Thermodynamics II Acidbase properties Andy Howard Biochemistry Lectures

Thermodynamics II; Acid-base properties Andy Howard Biochemistry Lectures, Spring 2019 22 January 2019, IIT

Thermodynamics; Acid-base reactions • Thinking carefully about entropy helps us • • make sense of biochemical thermodynamics Protonation and deprotonation matter Amino acids exemplify a lot of basic biochemical concepts, and they’re inherently important too 01/22/2019 Thermo II; Acid-base p. 2 of 71

What we’ll discuss • Thermodynamics II – – – Free energy Le Chatelier ATP Energy examples Protein folding • Acid-base concepts – Weak acids – Henderson. Hasselbalch • Amino Acids – What they are – -amino acids 01/22/2019 Thermo II; Acid-base p. 3 of 71

Free energy and equilibrium • Go = -RT ln keq, or keq = exp(- Go/RT) • keq is equilibrium constant; formula depends on reaction type • For a. A + b. B c. C + d. D, keq = ([C]c[D]d)/([A]a[B]b) • If all proportions are equal, keq = ([C][D])/([A][B]) • These values ([C], [D] …) denote the concentrations at equilibrium 01/22/2019 Thermo II; Acid-base p. 4 of 71

Spontaneity and free energy • Thus if reaction is just spontaneous, • • • i. e. Go = 0, then keq = 1 If Go < 0, then keq > 1: Exergonic If Go > 0, then keq < 1: Endergonic Distinguishable from exothermic and endothermic, which are concerned only with enthalpy, not the whole free-energy package. 01/22/2019 Thermo II; Acid-base p. 5 of 71

Free energy as a source of work • Change in free energy indicates that • • 01/22/2019 the reaction could be used to perform useful work If Go < 0, the system supplies work If Go > 0, we need to do work on the system to make the reaction occur Thermo II; Acid-base p. 6 of 71

What kind of work? • Movement (flagella, muscles) • Chemical work: – Transport molecules against concentration gradients – Transport ions against potential gradients • Driving otherwise endergonic reactions – by direct coupling of reactions – by depletion of products 01/22/2019 Thermo II; Acid-base p. 7 of 71

Coupled reactions • Often a single enzyme catalyzes 2 • • reactions, shoving them together: reaction 1, A B: Go 1 < 0 reaction 2, C D: Go 2 > 0 Coupled reaction: A + C B + D: Go. C = Go 1 + Go 2 If Go. C < 0, reaction 1 is driving reaction 2! 01/22/2019 Thermo II; Acid-base p. 8 of 71

How else can we win? • Concentration of products & reactants may • • play a role As we’ll discuss in a moment, the actual free energy depends on Go and on concentration of products and reactants So if the first reaction withdraws product of reaction B away, that drives the equilibrium of reaction 2 to the right 01/22/2019 Thermo II; Acid-base p. 9 of 71

Le Chatelier’s Principle & ΔGo • Le Chatelier’s Principle says that a reaction can be • driven to the right if one of the products is being removed from availability This can happen in various ways: – A gaseous product can evaporate – An insoluble product can precipitate – A reactive product can get converted to something else. 01/22/2019 Thermo II; Acid-base p. 10 of 71

Quantitation of Le Chatelier’s Principle • Relationship between G (actual free energy • • • 01/22/2019 experienced in a reaction under real conditions) & standard free energy Go : G = Go + RT ln([products]/[reactants]) So for a typical bimolecular reaction A + B C + D, G = Go + RT ln{[C][D]/([A][B])} Thermo II; Acid-base p. 11 of 71

Why does this embody Le Chatelier’s Principle? • Suppose product D is being taken out of the • reaction vessel (e. g. the mitochondrion) either by being converted to something else or by being transported away. Then [D] will be small, so Q=[C][D]/([A][B]) will be small 01/22/2019 Thermo II; Acid-base p. 12 of 71

Consequence of that! • Therefore ln. Q will be negative, and G = Go + RT ln. Q can become negative even if Go is positive (especially at high T) 01/22/2019 Thermo II; Acid-base p. 13 of 71

How hard is it to o measure G ? • Not as bad as you might think. • It’s true that it’s hard to set up [C]=[D]=[A]=[B] = 1 M; but we don’t actually have to do that. • Note that since G = Go +RTln{[C][D]/([A][B])}, we can get to G = Go simply by ensuring that [C][D]/([A][B]) = 1, so that ln{[C][D]/([A][B]) = 0. 01/22/2019 Thermo II; Acid-base p. 14 of 71

