Thermodynamics Energy Energy is the ability to do

Thermodynamics

Energy �Energy- is the ability to do work or produce heat. �Potential Energy- energy due to the composition or position of an object. �Kinetic Energy- is energy of motion.

Thermodynamics �Law of Conservation of Energy- states that in any chemical reaction or physical process, energy can be converted from one form to another, but it is neither created nor destroyed. �Chemical Potential Energy- the energy stored in a substance because of its composition. (like food or gasoline) �Thermochemistry- is the study of heat change that accompany chemical reaction and phase changes.

Thermodynamics �Heat – (q) is energy that is in the process of flowing from a warmer object to a cooler object. *Heat flows from ___hot___ to __cold__* �calorie- cal unit of heat. 1 calorie = 4. 184 J �Joule- J- the SI unit of heat and energy.

Thermodynamics �Temperature: A measure of the average kinetic energy of the atoms or molecules of substance �Units: Celsius, Kelvin and Fahrenheit �Thermal Energy: TOTAL kinetic energy of the particles of the substance �Units: Joules (J)

Conversions � 1000 J = 1 k. J � 1 calorie = 4. 184 J � 1 kcal= 1 Calorie = 1000 cal

Practice Problem 1 A fruit and oatmeal bar contains 142 Calories. Convert this energy to calories.

Practice Problem 2 If an endothermic process absorbs 256 J, how many kilocalories are absorbed?

Practice Problem 3 The breakfast I ate this morning contains 230 nutritional Calories. How much energy in joules will this breakfast supply?

Difference between Heat and Temperature �Temperature is the AVERAGE kinetic energy of all the particles (does NOT depend on the amount) �Units: Celsius, Fahrenheit, Kelvin �Heat (thermal) energy is the TOTAL kinetic energy of all of the particles (depends on the amount present) �Units: Joules, calories �Heat is the TRANSFER of thermal energy from hot to cold

Try it! �Compare the temperature and thermal (heat) energy of a small pot of boiling water vs. the entire Atlantic ocean!

Try it! �Compare the temperature and thermal (heat) energy of a small pot of boiling water vs. the entire Atlantic ocean! �The temperature would be higher in the pot of boiling water (higher average kinetic energy), but thermal energy would be higher for the Atlantic Ocean (so many more particles to the total KE would be greater)

Enthalpy �Enthalpy of reaction is the amount of heat gained or lost in a chemical reaction. �Enthalpy = ΔH �ΔH = Heatproducts - Heatreactants �Δ means “change in”

Endothermic / Exothermic �Endothermic – heat/energy must be added for a reaction. (ΔH = +) �Exothermic – heat/energy are released from the reaction. (ΔH = - )

Endothermic vs. Exothermic

Practice! Label the following reactions as exothermic or endothermic: H 2 + ½ O 2 H 2 O ΔH = -241. 8 k. J H 2 O H 2 + ½ O 2 ΔH = 241. 8 k. J

Practice! Rewrite these equations to include the ΔH value as a reactant or product: H 2 + ½ O 2 H 2 O ΔH = -241. 8 k. J H 2 O H 2 + ½ O 2 ΔH = 241. 8 k. J

Try these! Label as exothermic or endothermic and rewrite the equation to include the ΔH as a product or reactant. 4 Fe + 3 O 2 2 Fe 2 O 3 ∆H = -1625 k. J NH 4 + NO 3 - NH 4 NO 3 ∆H = +27 k. J

Answers! Label as exothermic or endothermic and rewrite the equation to include the ΔH as a product or reactant. 4 Fe + 3 O 2 2 Fe 2 O 3 ∆H = -1625 k. J exothermic 4 Fe + 3 O 2 2 Fe 2 O 3 + 1625 k. J NH 4 + NO 3 - NH 4 NO 3 ∆H = +27 k. J endothermic NH 4 + NO 3 - + 27 k. J NH 4 NO 3

Changes of State � Heat of vaporization (ΔHvap) heat required to vaporize (boil) one gram of a liquid �Heat of fusion (ΔHfus) heat required to melt one gram of a solid substance

Equations for change in state q = mΔHf q = mΔHv q = heat (J) m= mass (g) ΔHf = heat of fusion (J/g); found on reference table ΔHv = heat of vaporization (J/g); found on reference table *These formulas are in your reference tables on the formula page!

During a phase change, temperature remains constant because kinetic energy remains constant as potential energy increases or decreases.

Practice Problem 1 Calculate the heat required to melt 25. 7 g of solid water at its melting point.

Practice Problem 2 How much heat is evolved when 275 g of water vapor condenses to a liquid at its boiling point?

