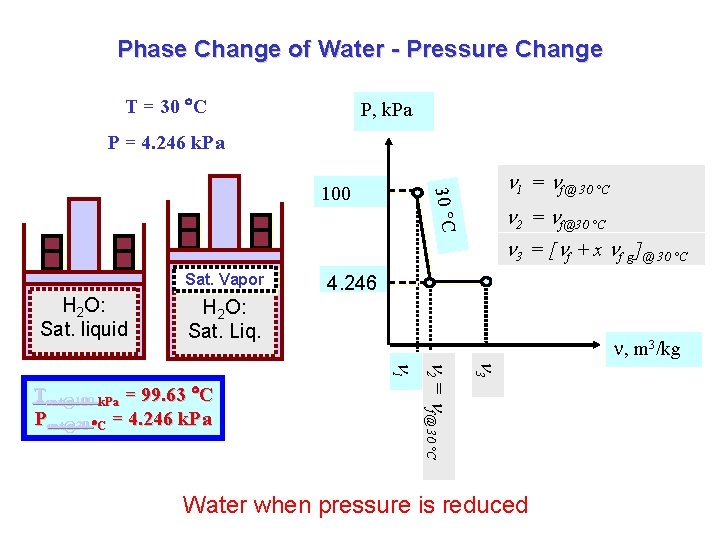
Phase Change of Water - Pressure Change T = 30 C P, k. Pa P = 4. 246 k. Pa 1 = f@ 30 °C 30 C 100 2 = f@30 °C 3 = [ f + x f g]@ 30 °C Sat. Vapor H 2 O: Sat. liquid 4. 246 H 2 O: Sat. Liq. , m 3/kg 3 2 = f@ 30 °C 1 Tsat@100 k. Pa = 99. 63 C Psat@30 C = 4. 246 k. Pa Water when pressure is reduced
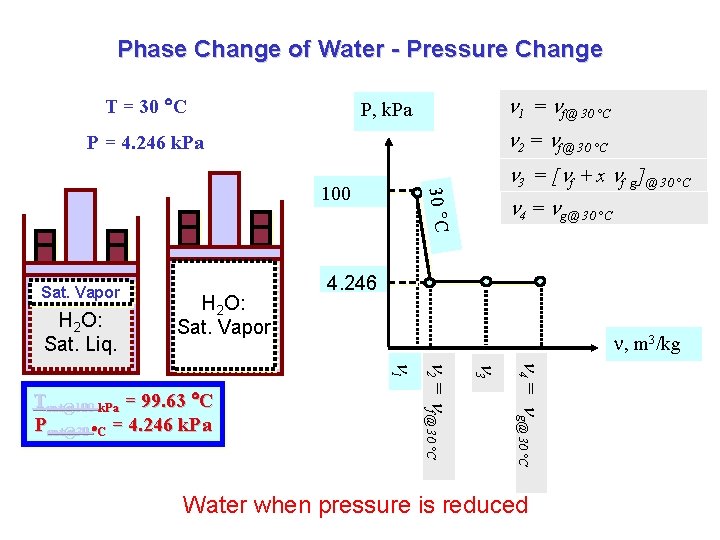
Phase Change of Water - Pressure Change T = 30 C 1 = f@ 30 °C P, k. Pa 2 = f@ 30 °C P = 4. 246 k. Pa Sat. Vapor H 2 O: Sat. Liq. H 2 O: Sat. Vapor 30 C 100 4 = g@ 30 °C 4. 246 , m 3/kg 4 = g@ 30 °C 3 2 = f@ 30 °C 1 Tsat@100 k. Pa = 99. 63 C Psat@30 C = 4. 246 k. Pa 3 = [ f + x f g]@ 30 °C Water when pressure is reduced
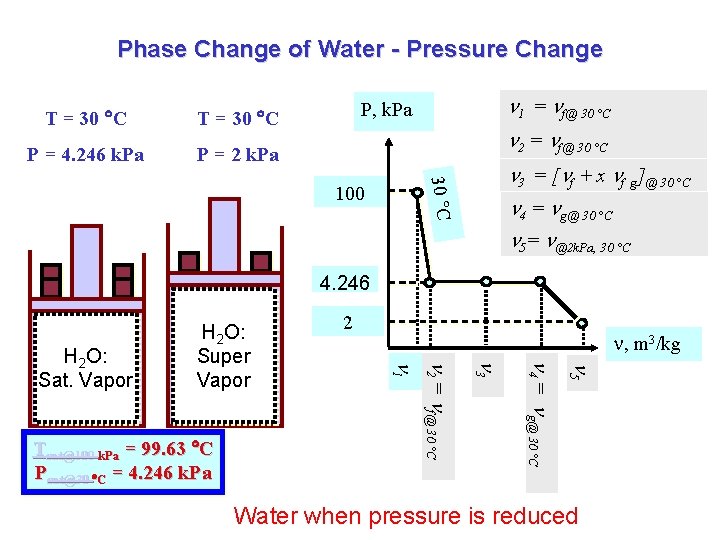
Phase Change of Water - Pressure Change T = 30 C P = 4. 246 k. Pa P = 2 k. Pa 1 = f@ 30 °C P, k. Pa 2 = f@ 30 °C 30 C 3 = [ f + x f g]@ 30 °C 100 4 = g@ 30 °C 5= @2 k. Pa, 30 °C 4. 