THERMODYNAMICS ENTROPY AND FREE ENERGY Thermodynamics studies the



















































- Slides: 59

THERMODYNAMICS – ENTROPY AND FREE ENERGY Thermodynamics studies the energy of a system, how much work a system could produce, and how to predict the spontaneous changes a system will undergo INTERNAL ENERGY (E) – The total energy of a system Energy is conserved during all processes 3 A-1 (of 25)

1847 HERMANN VON HELMHOLTZ Presented a mathematical argument for the Law of Conservation of Energy FIRST LAW OF THERMODYNAMICS – The change in energy of a system is equal to heat that enters the system plus the work done on the system ΔE = q + w HEAT (q) – Energy transferred causing a temperature change WORK (w) – Energy transferred causing an object to move 3 A-2 (of 25)

1847 HERMANN VON HELMHOLTZ Presented a mathematical argument for the Law of Conservation of Energy STATE FUNCTION – A property of a system whose change can be determined by only knowing its initial and final states, regardless of the pathway of the change Internal Energy is a state function, so ΔE is independent of the pathway of the change ΔE Kinetics Thermodynamics 3 A-3 (of 25)

ENTHALPY (H) – The internal energy of a system plus the pressure-volume product of a system H = E + p. V ΔH = ΔE + Δ(p. V) o ΔH = ΔE + (Δp)V + p(ΔV) At constant pressure, Δp = 0: ΔH = ΔE + p(ΔV) From the First Law: ΔE = q + w At constant pressure, w = -p(ΔV): ΔE = q + -p(ΔV) ΔE + p(ΔV) = q At constant pressure the enthalpy change of a system is equal to the heat that enters the system ΔH = qp 3 A-4 (of 25)

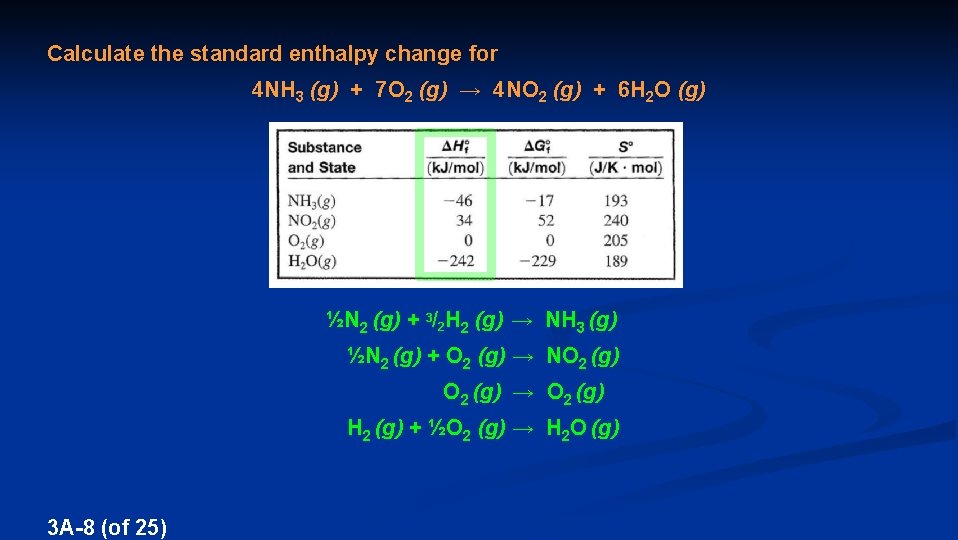
Calculate the standard enthalpy change for 4 NH 3 (g) + 7 O 2 (g) → 4 NO 2 (g) + 6 H 2 O (g) This can be done three ways: (1) Experimentally using a calorimeter (because qrxn equals ΔH) (2) Theoretically using standard enthalpies of formation (3) Theoretically using bond energies 3 A-5 (of 25)

Calculate the standard enthalpy change for 4 NH 3 (g) + 7 O 2 (g) → 4 NO 2 (g) + 6 H 2 O (g) Handout 5 of the class website (data at 298 K) Thermodynamic data is tabulated under specified conditions called the STANDARD STATE, identified with a “º”, such as ΔHº STANDARD STATE – Gaseous matter is at 1 atm, dissolved matter is 1 M, and elemental matter is in its natural state at room temperature and pressure 3 A-6 (of 25)

Calculate the standard enthalpy change for 4 NH 3 (g) + 7 O 2 (g) → 4 NO 2 (g) + 6 H 2 O (g) Handout 5 of the class website (data at 298 K) ENTHALPY OF FORMATION (ΔHf) – The enthalpy change for a formation reaction FORMATION REACTION – A reaction that forms one mole of a substance from the elements composing it 3 A-7 (of 25)

Calculate the standard enthalpy change for 4 NH 3 (g) + 7 O 2 (g) → 4 NO 2 (g) + 6 H 2 O (g) ½N 2 (g) + 3/2 H 2 (g) → NH 3 (g) ½N 2 (g) + O 2 (g) → NO 2 (g) O 2 (g) → O 2 (g) H 2 (g) + ½O 2 (g) → H 2 O (g) 3 A-8 (of 25)

