Thermodynamics First Law of Thermodynamics Energy Conservation The









- Slides: 16

Thermodynamics

First Law of Thermodynamics Energy Conservation The change in internal energy of a system ( U) is equal to the heat flow into the system (Q) minus the work done by the system (W) U = Q - W Increase in internal energy of system Heat flow into system Work done by system P P 1 Equivalent ways of writing 1 st Law: Q = U + W P 3 1 2 3 V 1 V 2 V 07

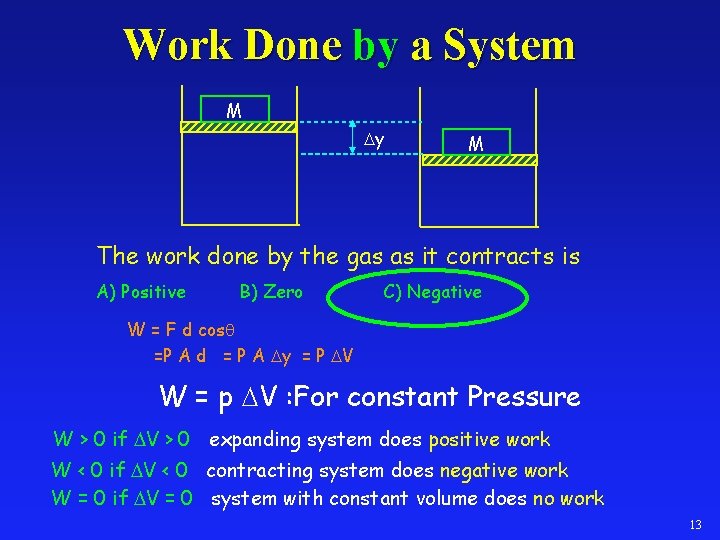
Work Done by a System M y M The work done by the gas as it contracts is A) Positive B) Zero C) Negative W = F d cosq =P A d = P A y = P V W = p V : For constant Pressure W > 0 if V > 0 expanding system does positive work W < 0 if V < 0 contracting system does negative work W = 0 if V = 0 system with constant volume does no work 13

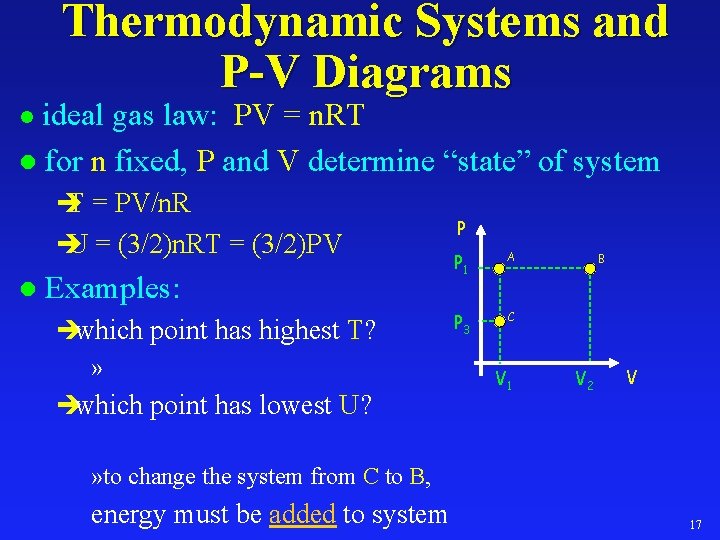
Thermodynamic Systems and P-V Diagrams ideal gas law: PV = n. RT l for n fixed, P and V determine “state” of system l è T = PV/n. R è U = (3/2)n. RT = (3/2)PV l Examples: èwhich point has highest T? » èwhich point has lowest U? P P 1 A P 3 C V 1 B V 2 V » to change the system from C to B, energy must be added to system 17

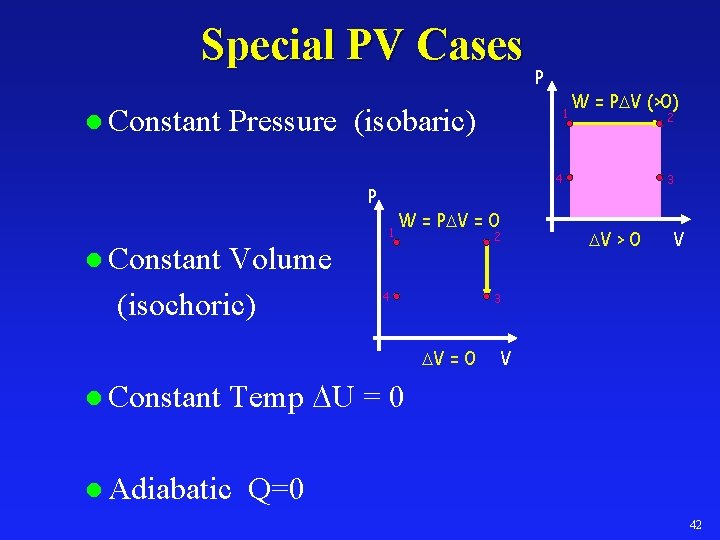
Special PV Cases l Constant Pressure (isobaric) 1 W = P V = 0 2 l Constant 4 W = P V (>0) 2 3 V > 0 V 3 V = 0 l Constant 1 4 P Volume (isochoric) P V Temp U = 0 l Adiabatic Q=0 42