246 , m 3/kg 5 4 = g@ 30 °C 3 2 = f@ 30 °C f@100 k. Pa Tsat@100 k. Pa = 99. 63 C Psat@30 C = 4. 246 k. Pa 2 1 H 2 O: Sat. Vapor H 2 O: Super Vapor Water when pressure is reduced
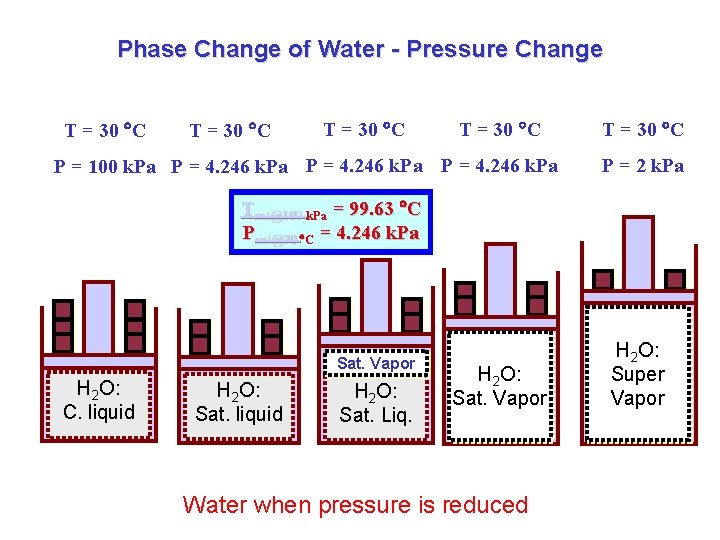
Phase Change of Water - Pressure Change T = 30 C P = 100 k. Pa P = 4. 246 k. Pa T = 30 C P = 2 k. Pa Tsat@100 k. Pa = 99. 63 C Psat@30 C = 4. 246 k. Pa Sat. Vapor H 2 O: C. liquid H 2 O: Sat. Liq. H 2 O: Sat. Vapor Water when pressure is reduced H 2 O: Super Vapor
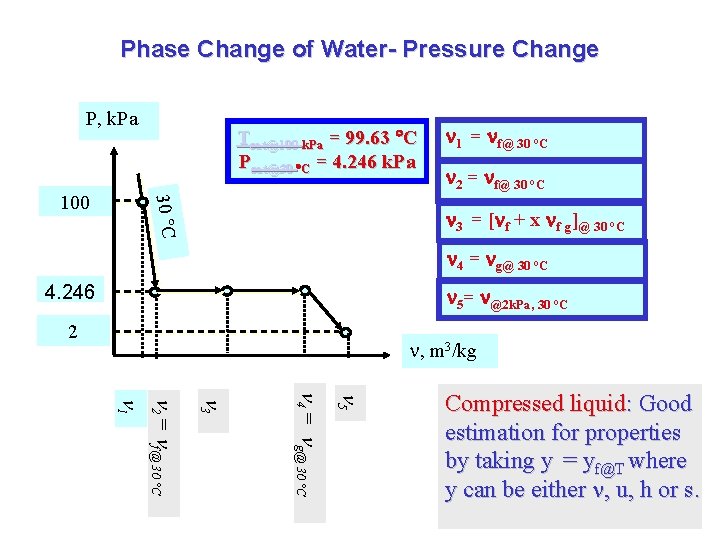
Phase Change of Water- Pressure Change P, k. Pa 30 C 100 Tsat@100 k. Pa = 99. 63 C Psat@30 C = 4. 246 k. Pa 1 = f@ 30 °C 2 = f@ 30 °C 3 = [ f + x f g]@ 30 °C 4 = g@ 30 °C 4. 246 5= @2 k. Pa, 30 °C 2 , m 3/kg 5 4 = g@ 30 °C 3 2 = f@ 30 °C 1 Compressed liquid: Good estimation for properties by taking y = yf@T where y can be either , u, h or s.