Adenosine Triphosphate • ATP readily available in cells • Derived from catabolic reactions • Contains two high-energy phosphate bonds that can be hydrolyzed to release energy: 01/22/2019 O O|| | (AMP)-O~P-O| || Thermo II; Acid-base O- O p. 15 of 71

Hydrolysis of ATP • Hydrolysis at the rightmost high-energy bond: • • • 01/22/2019 ATP + H 2 O ADP + Pi , Go = -33 k. J/mol Hydrolysis of middle bond: ATP + H 2 O AMP + PPi Go ~ -40 k. J/mol BUT PPi + H 2 O 2 Pi, Go = -31 k. J/mol So, appropriately coupled, we get roughly twice as much! Thermo II; Acid-base p. 16 of 71

ATP as energy currency • Any time we wish to drive a reaction that has • • Go < +30 k. J/mol, we can couple it to ATP hydrolysis and come out ahead If the reaction we want has Go < +60 k. J/mol, we can couple it to ATP AMP and come out ahead So ATP is a convenient source of energy — an energy currency for the cell 01/22/2019 Thermo II; Acid-base p. 17 of 71

Coin analogy • Think of store of ATP • • • as a roll of quarters Vendors don’t give change Use one quarter for some reactions, two for others Inefficient for buying $0. 35 items 01/22/2019 Thermo II; Acid-base p. 18 of 71

Other high-energy compounds • Creatine phosphate: ~ $0. 35 • Phosphoenolpyruvate: ~ $0. 40 • So for some reactions, they’re more efficient than ATP 01/22/2019 Thermo II; Acid-base p. 19 of 71

Why not use those always? • There’s no such thing as a free • • 01/22/2019 lunch! In order to store a compound, you have to create it in the first place So an intermediate-energy currency is the most appropriate Thermo II; Acid-base p. 20 of 71
![Le Chatelier’s principle in ATP-dependent reactions • G = Go + RT ln{[C][D]/([A][B]) • Le Chatelier’s principle in ATP-dependent reactions • G = Go + RT ln{[C][D]/([A][B]) •](http://slidetodoc.com/presentation_image_h/30469319ccaffc79a91bd51b24c3f5a1/image-21.jpg)
Le Chatelier’s principle in ATP-dependent reactions • G = Go + RT ln{[C][D]/([A][B]) • In an ATP-coupled reaction, D=ADP, B=ATP; • 01/22/2019 C and A are the other reactants But note that G = Go + RT ln{[C][D]/([A][B]) = Go + RT { ln([C]/[A])+ ln([ADP]/[ATP])} Thermo II; Acid-base p. 21 of 71
![Why that matters in cells… • The fact is that often [ADP]/[ATP] ~ 0. Why that matters in cells… • The fact is that often [ADP]/[ATP] ~ 0.](http://slidetodoc.com/presentation_image_h/30469319ccaffc79a91bd51b24c3f5a1/image-22.jpg)
Why that matters in cells… • The fact is that often [ADP]/[ATP] ~ 0. 1, so at T=300. 6 K, RTln[ADP]/[ATP] = -5. 8 k. J /mol, so that provides even more available energy to drive the reaction! 01/22/2019 Thermo II; Acid-base p. 22 of 71

How else can Le Chatelier’s principle help? • Often coupled reactions involve • 01/22/2019 withdrawal of a product from availability If that happens, [product] / [reactant] shrinks, the second term becomes negative, and G < 0 even if Go > 0 Thermo II; Acid-base p. 23 of 71

Example: glycolysis • Later this semester we’ll spend at least • • one lecture looking at glycolysis, one of the fundamental pathways Some of the glycolytic reactions have Go’ or Go > 0 But all have G values that are negative or zero because of this concentration effect 01/22/2019 Thermo II; Acid-base p. 24 of 71

How to solve energy problems involving coupled equations • General principles: – If two equations are added, their energetics add – An item that appears on the left and right side of the combined equation can be cancelled – Reversing a reaction reverses the sign of G. • “Hydrolysis” means “reacting with water” 01/22/2019 Thermo II; Acid-base p. 25 of 71

A bit more detail • Suppose we couple two equations: • • A + B C + D, Go’ = x C + F B + G, Go’ = y The result is: A + B + C + F B + C + D + G, or A + F D + G, Go’ = x + y … since B & C appear on both sides 01/22/2019 Thermo II; Acid-base p. 26 of 71

Slightly more complex… • Suppose we couple two equations: • • A + B C + D, Go’ = x H + A J + C, Go’ = z Reverse the second equation: J + C A + H, Go’ = -z Add this to 1 st eqn. & simplify: B + J D + H, Go’ = x - z … since A & C appear on both sides • 01/22/2019 Thermo II; Acid-base p. 27 of 71