Specific Heat amount of heat required to raise the temperature of one gram of a substance by one degree Celsius • You can find specific heat values in the reference table

Specific Heat-the formula! Specific Heat J/g °C q= mcΔt Amount of Heat (J) Mass (g) Change in Temperature (K or ° C)

Try it! �Which has a higher specific heat? �Water or sand �Metal pan or oven mitts

Try it! �Which has a higher specific heat? �Water or sand �Metal pan or oven mitts Why? The more difficult it is to heat something up, the higher the specific heat because more heat is required to raise the temperature of that substance

Practice Problem 1 How much heat is lost when 4110 g of aluminum cools from 660°C to 25. 0°C?

Practice Problem 2 How much heat is required to increase the temperature of 124 g of water from 17. 5°C to 45. 8°C?
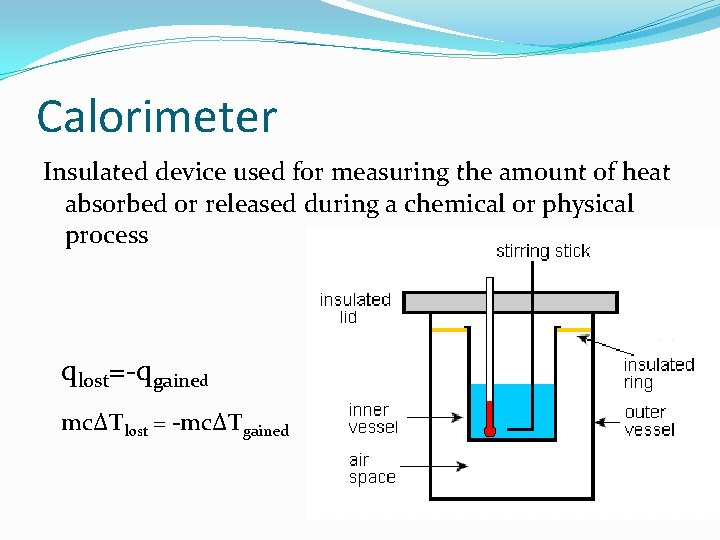
Calorimeter Insulated device used for measuring the amount of heat absorbed or released during a chemical or physical process qlost=-qgained mc∆Tlost = -mc∆Tgained

Example A 20. 0 g piece of metal at a temperature of 90. 0˚C is dropped into an insulated container holding 125 g of water at 20. 0˚C. If the final temperature is 23. 0˚C, what is the specific heat of the metal?
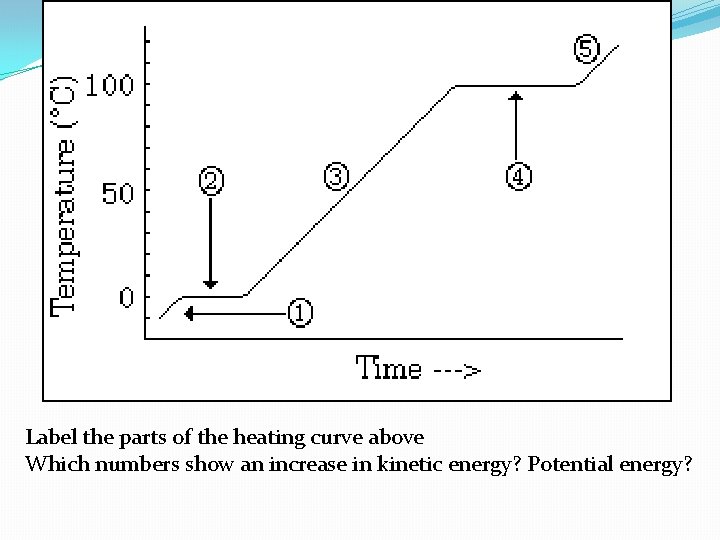
Label the parts of the heating curve above Which numbers show an increase in kinetic energy? Potential energy?

Review! Exothermic or endothermic? a) Condensation b)Melting c) Freezing d)Boiling e) Sublimation (s g) f) ΔH = -13. 6 J g) A + B + 159. 8 J C + D

Review! – Answers! Exothermic or endothermic? a) Condensation exothermic b)Melting endothermic c) Freezing exothermic d)Boiling endothermic e) Sublimation (s g) endothermic f) ΔH = -13. 6 J exothermic g) A + B + 159. 8 J C + D endothermic

Quiz Topics �Heat calculations (phase and temperature changes) �Energy conversions �Heat vocab �Exothermic vs. Endothermic �Heating Curves �Calorimeter (concepts only, NO calculations)

Entropy �Entropy – is the measure of disorder or randomness of the particles that make up a system. �Variable: S �Unit: Joules (J) �The Law of Disorder –states that spontaneous processes always proceed in such a way that the entropy of the universe increases. (The second law of Thermodynamics)

Entropy From each group, select the part with the highest entropy: a. Increasing temperature or decreasing temperature of a substance b. Reactants or products: 2 NH 3(g) 3 H 2(g) + N 2(g) c. ice or water or steam