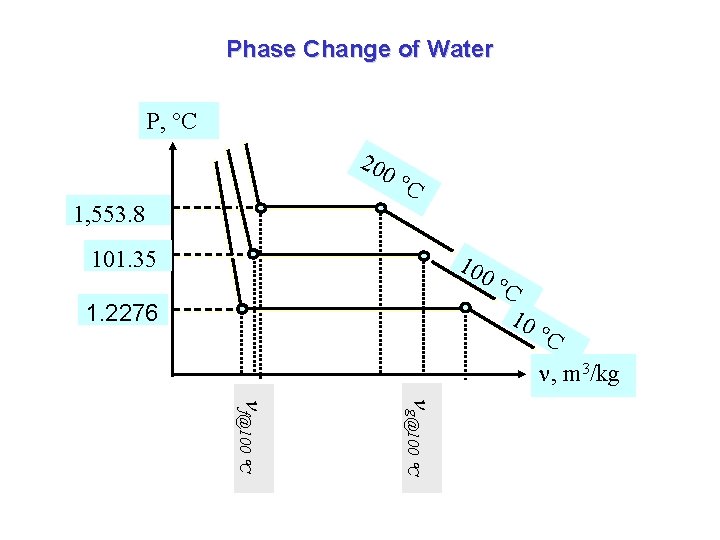
Phase Change of Water P, C 20 0 C 1, 553. 8 101. 35 10 1. 2276 0 C 10 C , m 3/kg g@100 C f@100 C
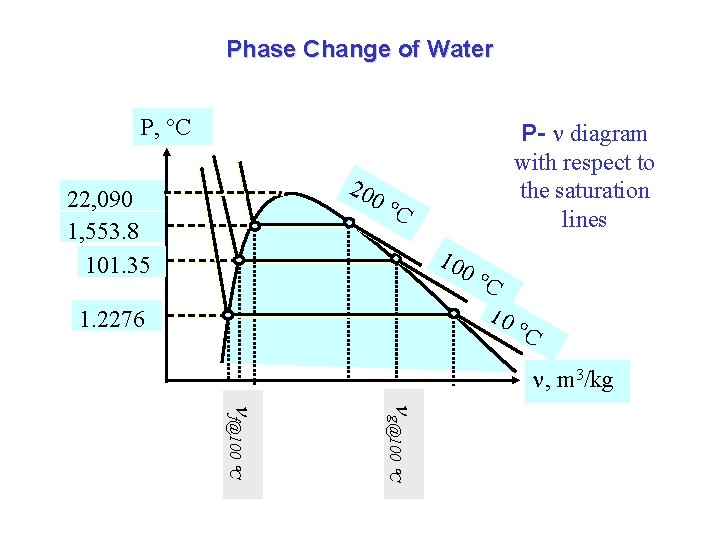
Phase Change of Water P, C 20 22, 090 1, 553. 8 101. 35 0 P- diagram with respect to the saturation lines C 10 1. 2276 0 C 10 C , m 3/kg g@100 C f@100 C
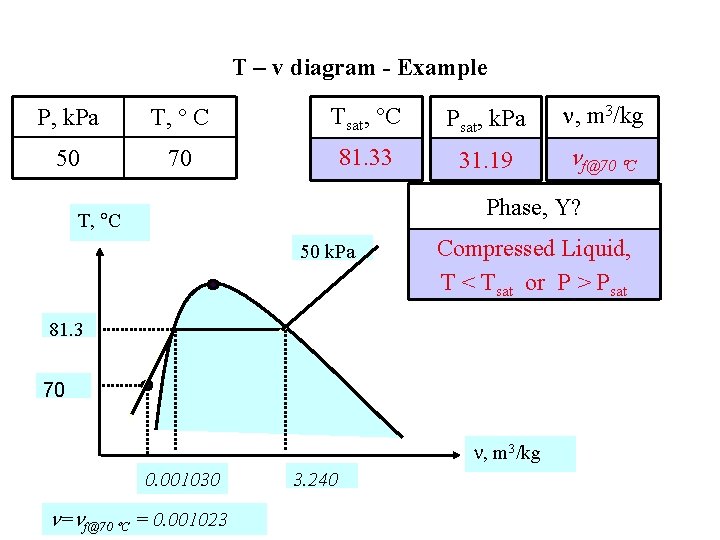
T – v diagram - Example P, k. Pa T, C Tsat, C Psat, k. Pa , m 3/kg 50 70 81. 33 31. 19 f@70 C Phase, Y? T, C 50 k. Pa Compressed Liquid, T < Tsat or P > Psat 81. 3 70 , m 3/kg 0. 001030 = f@70 C = 0. 001023 3. 240
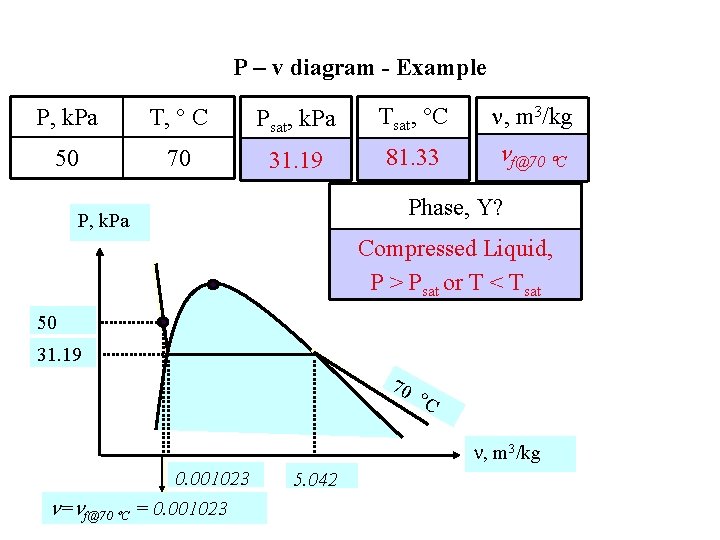
P – v diagram - Example P, k. Pa T, C Psat, k. Pa Tsat, C , m 3/kg 50 70 31. 19 81. 33 f@70 C Phase, Y? P, k. Pa Compressed Liquid, P > Psat or T < Tsat 50 31. 19 70 C , m 3/kg 0. 001023 = f@70 C = 0. 001023 5. 042