What do we mean by hydrolysis? • It simply means a reaction with water • Typically involves cleaving a bond: • U + H 2 O V + W is described as hydrolysis of U to yield V and W 01/22/2019 Thermo II; Acid-base p. 28 of 71

Protein Folding • Proteins (about which we’ll say a lot soon) are • • typically folded into a definite conformation at room temp in solution They unfold into a state where they have no definite conformation, at higher temperature We describe this as melting, and it’s frequently a first-order phase transition 01/22/2019 Thermo II; Acid-base p. 29 of 71

How does that work thermodynamically? • Consider the reaction • • (Protein+Solvent)folded (Protein+Solvent)unfolded This reaction has a free energy: G = H - T S System includes both protein and solvent: G = Gprotein + Gsolvent = Hprotein + Hsolvent - T Sprotein - T Ssolvent 01/22/2019 Thermo II; Acid-base p. 30 of 71

Unfolding: effect on enthalpy • Typically Hprotein > 0 because we lose • some hydrogen bonds and van der Waals interactions when the protein unfolds Typically Hsolvent ~ 0 because we make extra protein-solvent H-bonds and break some protein-protein H-bonds 01/22/2019 Thermo II; Acid-base p. 31 of 71

Unfolding: entropy • Sprotein > 0 because the protein • • 01/22/2019 becomes more disordered Therefore -T Sprotein < 0. Ssolvent < 0 because some water molecules become aggregated around the protein; therefore -T Ssolvent > 0. Thermo II; Acid-base p. 32 of 71

Putting the entropic terms together • Overall entropic term in free energy is • • -T( Sprotein + Ssolvent) These 2 terms have opposite signs but typically the protein term dominates, just barely Therefore the overall term is slightly negative, and it becomes more negative at higher temperature 01/22/2019 Thermo II; Acid-base p. 33 of 71
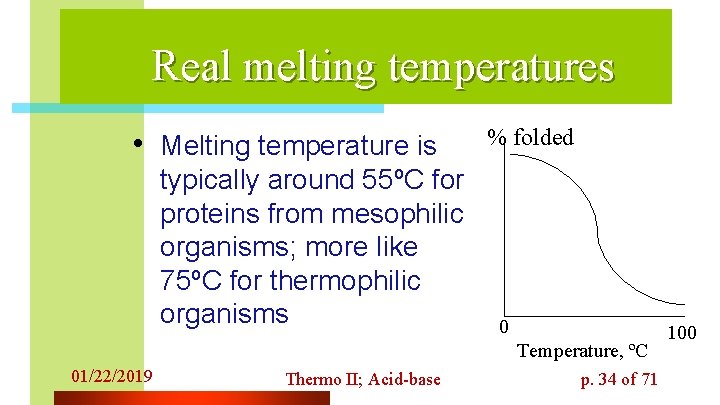
Real melting temperatures • Melting temperature is typically around 55ºC for proteins from mesophilic organisms; more like 75ºC for thermophilic organisms % folded 0 Temperature, ºC 01/22/2019 Thermo II; Acid-base p. 34 of 71 100

Let’s begin, chemically! • Amino acids are important on their own • and as building blocks We need to start somewhere: – Proteins are made up of amino acids – Free amino acids and peptides play significant roles in cells, even though their resting concentrations are low – We’ll build from small to large 01/22/2019 Thermo II; Acid-base p. 35 of 71

Will we really start with amino acids immediately? • Not quite yet. • We need to say a few things about • acid-base reactions We’ll use amino acids to exemplify those in a few minutes. 01/22/2019 Thermo II; Acid-base p. 36 of 71
![Acid-Base Equilibrium • In aqueous solution, [H+] ≠ 0, [OH-] ≠ 0. • Define: Acid-Base Equilibrium • In aqueous solution, [H+] ≠ 0, [OH-] ≠ 0. • Define:](http://slidetodoc.com/presentation_image_h/30469319ccaffc79a91bd51b24c3f5a1/image-37.jpg)
Acid-Base Equilibrium • In aqueous solution, [H+] ≠ 0, [OH-] ≠ 0. • Define: – p. H -log 10[H+] – p. OH -log 10[OH-] • Product [H+][OH-] = 10 -14 M 2 (+/-) fact • Take common (base-10) log of both sides: log 10([H+][OH-]) = log 10(10 -14) = -14 01/22/2019 Thermo II; Acid-base p. 37 of 71

Acid-base equilibrium, continued • Remember: • • • log(AB) = log(A) + log(B) So log 10([H+][OH-]) = log 10([H+]) + log 10([OH-]) = -14 So -log 10([H+]) -log 10([OH-]) = 14 So p. H + p. OH = 14 derived formula 01/22/2019 Thermo II; Acid-base p. 38 of 71