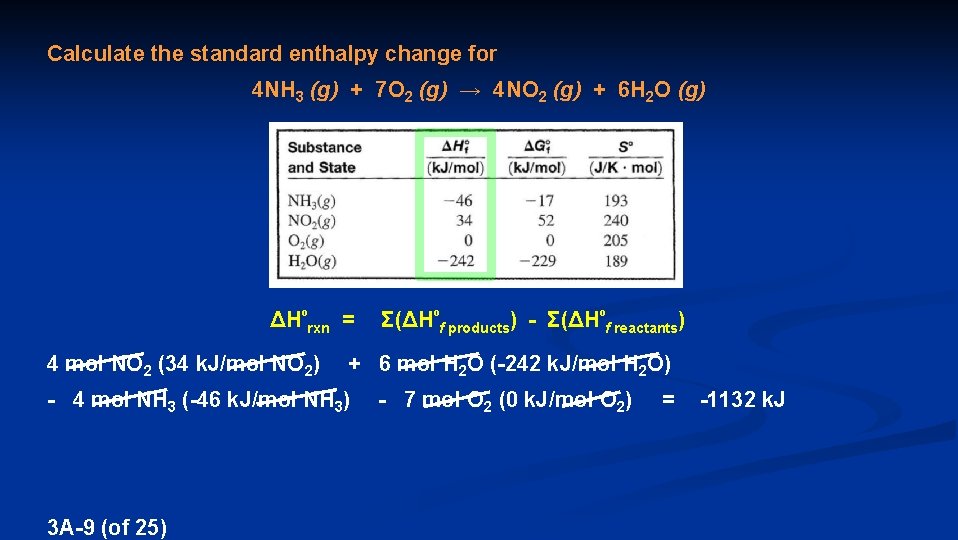
Calculate the standard enthalpy change for 4 NH 3 (g) + 7 O 2 (g) → 4 NO 2 (g) + 6 H 2 O (g) ΔHºrxn = 4 mol NO 2 (34 k. J/mol NO 2) + 6 mol H 2 O (-242 k. J/mol H 2 O) - 4 mol NH 3 (-46 k. J/mol NH 3) 3 A-9 (of 25) Σ(ΔHºf products) - Σ(ΔHºf reactants) - 7 mol O 2 (0 k. J/mol O 2) = -1132 k. J

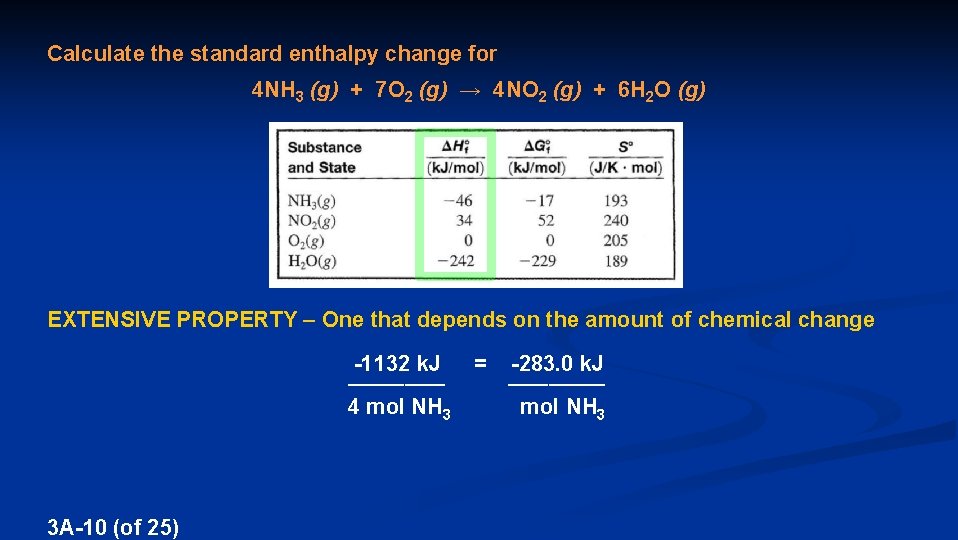
Calculate the standard enthalpy change for 4 NH 3 (g) + 7 O 2 (g) → 4 NO 2 (g) + 6 H 2 O (g) EXTENSIVE PROPERTY – One that depends on the amount of chemical change -1132 k. J = -283. 0 k. J ____________ 4 mol NH 3 mol NH 3 3 A-10 (of 25)