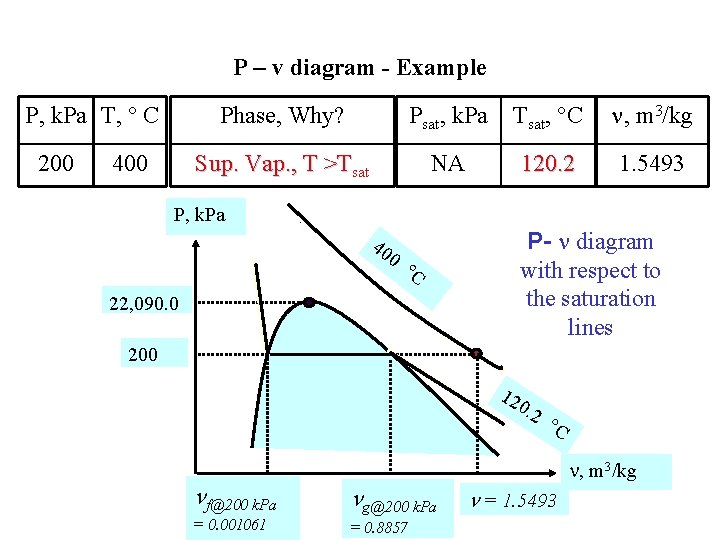
P – v diagram - Example P, k. Pa T, C 200 400 Phase, Why? Psat, k. Pa Tsat, C , m 3/kg Sup. Vap. , T >Tsat NA 120. 2 1. 5493 P, k. Pa 40 0 P- diagram with respect to the saturation lines C 22, 090. 0 200 12 0. 2 f@200 k. Pa g@200 k. Pa = 0. 001061 = 0. 8857 C , m 3/kg = 1. 5493
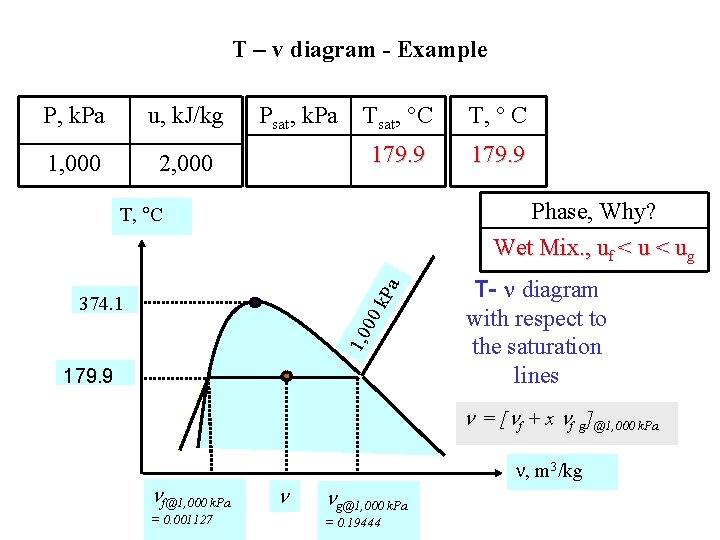
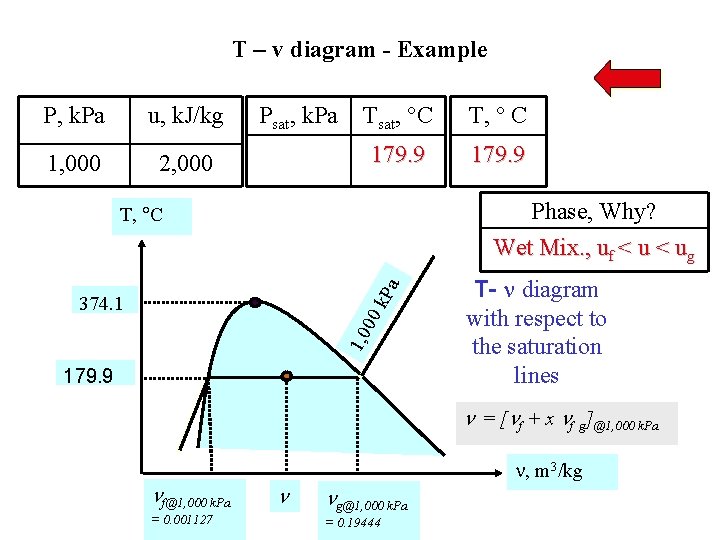
T – v diagram - Example P, k. Pa u, k. J/kg 1, 000 2, 000 Psat, k. Pa Tsat, C T, C 179. 9 Phase, Why? Wet Mix. , uf < ug k. P a T, C 1, 0 00 374. 1 179. 9 T- diagram with respect to the saturation lines = [ f + x f g]@1, 000 k. Pa f@1, 000 k. Pa = 0. 001127 , m 3/kg g@1, 000 k. Pa = 0. 19444
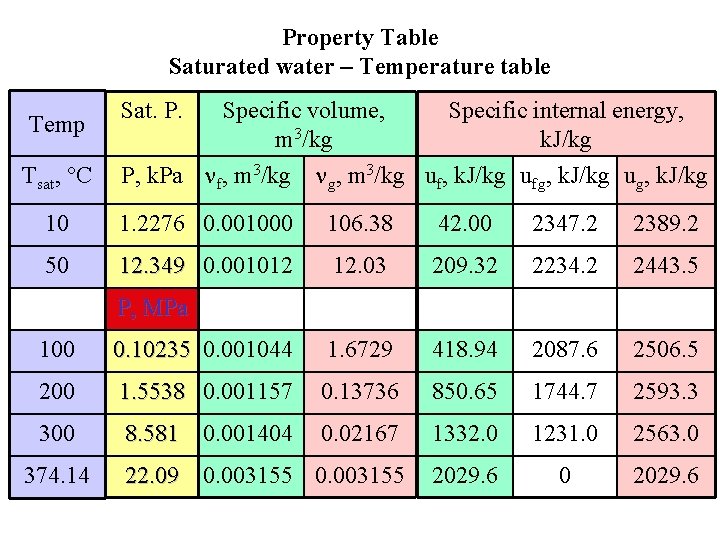