First Law of Thermodynamics Isobaric Example P 2 moles of monatomic ideal gas is taken from state 1 to state 2 at constant pressure p=1000 Pa, where V 1 =2 m 3 and V 2 =3 m 3. Find T 1, T 2, U, W, Q. (R=8. 31 J/k mole) P 1 V 1 2 V 21

First Law of Thermodynamics Isochoric Example 2 moles of monatomic ideal gas is taken from state 1 to state 2 at constant volume V=2 m 3, where T 1=120 K and T 2 =180 K. Find Q. P P 2 2 P 1 1 V V 24

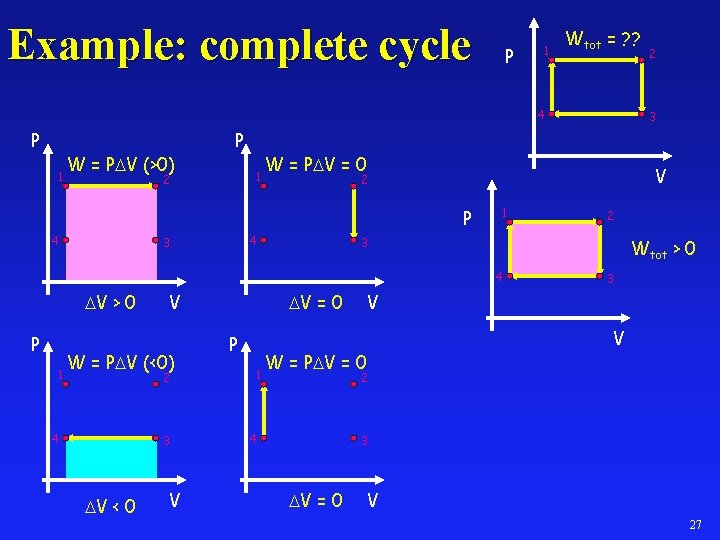
Example: complete cycle P 1 Wtot = ? ? 4 P 1 W = P V (>0) P 1 2 V 2 4 3 1 P 1 V W = P V (<0) 2 4 3 V < 0 V V = 0 P 1 2 3 Wtot > 0 4 V > 0 3 W = P V = 0 P 4 2 3 V V W = P V = 0 2 4 3 V = 0 V 27

WORK Question If we go the opposite direction for the cycle (4, 3, 2, 1) the net work done by the system will be A) Positive B) Negative P 1 4 2 If we go the other way then 3 V 30

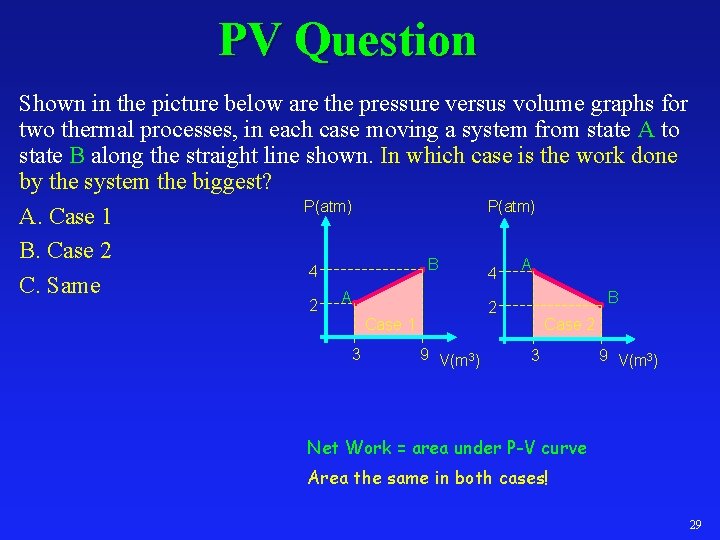
PV Question Shown in the picture below are the pressure versus volume graphs for two thermal processes, in each case moving a system from state A to state B along the straight line shown. In which case is the work done by the system the biggest? P(atm) A. Case 1 B. Case 2 A B 4 4 C. Same A B 2 2 Case 1 3 9 V(m 3) Case 2 3 9 V(m 3) Net Work = area under P-V curve Area the same in both cases! 29