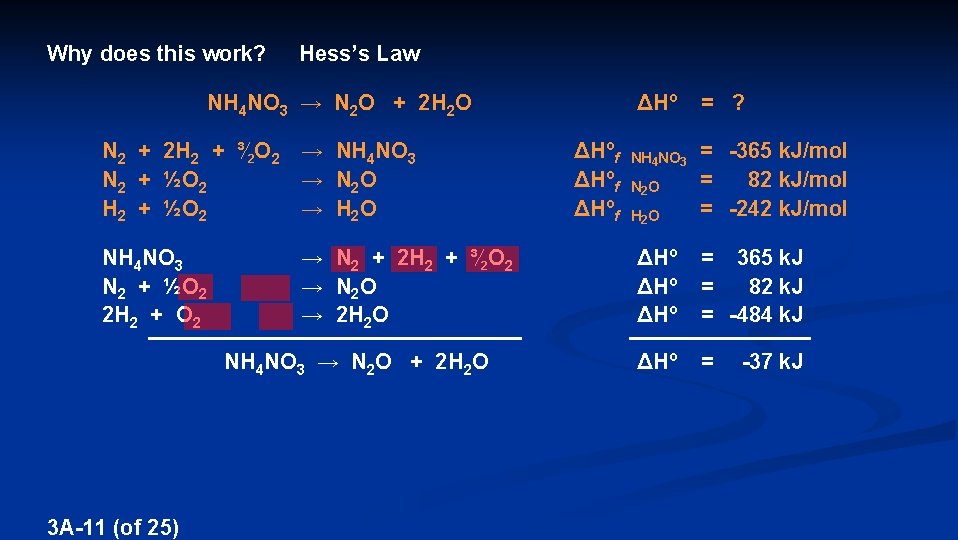
Why does this work? Hess’s Law ΔHº = ? NH 4 NO 3 → N 2 O + 2 H 2 O N 2 + 2 H 2 + ³∕ 2 O 2 → NH 4 NO 3 N 2 + ½O 2 → N 2 O H 2 + ½O 2 → H 2 O NH 4 NO 3 N 2 + ½O 2 2 H 2 + O 2 → N 2 + 2 H 2 + ³∕ 2 O 2 → N 2 O → 2 H 2 O NH 4 NO 3 → N 2 O + 2 H 2 O 3 A-11 (of 25) ΔHºf = -365 k. J/mol 82 k. J/mol N 2 O = = -242 k. J/mol H 2 O NH 4 NO 3 ΔHº = 365 k. J ΔHº = 82 k. J ΔHº = -484 k. J ΔHº = -37 k. J

Calculate the standard enthalpy change for N 2 + 2 H 2 → N 2 H 4 From Chem 1 A Handout 8: Energy must be absorbed to break the reactant bonds Energy must be released to form the product bonds : N ≡ N : H – H → H H H – N – H . . (BE’s are positive) (BE’s are negative) + 1 mol (941 k. J/mol) = 941 k. J + 2 mol (432 k. J/mol) = 864 k. J 1 mol (160 k. J/mol) = -160 k. J 4 mol (391 k. J/mol) = -1564 k. J 81 k. J 3 A-12 (of 25)

1865 RUDOLF CLAUSIUS Proposed that there is always some internal energy that cannot be converted into work ( ΔT ) He called this ENTROPY 3 A-13 (of 25)

A large amount of the internal energy of the blocks can be used for work a low amount cannot be used for work low entropy 3 A-14 (of 25) A small amount of internal energy of the blocks can be used for work a high amount cannot be used for work high entropy

ordered disordered Systems with low entropy are ordered, systems with high entropy are disordered SECOND LAW OF THERMODYNAMICS – In any spontaneous process (one that occurs naturally on its own) there is always an increase in total entropy The amount of disorder increases when a process is spontaneous the spontaneous process would be for the blocks to fall, not to stack 3 A-15 (of 25)

1890 LUDWIG BOLTZMANN Proposed a statistical meaning for entropy ENTROPY (S) – A measure of the number of arrangements available to a system in a given state 3 A-16 (of 25)

Boltzmann calculated the entropy of a system by S = k log W S = entropy k = Boltzmann Constant (R/NA, or 1. 38 x 10 -23 J/molecule. K) W = number of ways particles can be arranged in a given state while keeping the total energy constant 3 A-17 (of 25)

Calculate the entropies of the following samples of CO 2 (g) and CO 2 (s) W = 20 x 19 = 380 (1. 38 x 10 -23 J/K) log 380 = 3. 56 x 10 -23 J/K W = 2 x 1 = 2 (1. 38 x 10 -23 J/K) log 2 = 4. 15 x 10 -24 J/K For a particular type of matter, the gaseous state has a higher entropy than the solid state 3 A-18 (of 25)

FACTORS IN PREDICTING S FOR CHEMICAL SUBSTANCES 1) State of Matter Sgas > Sliquid > Ssolid More possible arrangements, higher entropy 2) Mass Slarge mass > Slow mass Ei = h 2 i 2 i = 1, 2, … ______ 8 m. X 2 3 A-19 (of 25) A larger mass means closer spacing of energy levels, so more possible arrangements

FACTORS IN PREDICTING S FOR CHEMICAL SUBSTANCES 3) complexity S molecular less elements < Smore elements Molecules with different elements allows for more possible arrangements Fa Fb Ha Hb Fa – Fb and Ha – Hb Ha – Fa and Hb – Fb or Ha – Fb and Hb – Fa Explain each difference in absolute entropy: Cl 2 (g) > Br 2 (l) Sgas is higher than Sliquid Cl 2 (g) > F 2 (g) S is higher for larger masses C 3 H 8 (g) > C 2 H 6 (g) S is higher for larger masses CO (g) > N 2 (g) S is higher for more internal complexity 3 A-20 (of 25)

THIRD LAW OF THERMODYNAMICS – The entropy of a perfect crystal at 0 K is 0 At 0 K C O C O C O C O C O C O O C C O C O C O W = 1 (1. 38 x 10 -23 J/K) log 1 = 0 J/K W = 16 (1. 38 x 10 -23 J/K) log 16 = 1. 66 x 10 -23 J/K Because of the 3 rd Law, all substances > 0 K have entropy values > 0 3 A-21 (of 25)