Property Table Saturated water – Temperature table Temp Tsat, C Specific volume, Specific internal energy, m 3/kg k. J/kg P, k. Pa f, m 3/kg g, m 3/kg uf, k. J/kg ufg, k. J/kg ug, k. J/kg Sat. P. 10 1. 2276 0. 001000 106. 38 42. 00 2347. 2 2389. 2 50 12. 349 0. 001012 12. 03 209. 32 2234. 2 2443. 5 P, MPa 100 0. 10235 0. 001044 1. 6729 418. 94 2087. 6 2506. 5 200 1. 5538 0. 001157 0. 13736 850. 65 1744. 7 2593. 3 300 8. 581 0. 001404 0. 02167 1332. 0 1231. 0 2563. 0 22. 09 0. 003155 2029. 6 0 2029. 6 374. 14
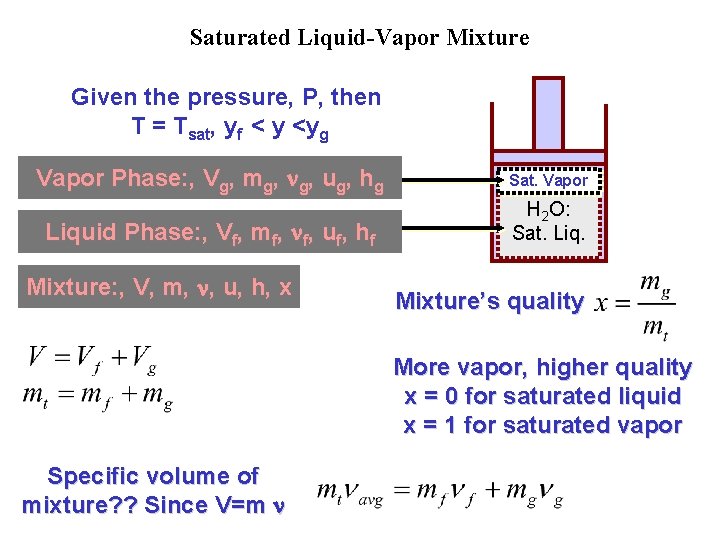
Saturated Liquid-Vapor Mixture Given the pressure, P, then T = Tsat, yf < y <yg Vapor Phase: , Vg, mg, g, ug, hg Sat. Vapor Liquid Phase: , Vf, mf, f, uf, hf H 2 O: Sat. Liq. Mixture: , V, m, , u, h, x Mixture’s quality More vapor, higher quality x = 0 for saturated liquid x = 1 for saturated vapor Specific volume of mixture? ? Since V=m