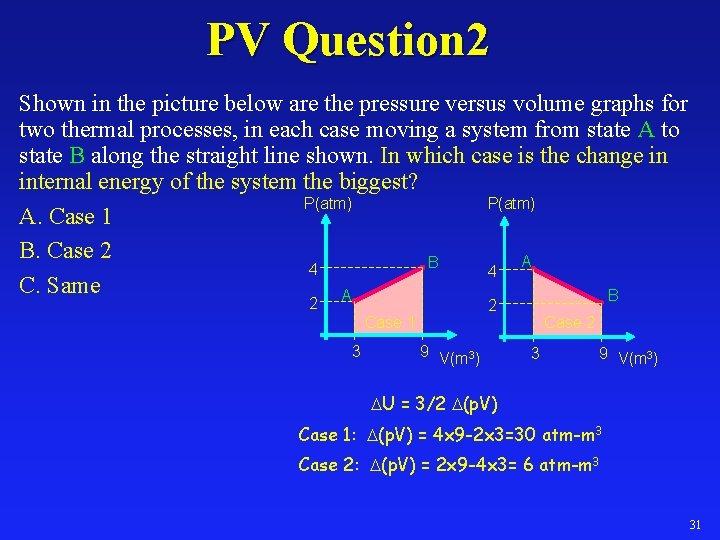
PV Question 2 Shown in the picture below are the pressure versus volume graphs for two thermal processes, in each case moving a system from state A to state B along the straight line shown. In which case is the change in internal energy of the system the biggest? P(atm) A. Case 1 B. Case 2 A B 4 4 C. Same A B 2 2 Case 1 3 9 V(m 3) Case 2 3 9 V(m 3) U = 3/2 (p. V) Case 1: (p. V) = 4 x 9 -2 x 3=30 atm-m 3 Case 2: (p. V) = 2 x 9 -4 x 3= 6 atm-m 3 31

PV Question 3 Shown in the picture below are the pressure versus volume graphs for two thermal processes, in each case moving a system from state A to state B along the straight line shown. In which case is the heat added to the system the biggest? A. Case 1 P(atm) B. Case 2 A B C. Same 4 4 2 A Case 1 Q = U + W 3 B 2 9 V(m 3) Case 2 3 9 V(m 3) W is same for both U is larger for Case 1 Therefore, Q is larger for Case 1 34

First Law Questions Q = U + W Work done by system Increase in internal energy of system Heat flow into system Some questions: P P 1 P 3 1 2 3 V 1 V 2 l Which part of cycle has largest change in internal energy, DU ? 2 3 (since U = 3/2 p. V) l Which part of cycle involves the least work W ? 3 1 (since W = p V) l What is change in internal energy for full cycle? U = 0 for closed cycle (since both p & V are back where they started) l What is net heat into system for full cycle (positive or negative)? U = 0 Q = W = area of triangle (>0) V 37

Question Consider a hypothetical device that takes 1000 J of heat from a hot reservoir at 300 K, ejects 200 J of heat to a cold reservoir at 100 K, and produces 800 J of work. Does this device violate the first law of thermodynamics ? 1. Yes 2. No W (800) = Qhot (1000) - Qcold (200) l Efficiency = W/Qhot = 800/1000 = 80% l 45

Reversible? l Most “physics” processes are reversible, you could play movie backwards and still looks fine. (drop ball vs throw ball up) l Exceptions: èNon-conservative forces (friction) èHeat Flow: » Heat never flows spontaneously from cold to hot 47

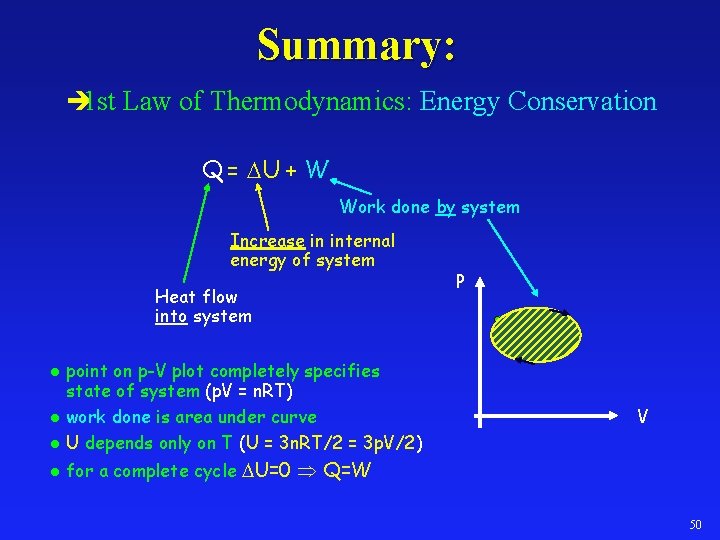
Summary: è 1 st Law of Thermodynamics: Energy Conservation Q = U + W Work done by system Increase in internal energy of system Heat flow into system point on p-V plot completely specifies state of system (p. V = n. RT) l work done is area under curve l U depends only on T (U = 3 n. RT/2 = 3 p. V/2) P l l V for a complete cycle U=0 Q=W 50