CALCULATING ΔS FOR CHEMICAL CHANGES Third Law Entropies of 1 mole of many substances in their standard states at 298 K (298 Sº or just Sº) are found on Handout 5 Be (s) + O 2 (g) + H 2 (g) → Be(OH)2 (s) 3 A-22 (of 25)

CALCULATING ΔS FOR CHEMICAL CHANGES Third Law Entropies of 1 mole of many substances in their standard states at 298 K (298 Sº or just Sº) are found on Handout 5 Sº values are not ΔSºf values, Sº values are the absolute entropy (calculated from the Boltzmann equation) of 1 mole of each substance All Sº values at temperatures > 0 K are positive For a chemical reaction, a 298ΔSº can be calculated from Handout 5 298ΔSº reaction = Σ 298 Sº products - Σ 298 Sº reactants Be (s) + O 2 (g) + H 2 (g) → Be(OH)2 (s) 3 A-23 (of 25) NO!!!

Calculate 298ΔSº, the standard entropy change at 298 K, for 2 Ni. S (s) + 3 O 2 (g) → 2 SO 2 (g) + 2 Ni. O (s) 2 mol SO 2 (248 J/mol. K SO 2) + 2 mol Ni. O (38 J/mol. K Ni. O) - 2 mol Ni. S (53 J/mol. K Ni. S) - 3 mol O 2 (205 J/mol. K O 2) = -149 J/K ΔS predicts whether a system is becoming more ordered or disordered This says the system is becoming more ordered 3 A-24 (of 25)

PREDICTING ΔS FOR CHEMICAL CHANGES ΔS is + for reactions that have 1) more gaseous products than reactants 2) solid reactants dissolving Ca. CO 3 (s) → Ca. O (s) + CO 2 (g) Ag+ (aq) + Cl- (aq) → Ag. Cl (s) 0 gas molecules → 1 gas molecule dissolved ions precipitate ΔS = + ΔS = – 2 SO 2 (g) + O 2 (g) → 2 SO 3 (g) H+ (aq) + HCO 3 - (aq) → H 2 O (l) + CO 2 (g) 3 gas molecules → 2 gas molecules 0 gas molecules → 1 gas molecule ΔS = – ΔS = + 3 A-25 (of 25)


SPONTANEITY SPONTANEOUS PROCESS – One that proceeds without outside intervention SECOND LAW OF THERMODYNAMICS – In any spontaneous process there is always an increase in total entropy For a spontaneous process: ΔSuniv > 0 ΔSsys + ΔSsurr = ΔSuniv HO 5 -ΔHsys _____ T 3 B-1 (of 19)

1873 JOSIAH WILLARD GIBBS Proposed a way to determine the maximum amount of internal energy that could be converted into work GIBBS FREE ENERGY (G) – The maximum amount of internal energy of a system that can be converted into work 3 B-2 (of 19)

G = H - TS ΔGsys = ΔHsys - TΔSsys -ΔGsys = -ΔHsys + ΔSsys ________ T ΔSsurr -ΔGsys = ΔSsurr + ΔSsys ____ T ΔSuniv 3 B-3 (of 19)

-ΔGsys = ΔSuniv > 0 for a spontaneous process T ____ ΔGsys < 0 for a spontaneous process The change in Gibbs Free Energy (ΔG) indicates if a process is spontaneous or nonspontaneous 3 B-4 (of 19)

ΔG = ΔH - TΔS Spontaneous processes are favored by (1) a decrease in enthalpy (processes that are exothermic ΔH negative) (2) an increase in entropy (processes that increase disorder ΔS positive) 3 B-5 (of 19)

CONTRIBUTIONS OF ΔH AND ΔS TO SPONTANEITY ΔG = ΔH - TΔS ΔH - exothermic endothermic + endothermic exothermic + - 3 B-6 (of 19) = (-) - (+) = (+) - (-) = (+) - (+) = (-) - (-) = (+) = (? ) = (-) ΔS + more disorder more order - more disorder more order + - ΔG - spontaneous nonspontaneous + spontaneous at high T spontaneous at low T ? ?

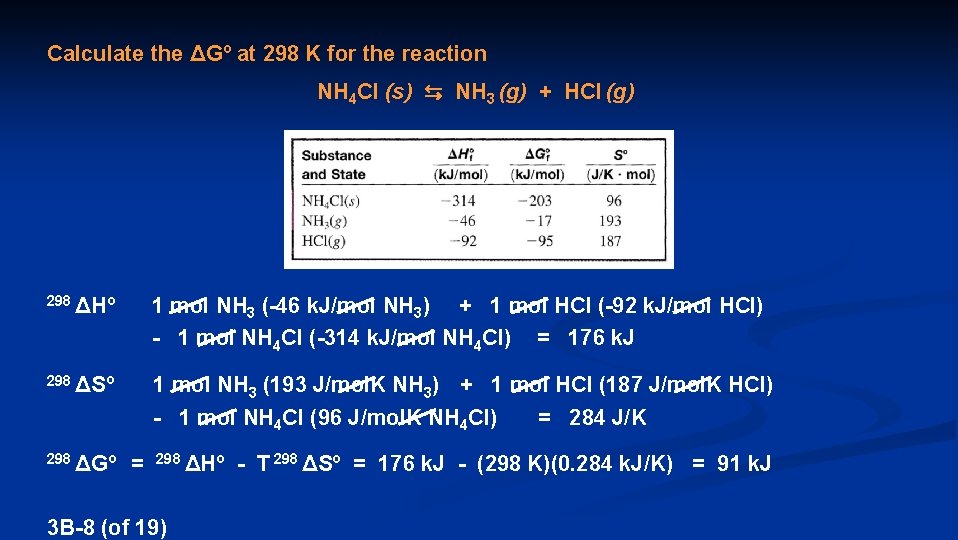
Calculate the ΔGº at 298 K for the reaction NH 4 Cl (s) ⇆ NH 3 (g) + HCl (g) 298ΔGº 1 mol NH 3 (-17 k. J/mol NH 3) + 1 mol HCl (-95 k. J/mol HCl) - 1 mol NH 4 Cl (-203 k. J/mol NH 4 Cl) = 91 k. J When starting with solid NH 4 Cl, 1 atm NH 3, and 1 atm HCl, the reaction is nonspontaneous reverse reaction is spontaneous 3 B-7 (of 19)