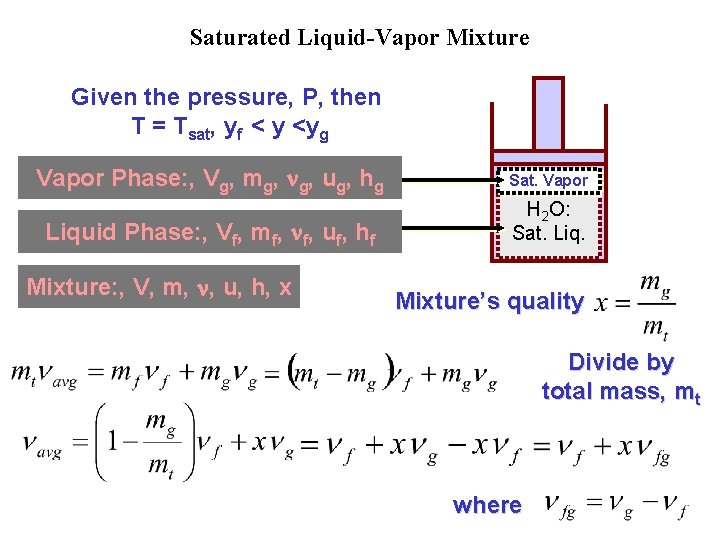
Saturated Liquid-Vapor Mixture Given the pressure, P, then T = Tsat, yf < y <yg Vapor Phase: , Vg, mg, g, ug, hg Sat. Vapor Liquid Phase: , Vf, mf, f, uf, hf H 2 O: Sat. Liq. Mixture: , V, m, , u, h, x Mixture’s quality Divide by total mass, mt where
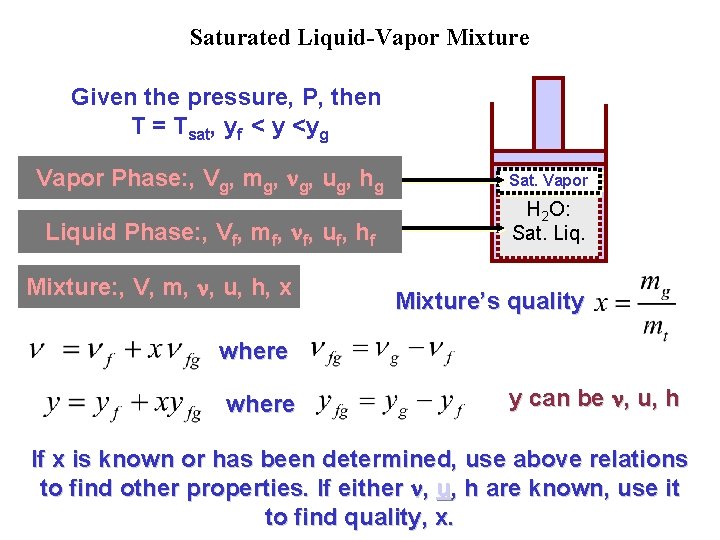
Saturated Liquid-Vapor Mixture Given the pressure, P, then T = Tsat, yf < y <yg Vapor Phase: , Vg, mg, g, ug, hg Sat. Vapor Liquid Phase: , Vf, mf, f, uf, hf H 2 O: Sat. Liq. Mixture: , V, m, , u, h, x Mixture’s quality where y can be , u, h If x is known or has been determined, use above relations to find other properties. If either , u, h are known, use it to find quality, x.