That’s true everywhere… • That equation is true in general. • • • What happens specifically at p. H = 7? There, p. H = 7, p. H + p. OH = 14, so p. OH = 14 – 7 = 7, so p. H = p. OH That’s not true at any other p. H. 01/22/2019 Thermo II; Acid-base p. 39 of 71

So what’s the equilibrium constant for this reaction? • Note that the chemical reaction is • • H 2 O H+ + OHTherefore keq = [H+][OH-] / [H 2 O] But we just said that [H+][OH-] = 10 -14 M 2 We also know that [H 2 O] = 55. 5 M (= (1000 g / L )/(18 g/mole)) keq = (10 -14 M 2) /55. 5 M = 1. 8 * 10 -16 M 01/22/2019 Thermo II; Acid-base p. 40 of 71

Keep clear in your mind… • Often students make mistakes that are easy • • 01/22/2019 to avoid if you remember a simple rule: At low p. H, there are many protons available and few hydroxide ions available At high p. H, there are few protons available and many hydroxide ions available Thermo II; Acid-base p. 41 of 71

Henderson-Hasselbalch Equation • If ionizable solutes are present, their • • • ionization will depend on p. H Assume a weak acid HA H+ + Asuch that the ionization equilibrium constant is Ka = [A-][H+] / [HA] Define p. Ka -log 10 Ka Then p. H = p. Ka + log 10([A-]/[HA]) 01/22/2019 Thermo II; Acid-base p. 42 of 71
![The Derivation is Trivial! • Ho hum: Ka = [A-][H+]/[HA] • p. Ka = The Derivation is Trivial! • Ho hum: Ka = [A-][H+]/[HA] • p. Ka =](http://slidetodoc.com/presentation_image_h/30469319ccaffc79a91bd51b24c3f5a1/image-43.jpg)
The Derivation is Trivial! • Ho hum: Ka = [A-][H+]/[HA] • p. Ka = -log 10([A-][H+]/[HA]) • • = -log 10([A-]/[HA]) - log 10([H+]) = -log 10([A-]/[HA]) + p. H Therefore p. H = p. Ka + log 10([A-]/[HA]) Often written p. H = p. Ka + log([base]/[acid]) 01/22/2019 Thermo II; Acid-base p. 43 of 71
![How do we use this? • Often we’re interested in calculating • [base]/[acid] for How do we use this? • Often we’re interested in calculating • [base]/[acid] for](http://slidetodoc.com/presentation_image_h/30469319ccaffc79a91bd51b24c3f5a1/image-44.jpg)
How do we use this? • Often we’re interested in calculating • [base]/[acid] for a dilute solute Clearly if we can calculate log 10([base]/[acid]) = p. H - p. Ka then you can determine [base]/[acid] = 10(p. H - p. Ka) 01/22/2019 Thermo II; Acid-base p. 44 of 71

At, below, above the p. Ka • If p. H = p. Ka, the concentration of the • • “acid” (more protonated) form and the “base” (less protonated) form are equal If p. H < p. Ka, the acid form predominates If p. H > p. Ka, the base form predominates 01/22/2019 Thermo II; Acid-base p. 45 of 71

What if a molecule has more than one ionizable group? • That’s okay: there will be a p. Ka • characteristic of each group’s ionization Typically the two p. Ka values will be far apart, so that at any given p. H, only at most two forms will be plentiful 01/22/2019 Thermo II; Acid-base p. 46 of 71

Is the “acid” form always positively charged? • No; it simply differs from the “base” • form by exactly one charge So they could be: (+1 and 0), or (0 and -1), or even (+2 and +1), or (-1 and -2). 01/22/2019 Thermo II; Acid-base p. 47 of 71

Using this notion • Many amino acid properties are expressed in these terms; but it’s relevant to other biological acids and bases too, like lactate and oleate 01/22/2019 Thermo II; Acid-base p. 48 of 71

Reading recommendations • If the material on ionization of weak acids isn’t pure review for you, I strongly encourage you to read sections 2. 3 -2. 5 of CF&M. in detail. We won’t go over this material in detail in class because it should be review, but you do need to know it! 01/22/2019 Thermo II; Acid-base p. 49 of 71

p. Ka values for weak acids Acid Formic acid Lactic acid H 2 PO 4 H 2 CO 3 CH 3 NH 3+ Alanine 0 01/22/2019 Base p. Ka Formate 3. 8 Lactate 3. 9 HPO 4 -2 7. 2 HCO 310. 2 CH 3 NH 2 10. 7 Alanine- 9. 9 Acid Form Acetic acid H 3 PO 4 HPO 4 -2 NH 4+ Alanine+ Thermo II; Acid-base Base Form Acetate H 2 PO 4 -3 NH 3 Alanine 0 p. Ka 4. 8 2. 2 12. 7 9. 2 2. 4 p. 50 of 71