Calculate the ΔGº at 298 K for the reaction NH 4 Cl (s) ⇆ NH 3 (g) + HCl (g) 298 ΔHº 1 mol NH 3 (-46 k. J/mol NH 3) + 1 mol HCl (-92 k. J/mol HCl) - 1 mol NH 4 Cl (-314 k. J/mol NH 4 Cl) = 176 k. J 298 ΔSº 1 mol NH 3 (193 J/mol. K NH 3) + 1 mol HCl (187 J/mol. K HCl) - 1 mol NH 4 Cl (96 J/mol. K NH 4 Cl) = 284 J/K 298 ΔGº = 298 ΔHº - T 298 ΔSº 3 B-8 (of 19) = 176 k. J - (298 K)(0. 284 k. J/K) = 91 k. J

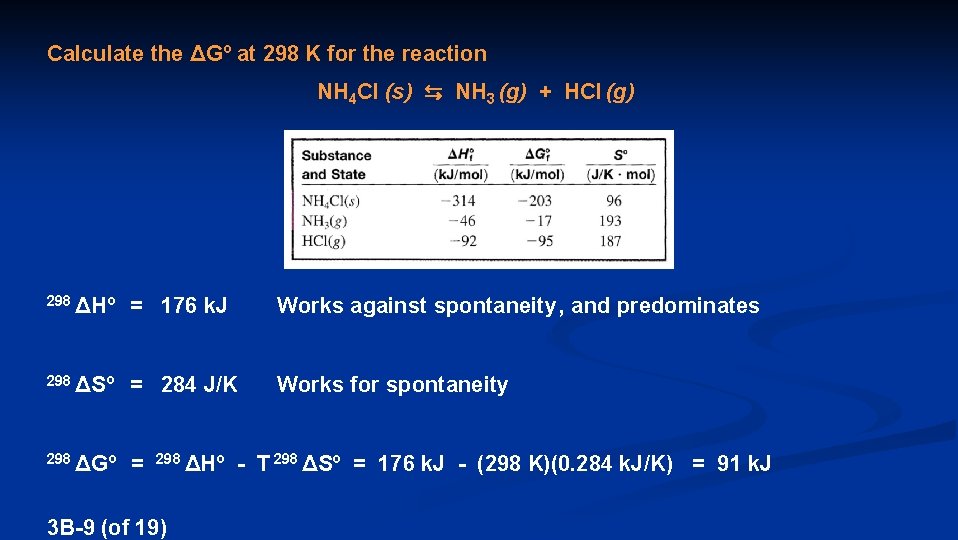
Calculate the ΔGº at 298 K for the reaction NH 4 Cl (s) ⇆ NH 3 (g) + HCl (g) 298 ΔHº = 176 k. J Works against spontaneity , and predominates 298 ΔSº = 284 J/K Works for spontaneity 298 ΔGº = 298 ΔHº - T 298 ΔSº 3 B-9 (of 19) = 176 k. J - (298 K)(0. 284 k. J/K) = 91 k. J

Calculate the ΔHº, ΔGº, and ΔSº at 298 K for the reaction 2 C 2 H 6 (g) + O 2 (g) ⇆ 2 C 2 H 5 OH (l) 298 ΔHº 2 mol C 2 H 5 OH (-278 k. J/mol C 2 H 5 OH) - 2 mol C 2 H 6 (-84. 7 k. J/mol C 2 H 6) - 1 mol O 2 (0 k. J/mol O 2) = -386. 6 k. J 298 ΔGº 2 mol C 2 H 5 OH (-175 k. J/mol C 2 H 5 OH) - 2 mol C 2 H 6 (-32. 9 k. J/mol C 2 H 6) - 1 mol O 2 (0 k. J/mol O 2) = -284. 2 k. J 3 B-10 (of 19)

Calculate the ΔHº, ΔGº, and ΔSº at 298 K for the reaction 2 C 2 H 6 (g) + O 2 (g) ⇆ 2 C 2 H 5 OH (l) 298 ΔSº 2 mol C 2 H 5 OH (161 J/mol. K C 2 H 5 OH) - 2 mol C 2 H 6 (229. 5 J/mol. K C 2 H 6) - 1 mol O 2 (0 J/mol. K O 2) = -137 J/K ΔGº = ΔHº - TΔSº ΔGº - ΔHº = ΔSº = -284. 2 k. J – (-386. 6 k. J) ______ ______________ -T -298 K 3 B-11 (of 19) = -0. 344 k. J/K