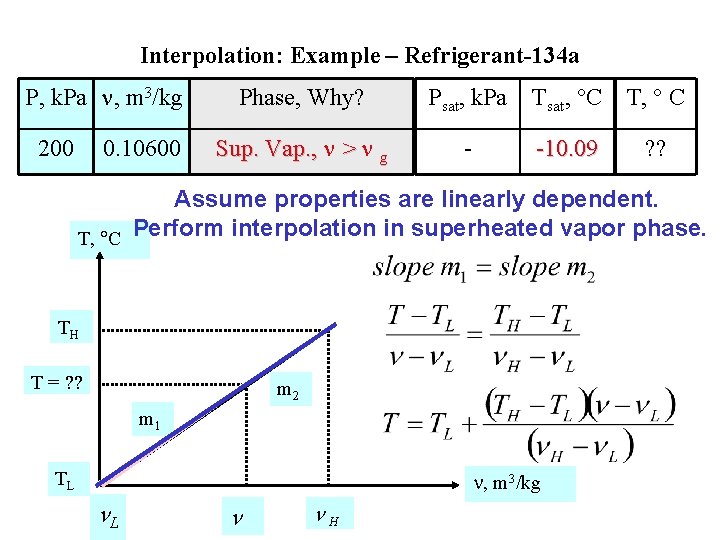
Interpolation: Example – Refrigerant-134 a P, k. Pa , m 3/kg 200 0. 10600 Phase, Why? Psat, k. Pa Tsat, C T, C Sup. Vap. , > g - -10. 09 ? ? Assume properties are linearly dependent. T, C Perform interpolation in superheated vapor phase. TH T = ? ? m 2 m 1 TL , m 3/kg L H

Interpolation: Example – Refrigerant-134 a P, k. Pa , m 3/kg 200 0. 10600 Phase, Why? Psat, k. Pa Tsat, C T, C Sup. Vap. , > g - -10. 09 ? ? T, C , m 3/kg u, k. J/kg T L= 0 L = 0. 10438 u. L = 229. 23 0 < TL < TH = 0. 10600 0 < u. L < u. H TH= 10 H = 0. 10922 u. H = 237. 05 Assume properties are linearly dependent. Perform interpolation in superheated vapor phase.
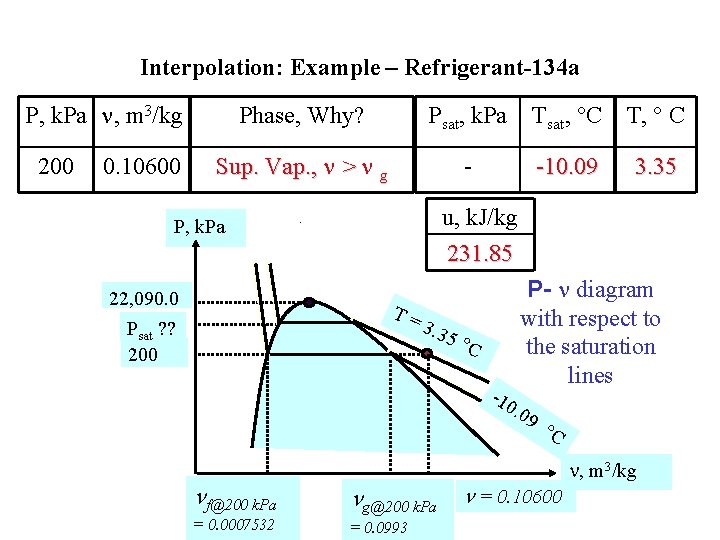
Interpolation: Example – Refrigerant-134 a P, k. Pa , m 3/kg 200 0. 10600 Phase, Why? Psat, k. Pa Tsat, C T, C Sup. Vap. , > g - -10. 09 3. 35 u, k. J/kg 231. 85 P, k. Pa 22, 090. 0 Psat ? ? 200 T= 3. 3 5 C P- diagram with respect to the saturation lines -10 . 09 f@200 k. Pa g@200 k. Pa = 0. 0007532 = 0. 0993 C = 0. 10600 , m 3/kg

Ideal Gases Properties of Pure Substances- Ideal Gases Equation of State

Ideal Gases • Equation of State – An equation relating pressure, temperature and specific volume of a substance. – Predicts P- -T behaviour quite accurately – Any properties relating to other properties – Simplest EQOS of substance in gas phase is ideal-gas (imaginary gas) equation of state

Ideal Gases • Equation of State for ideal gas – Boyle’s Law: Pressure of gas is inversely proportional to its specific volume P • Equation of State for ideal gas – Charles’s Law: At low pressure, volume is proportional to temperature