Calculating the p. H of a solution of a weak acid • Add a weak acid to water at a molarity B, with • dissociation constant ka= [H+][A-]/[HA] Then let x = [H+]. We assume that almost all protons in the solution come from solute, not the water, so to a good approximation [A-] = x also. 01/22/2019 Thermo II; Acid-base p. 51 of 71

p. H of a solution of a weak acid, continued • • 01/22/2019 Therefore ka = x*x/(B-x) = x 2/(B-x) We know B and ka: we want to solve this for x. ka (B-x) = x 2 + kax –Bka = 0 x = {-ka ± √[ka 2 – 4(1)(-Bka)]} / 2 x = (-ka/2) + √[ka 2/4 + Bka] This is an exact result and general. Thermo II; Acid-base p. 52 of 71

Appropriate simplifications • In most cases B >> x, so the initial • • • equation is roughly ka = x 2/B Solution is then x = (Bka)1/2 Typical values: B = 0. 1 M, ka= 1. 76*10 -5 M, so x = 1. 33*10 -3 M p. H = -log 10 x = 2. 88 01/22/2019 Thermo II; Acid-base p. 53 of 71
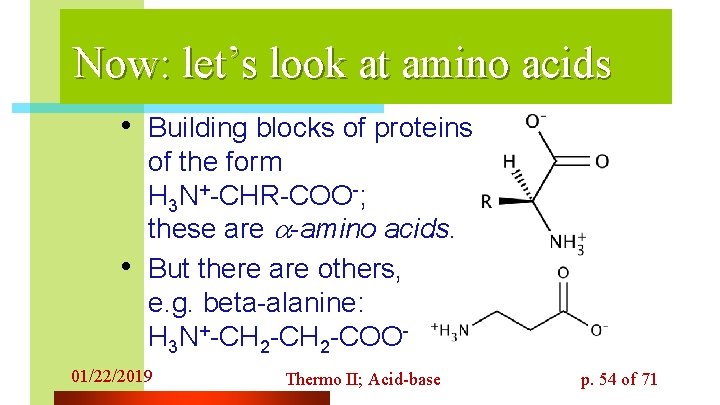
Now: let’s look at amino acids • Building blocks of proteins are • of the form H 3 N+-CHR-COO-; these are -amino acids. But there are others, e. g. beta-alanine: H 3 N+-CH 2 -COO- 01/22/2019 Thermo II; Acid-base p. 54 of 71

These are zwitterions • Over a broad range of p. H: – amino end is protonated and is + charged – carboxyl end is unprotonated and is - charged • Therefore both ends are charged • anywhere near neutral p. H Free -amino acids are therefore highly soluble, even if the side chain is apolar 01/22/2019 Thermo II; Acid-base p. 55 of 71
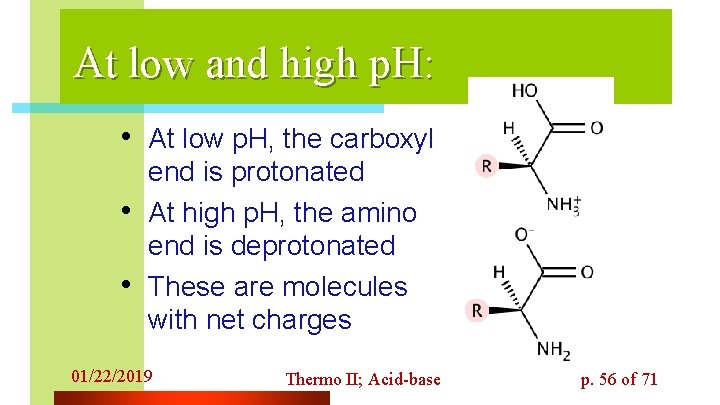
At low and high p. H: • At low p. H, the carboxyl • • end is protonated At high p. H, the amino end is deprotonated These are molecules with net charges 01/22/2019 Thermo II; Acid-base p. 56 of 71

Identities of the R groups • Nineteen of the twenty ribosomally • • • 01/22/2019 encoded amino acids fit this form Only variation is in identity of R group (side chain extending off alpha carbon) Complexity ranging from glycine (R=H) to tryptophan (R=-CH 2 -indole) Sometimes we care about non-ribosomal ornithine -amino acids—like ornithine Thermo II; Acid-base p. 57 of 71