Calculate the ΔHº, ΔGº, and ΔSº at 298 K for the reaction 2 C 2 H 6 (g) + O 2 (g) ⇆ 2 C 2 H 5 OH (l) ΔH and ΔS are temperature independent, so Handout 5 can be used to calculate a ΔHº and ΔSº at any temperature However, ΔG depends on temperature, so the ΔGºf’s on Handout 5 can ONLY be used to calculate ΔGº at 298 K To calculate ΔGº at any temperature other than 298 K, use ΔHº - TΔSº 3 B-12 (of 19)

Calculate ΔHº, ΔSº, and ΔGº at 308 K for 2 SO 2 (g) + O 2 (g) → 2 SO 3 (g) 298ΔHº = 308ΔHº (constant at all temperatures) 2 mol SO 3 (-396 k. J/mol SO 3) - 2 mol SO 2 (-297 k. J/mol SO 2) - 1 mol O 2 (0 k. J/mol O 2) = -198 k. J ∴ reaction is exothermic 3 B-13 (of 19)

Calculate ΔHº, ΔSº, and ΔGº at 308 K for 2 SO 2 (g) + O 2 (g) → 2 SO 3 (g) 298ΔSº = 308ΔSº (constant at all temperatures) 2 mol SO 3 (257 J/mol. K SO 3) - 2 mol SO 2 (248 J/mol. K SO 2) - 1 mol O 2 (205 J/mol. K O 2) = -187 J/K ∴ reaction becomes more ordered 3 B-14 (of 19)

Calculate ΔHº, ΔSº, and ΔGº at 308 K for 2 SO 2 (g) + O 2 (g) → 2 SO 3 (g) 298ΔGº ≠ 308ΔGº 308 ΔGº (not constant at all temperatures) = ΔHº - TΔSº = -198 k. J - (308 K)(-0. 187 k. J/K) = -140. k. J ∴ when all reactants and products are present and start in their standard states, the forward reaction is spontaneous at 308 K 3 B-15 (of 19)

For the reaction: H 2 O (s) ⇆ H 2 O (l) Find ΔGº at 20. 0ºC Calculated from Handout 5: 293. 2ΔGº 298 ΔHº fus = 6, 038 J 298 ΔSº = 293. 2 ΔHº - T 293. 2 ΔSº = 6, 038 J - (293. 2 K)(22. 1 J/K) = -442 J reaction is spontaneous forward reaction is spontaneous H 2 O (s) melts at 20. 0ºC 3 B-16 (of 19) fus = 22. 1 J/K

For the reaction: H 2 O (s) ⇆ H 2 O (l) Find ΔGº at -20. 0ºC Calculated from Handout 5: 253. 2ΔGº 298 ΔHº fus = 6, 038 J 298 ΔSº fus = 22. 1 J/K = 253. 2 ΔHº - T 253. 2 ΔSº = 6, 038 J - (253. 2 K)(22. 1 J/K) = 442 J reaction is nonspontaneous reverse reaction is spontaneous H 2 O (l) freezes at -20. 0ºC Notice ΔGº is different at the two different temperatures: -442 J and +442 J 3 B-17 (of 19)

For the reaction: H 2 O (s) ⇆ H 2 O (l) Find ΔGº at 0. 0ºC Calculated from Handout 5: 273. 2ΔGº 298 ΔHº fus = 6, 038 J 298 ΔSº fus = 22. 1 J/K = 273. 2 ΔHº - T 273. 2 ΔSº = 6, 038 J - (273. 2 K)(22. 1 J/K) = 0 J The temperature of a phase change is when the two phases are in equilibrium For any reaction at equilibrium, ΔG = 0 If a standard state reaction is at equilibrium, ΔGº is 0 Then, if ΔHº and ΔSº are known, the temperature at which the standard state reaction is at equilibrium can be calculated 3 B-18 (of 19)

For the reaction: H 2 O (l) ⇆ H 2 O (g) Find the normal boiling point of water “Normal” means vapor pressure is 1 atm when boiling a standard state process Boiling is equilibrium between liquid and vapor, so ΔGº = 0 Calculated from Handout 5: 298 ΔHº vap = 44, 400 J TΔGº = ΔHº - TΔSº 0 = ΔHº - TeqΔSº -ΔHº = - TeqΔSº ΔHº = Teq = 44, 400 J ΔSº 119 J/K _____ 3 B-19 (of 19) ______ = 373 K = 100. ºC 298 ΔSº vap = 119 J/K


THERMODYNAMIC FUNCTIONS (1) Enthalpy (H) – The total energy plus p. V product of a system Change in Enthalpy (ΔHsys) – Indicates if a process is exothermic or endothermic at constant pressure (2) Entropy (S) – The energy of a system that cannot be converted to work, related to the number of arrangements available to a system in a given state Change in Entropy (ΔSsys) – Indicates if a process is becoming more ordered or more disordered Change in Entropy (ΔSuniv) – Indicates if a process is spontaneous or nonspontaneous (3) Gibbs Free Energy (G) – The maximum energy of a system that can be converted into work Change in Gibbs Free Energy (ΔGsys) – Indicates if a process is spontaneous or nonspontaneous 3 C-1 (of 13)