Ideal Gases • Equation of State for ideal gas – Combining Boyles and Charles laws: and where M is molar mass where gas constant R is and where Ru is universal gas constant Ru = 8. 314 k. J/kmol. K EQOS: Since the total volume is V = m , so : = V/m EQOS: since the mass m = MN where N is number of moles: So,

Ideal Gases • Equation of State for ideal gas – Real gases with low densities behaves like an ideal gas Hence real gases satisfying conditions P << Pcr, T >> Tcr Obeys EQOS where, Ru = 8. 314 k. J/kmol. K, m = MN V = m and
Gas Mixtures – Ideal Gases ØLow density (mass in 1 m 3) gases Molecules are further apart ØReal gases satisfying condition Pgas << Pcrit; Tgas >> Tcrit , have low density and can be treated as ideal gases High density Low density Molecules far
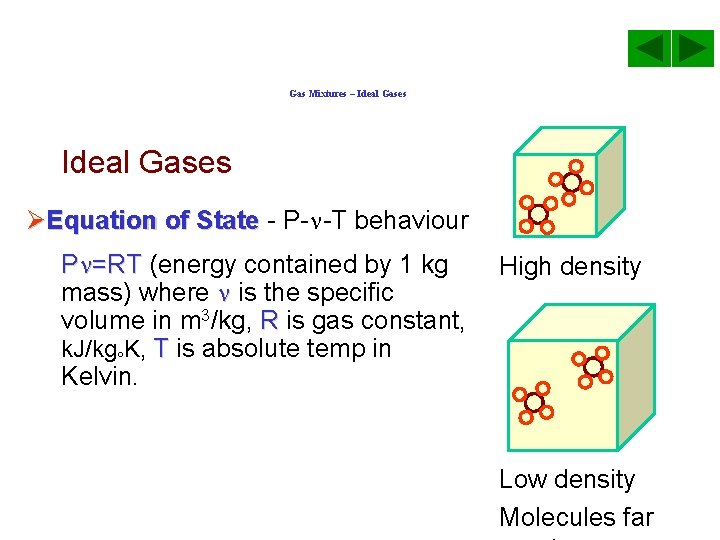
Gas Mixtures – Ideal Gases ØEquation of State - P- -T behaviour P =RT (energy contained by 1 kg mass) where is the specific volume in m 3/kg, R is gas constant, k. J/kg K, T is absolute temp in Kelvin. High density Low density Molecules far
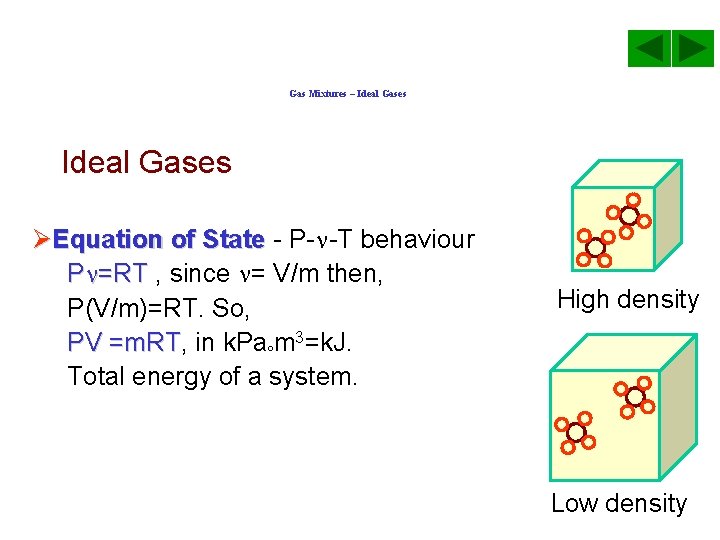
Gas Mixtures – Ideal Gases ØEquation of State - P- -T behaviour P =RT , since = V/m then, P(V/m)=RT. So, PV =m. RT, m. RT in k. Pa m 3=k. J. Total energy of a system. High density Low density

Gas Mixtures – Ideal Gases ØEquation of State - P- -T behaviour PV =m. RT = NMRT = N(MR)T Hence, can also write PV = NRu. T where N is no of kilomoles, kmol, M is molar mass in kg/kmole and Ru is universal gas constant; Ru=MR. =MR Ru = 8. 314 k. J/kmol K High density Low density
T – v diagram - Example P, k. Pa u, k. J/kg 1, 000 2, 000 Psat, k. Pa Tsat, C T, C 179. 9 Phase, Why? Wet Mix. , uf < ug k. P a T, C 1, 0 00 374. 1 179. 9 T- diagram with respect to the saturation lines = [ f + x f g]@1, 000 k. Pa f@1, 000 k. Pa = 0. 001127 , m 3/kg g@1, 000 k. Pa = 0. 19444
- Slides: 37