Why “ribosomal”? • It’s my slightly pompous way of emphasizing that these 20 amino acids are actually used in the ribosome to build proteins. Other -amino acids (like ornithine and citrulline) matter, but they don’t get used in making proteins. 01/22/2019 Thermo II; Acid-base p. 58 of 71

Let’s learn the ribosomal amino acids. • We’ll walk through the list of 20, • • • one or two at a time We’ll begin with proline because it’s weird Then we’ll go through the others sequentially You do need to memorize these names and structures, both actively and passively 01/22/2019 Thermo II; Acid-base p. 59 of 71

Special case: proline • Proline isn’t an amino acid: • • it’s an imino acid Hindered rotation around bond between amine N and alpha carbon is important to its properties Tends to abolish -helicity Main-chain because of that hindered p. Ka’s: 2. 0, 10. 6 rotation 01/22/2019 Thermo II; Acid-base p. 60 of 71
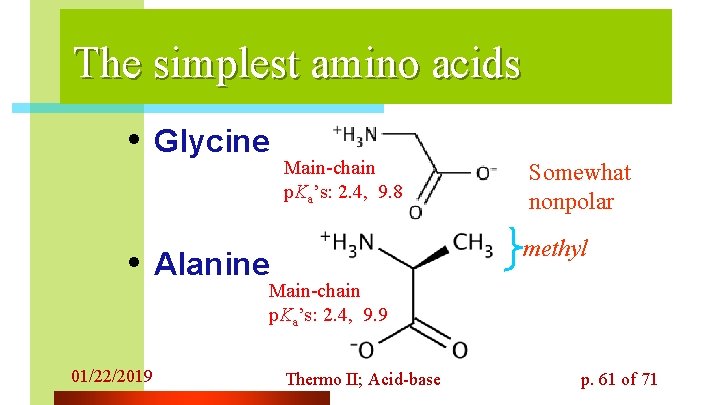
The simplest amino acids • Glycine Main-chain p. Ka’s: 2. 4, 9. 8 Somewhat nonpolar methyl • Alanine Main-chain p. Ka’s: 2. 4, 9. 9 01/22/2019 Thermo II; Acid-base p. 61 of 71
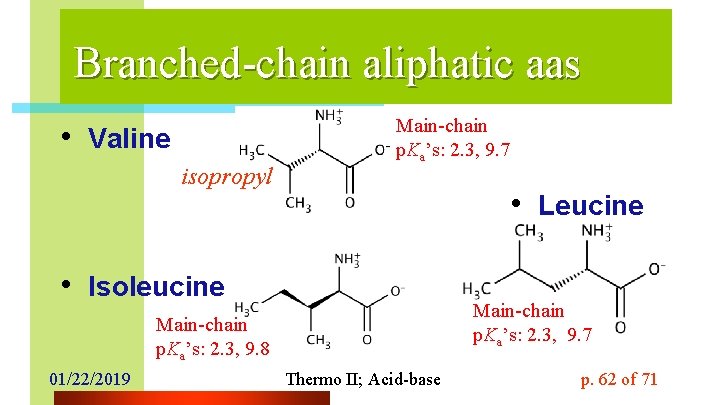