PREDICTING ΔG FOR NON-STANDARD STATE CHEMICAL REACTIONS ΔGº predicts spontaneity when reactants and products are all present in the container and in their standard state (1 M or 1 atm) ΔG predicts spontaneity when reactants and products are not in their standard state ΔG = ΔGº + RT ln Q 3 C-2 (of 13)

The standard free energy change at 298 K is -514 k. J for the reaction 2 CO (g) + O 2 (g) → 2 CO 2 (g) If a reaction vessel contains 0. 010 atm CO, 0. 020 atm O 2, and 3. 0 atm CO 2, calculate the free energy change for the reaction at 298 K ΔG = ΔGº + RT ln Q 298ΔG = 298ΔGº + RTln Q Q = p. CO 22 = (3. 0)2 p. CO 2 p. O 2 (0. 010)2(0. 020) _________________ = 4. 5 x 106 298ΔG = -514, 000 J + (8. 314 J/K)(298 K) ln 4. 5 x 106 = -476, 000 J 3 C-3 (of 13)

HIO 2 (aq) + H 2 O (l) ⇆ H 3 O+ (aq) + IO 2 - (aq) (a) Calculate the standard free energy change at 298 K for the reaction 298ΔGº = 1 mol H 3 O + (-237 k. J/mol H 3 O +) + 1 mol IO - (-23 k. J/mol IO -) 2 2 - 1 mol HIO 2 (-55 k. J/mol HIO 2) - 1 mol H 2 O (-237 k. J/mol H 2 O) = 32 k. J This means that the reverse reaction is spontaneous in a solution that is 1 M HIO 2, 1 M H 3 O+, and 1 M IO 2 - 3 C-4 (of 13)

HIO 2 (aq) + H 2 O (l) ⇆ H 3 O+ (aq) + IO 2 - (aq) (b) If a solution is 0. 500 M HIO 2, 1. 00 x 10 -7 M H 3 O+, and 0. 250 M IO 2 -, calculate the free energy change at 298 K for the reaction ΔG = ΔGº + RT ln Q 298ΔG = 298ΔGº + RTln Q Q = [H 3 O+][IO 2 -] = (1. 00 x 10 -7)(0. 250) [HIO 2] (0. 500) ___________________ = 5. 00 x 10 -8 298ΔG = 32, 000 J + (8. 314 J/K)(298 K) ln 5. 00 x 10 -8 = -10, 000 J This means that the forward reaction is spontaneous in a solution that is 0. 500 M HIO 2, 1. 00 x 10 -7 M H 3 O+, and 0. 250 M IO 2 - 3 C-5 (of 13)

THE MOST IMPORTANT REASON FOR KNOWING ΔGº For a reaction at equilibrium ΔG = 0 ΔG = ΔGº + RT ln Q 0 = ΔGº + RT ln Q what is Q at equilibrium? 0 = ΔGº + RT ln Keq -RT ln Keq = ΔGº ΔGº = -RT ln Keq Knowing ΔGº is valuable because it is related to a reaction’s equilibrium constant 3 C-6 (of 13)

4 NO (g) ⇆ 2 N 2 O (g) + O 2 (g) The 298ΔGº for the above reaction is -28. 2 k. J. Calculate Keq at 298 K. ΔGº = -RT ln Keq ΔGº = ln Keq _____ -RT e-ΔGº/RT = Keq = e-(-28, 200 J)/[(8. 314 J/K)(298 K)] = 8. 77 x 104 3 C-7 (of 13)

HF (aq) + H 2 O (l) ⇆ H 3 O+ (aq) + F- (aq) The Keq (Ka) for the above reaction is 7. 2 x 10 -4 at 298 K. Calculate 298ΔGº. ΔGº = -RT ln Keq 298ΔGº = -(8. 314 J/K)(298 K) ln (7. 2 x 10 -4) 298ΔGº = 18, 000 J 3 C-8 (of 13)

RELATIONSHIP BETWEEN ΔGº AND Keq The magnitude of the Keq determines the sign of ΔGº Keq >1 <1 ΔGº = -RT ln (>1) ΔGº = -RT ln (<1) ΔGº = -RT ln (1) ln (>1) = + ln (<1) = – ln (1) = 0 – + 0 1 1 Why? ΔGº means all chemicals start at 1 atm or 1 M if ΔGº is – it means Keq > 1 if ΔGº is + it means Keq < 1 if ΔGº is 0 it means Keq = Q = 1 If ΔGº = – , then the forward reaction must be spontaneous to get to equilibrium, so more products will be formed, making the numerator bigger, and making Keq > 1 3 C-9 (of 13)

Find 298ΔGº and Keq at 298 K for the reaction N 2 O 4 (g) ⇆ 2 NO 2 (g) 298ΔGº = 2 mol NO 2 (52 k. J/mol NO 2) - 1 mol N 2 O 4 (98 k. J/mol N 2 O 4) = 6 k. J e-ΔGº/RT = Keq = e-(6, 000 J)/[(8. 314 J/K)(298 K)] = 0. 09 3 C-10 (of 13)