Branched-chain aliphatic aas • Valine isopropyl Main-chain p. Ka’s: 2. 3, 9. 7 • Leucine • Isoleucine Main-chain p. Ka’s: 2. 3, 9. 7 Main-chain p. Ka’s: 2. 3, 9. 8 01/22/2019 Thermo II; Acid-base p. 62 of 71
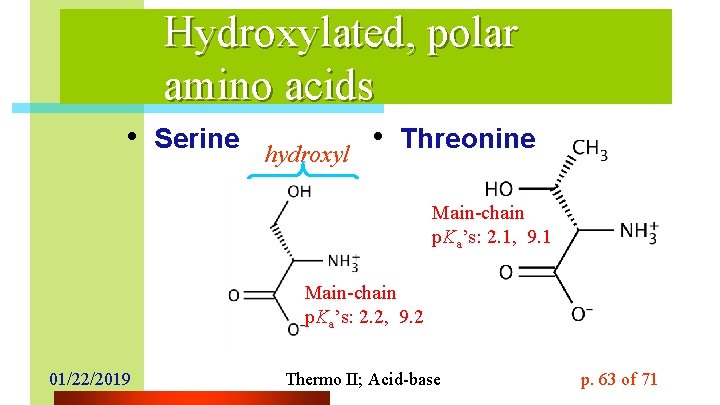
Hydroxylated, polar amino acids • Serine hydroxyl • Threonine Main-chain p. Ka’s: 2. 1, 9. 1 Main-chain p. Ka’s: 2. 2, 9. 2 01/22/2019 Thermo II; Acid-base p. 63 of 71
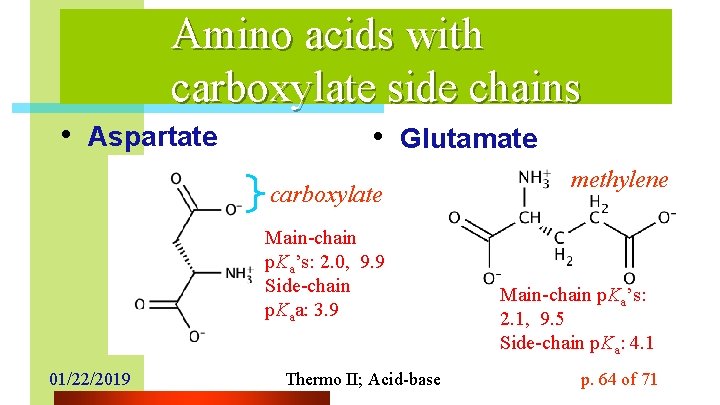
Amino acids with carboxylate side chains • Aspartate • Glutamate carboxylate Main-chain p. Ka’s: 2. 0, 9. 9 Side-chain p. Kaa: 3. 9 01/22/2019 Thermo II; Acid-base methylene Main-chain p. Ka’s: 2. 1, 9. 5 Side-chain p. Ka: 4. 1 p. 64 of 71
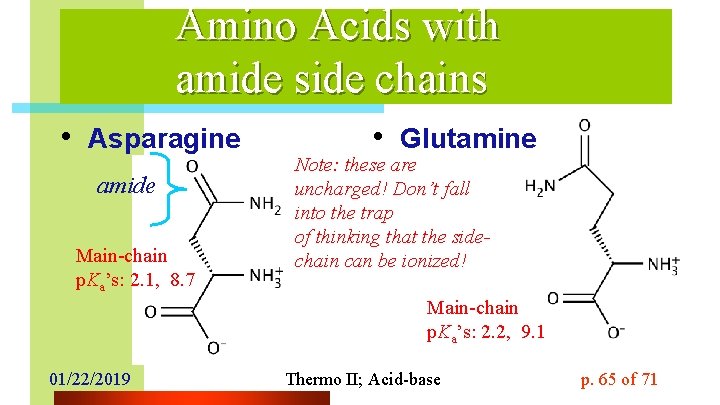
Amino Acids with amide side chains • Asparagine amide Main-chain p. Ka’s: 2. 1, 8. 7 • Glutamine Note: these are uncharged! Don’t fall into the trap of thinking that the sidechain can be ionized! Main-chain p. Ka’s: 2. 2, 9. 1 01/22/2019 Thermo II; Acid-base p. 65 of 71
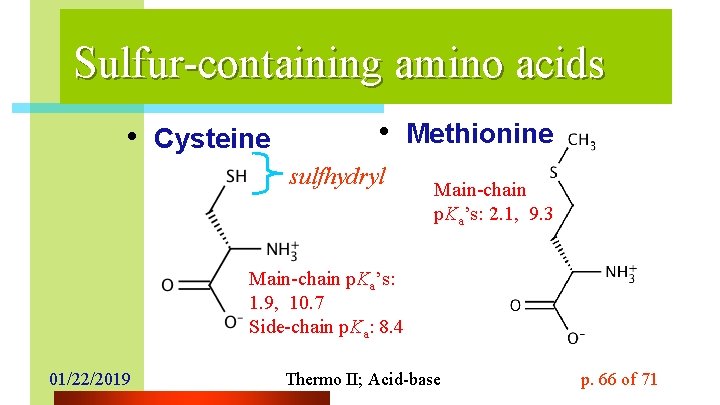
Sulfur-containing amino acids • Cysteine • Methionine sulfhydryl Main-chain p. Ka’s: 2. 1, 9. 3 Main-chain p. Ka’s: 1. 9, 10. 7 Side-chain p. Ka: 8. 4 01/22/2019 Thermo II; Acid-base p. 66 of 71
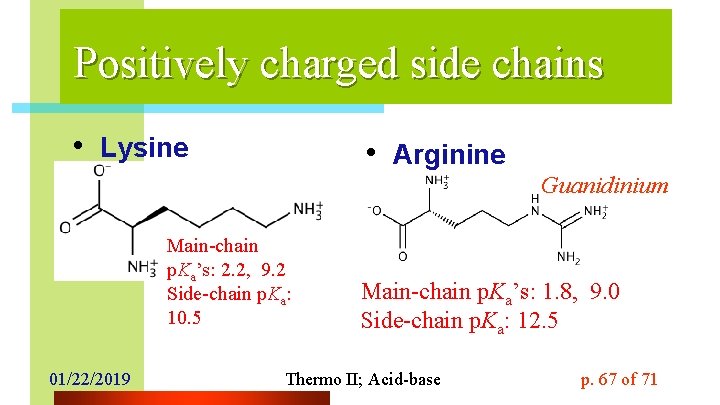
Positively charged side chains • Lysine • Arginine Guanidinium Main-chain p. Ka’s: 2. 2, 9. 2 Side-chain p. Ka: 10. 5 01/22/2019 Main-chain p. Ka’s: 1. 8, 9. 0 Side-chain p. Ka: 12. 5 Thermo II; Acid-base p. 67 of 71
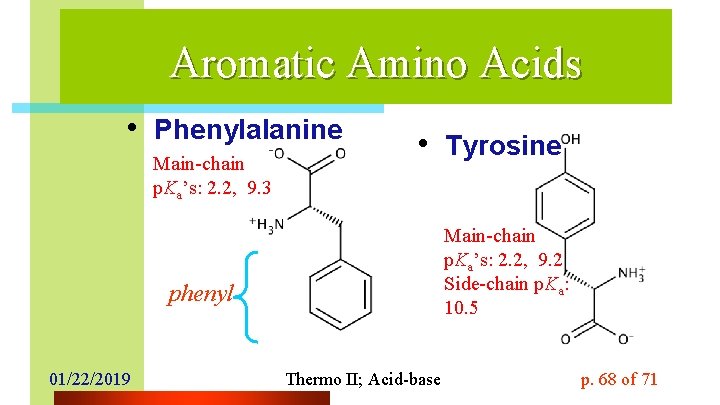
Aromatic Amino Acids • Phenylalanine Main-chain p. Ka’s: 2. 2, 9. 3 • Tyrosine Main-chain p. Ka’s: 2. 2, 9. 2 Side-chain p. Ka: 10. 5 phenyl 01/22/2019 Thermo II; Acid-base p. 68 of 71
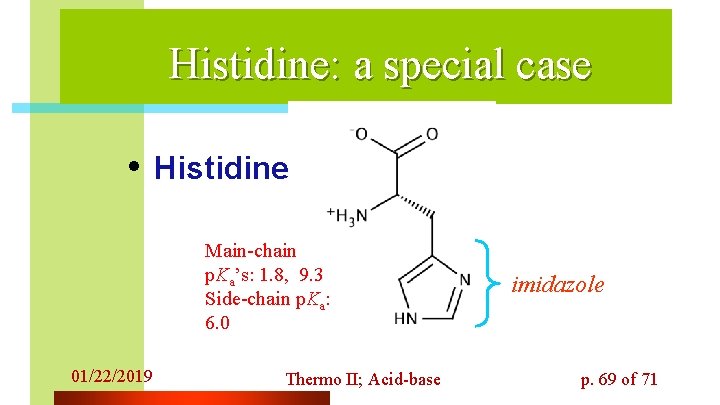
Histidine: a special case • Histidine Main-chain p. Ka’s: 1. 8, 9. 3 Side-chain p. Ka: 6. 0 01/22/2019 Thermo II; Acid-base imidazole p. 69 of 71
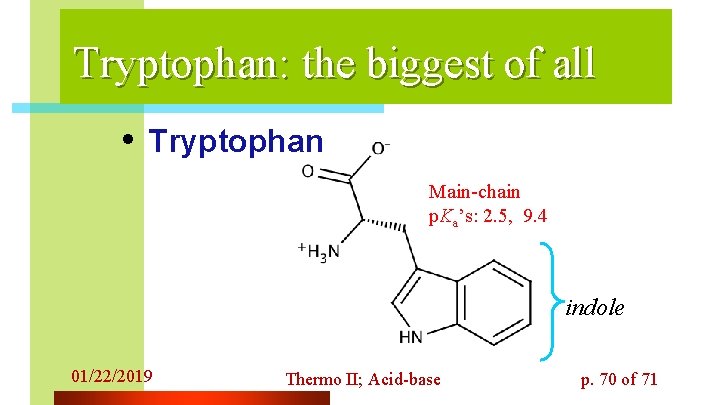
Tryptophan: the biggest of all • Tryptophan Main-chain p. Ka’s: 2. 5, 9. 4 indole 01/22/2019 Thermo II; Acid-base p. 70 of 71

Common mistakes • Thinking that asn and gln sidechains can be • • • ionized Thinking that trp’s side-chain can be ionized Misremembering the two differences between cys and met Misremembering that his is usually neutral 01/22/2019 Thermo II; Acid-base p. 71 of 71
- Slides: 71