Find ΔGº and Ka at 298 K for the reaction HNO 2 (aq) + H 2 O (l) ⇆ H 3 O+ (aq) + NO 2 - (aq) 298ΔGº = 1 mol H 3 O+ (-237 k. J/mol H 3 O+) + 1 mol NO 2 - (-35 k. J/mol NO 2 -) - 1 mol HNO 2 (-54 k. J/mol HNO 2) - 1 mol H 2 O (-237 k. J/mol H 2 O) = 19 k. J e-298ΔGº/RT = Keq = e-(19, 000 J)/[(8. 314 J/K)(298 K)] = 4. 7 x 10 -4 3 C-11 (of 13)

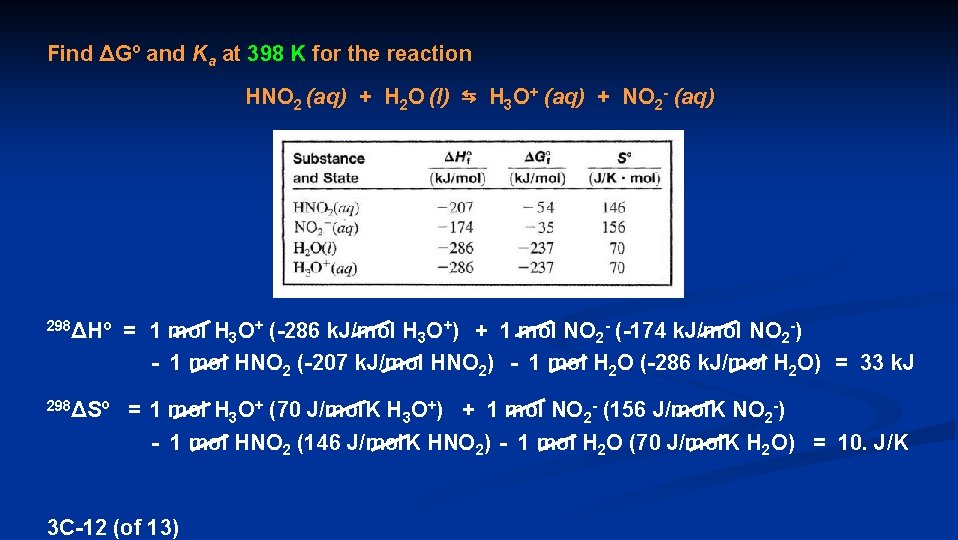
Find ΔGº and Ka at 398 K for the reaction HNO 2 (aq) + H 2 O (l) ⇆ H 3 O+ (aq) + NO 2 - (aq) 298ΔHº = 1 mol H 3 O+ (-286 k. J/mol H 3 O+) + 1 mol NO 2 - (-174 k. J/mol NO 2 -) - 1 mol HNO 2 (-207 k. J/mol HNO 2) - 1 mol H 2 O (-286 k. J/mol H 2 O) = 33 k. J 298ΔSº = 1 mol H 3 O+ (70 J/mol. K H 3 O+) + 1 mol NO 2 - (156 J/mol. K NO 2 -) - 1 mol HNO 2 (146 J/mol. K HNO 2) - 1 mol H 2 O (70 J/mol. K H 2 O) = 10. J/K 3 C-12 (of 13)

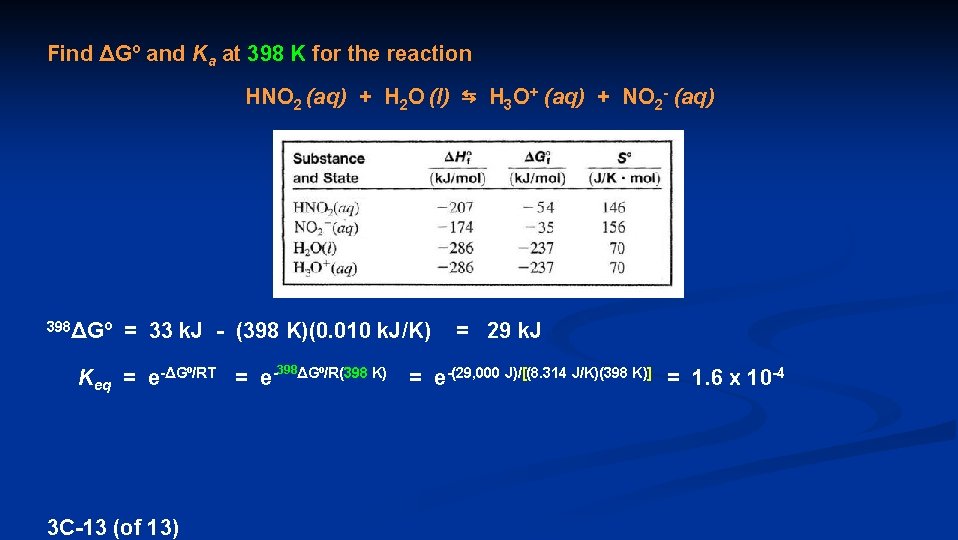
Find ΔGº and Ka at 398 K for the reaction HNO 2 (aq) + H 2 O (l) ⇆ H 3 O+ (aq) + NO 2 - (aq) 398ΔGº = 33 k. J - (398 K)(0. 010 k. J/K) = 29 k. J Keq = e-ΔGº/RT = e-398ΔGº/R(398 K) = e-(29, 000 J)/[(8. 314 J/K)(398 K)] = 1. 6 x 10 -4 3 C-13 (of 13)