Entropy �The states of matter from lowest entropy to highest entropy: �(s) (l) (aq) (g) �When S is positive that means that entropy is increasing �When S is negative that means that entropy is decreasing

Entropy Practice �Predict the sign of ∆Ssystem for each of the following changes. �CIF(g) + F 2(g) CIF 3(g) �NH 3(g) NH 3(aq) �CH 3 OH(l) CH 3 OH(aq) �C 10 H 8(l) C 10 H 8(s)

Kinetics and Equilibrium

Collision Theory 1. Reacting substances (atoms/ions/molecules) must collide. 2. Reacting substances must collide with sufficient energy to form activated complex. (Activation Energy = Ea)

Collision theory 3. Reacting substance must collide with the correct orientation.

Factors that affect reaction rates �Nature of reactants �More reactive the substance, the faster the reaction �Concentration �When increased, the rate increases (more collisions) �Surface area �More surface area quicker reaction �Temperature �Usually increasing temperature increases rate �Catalysts �Reaction takes a different path and lowers activation energy

Potential Energy Diagram
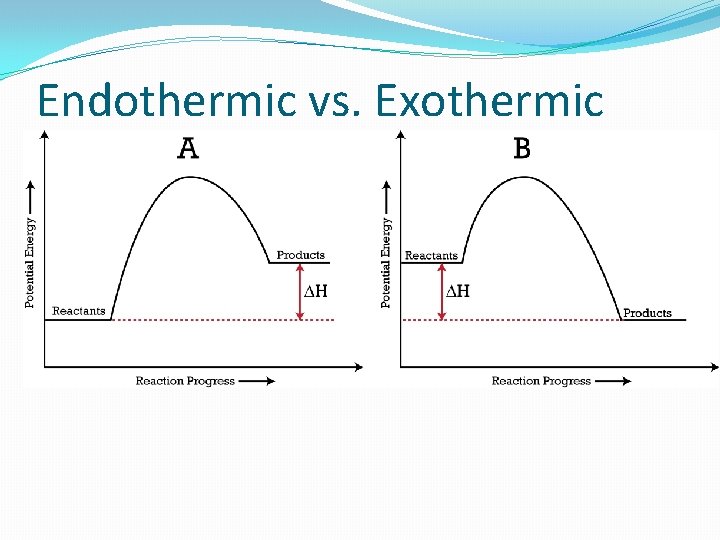
Endothermic vs. Exothermic
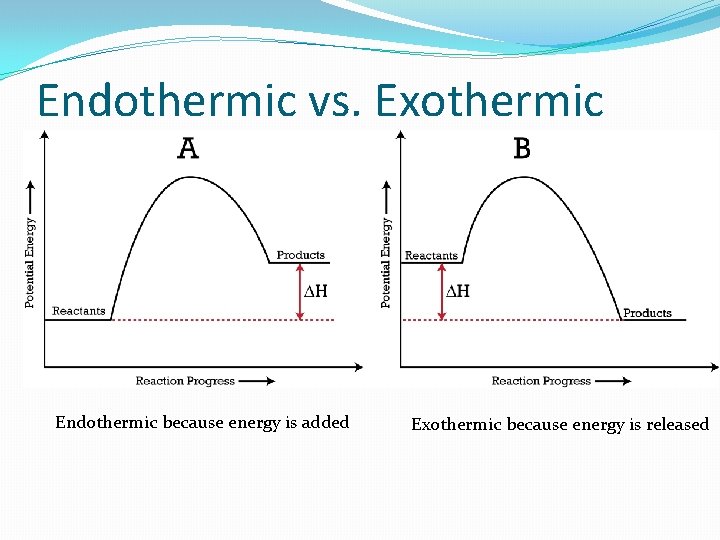
Endothermic vs. Exothermic Endothermic because energy is added Exothermic because energy is released

Catalyst �A substance that increases the rate of a chemical reaction without itself being consumed in the reaction. An example of a catalyst is an enzyme

Equilibrium �Reversible Reaction is one that can occur in both forward and reverse directions. �Chemical equilibrium is a state in which the forward and reverse reactions balance each other because they take place at equal rates. �Rateforward reaction = Ratereverse reaction

Equilibrium �Equilibrium is DYNAMIC, meaning that Equilibrium is a state of action. The reaction is constantly shifting and moving to maintain equilibrium even though we can not observe this.

Equilibrium �Law of chemical equilibrium states that at a given temperature, a chemical system may reach a state in which a particular ratio of reactant to product concentrations has a constant value. �Equilibrium constant (Keq) is the numerical value of the ratio of products concentrations to reactant concentrations, with each concentration raised to the power of its coefficients in the balanced equation.

Generic reaction and formula �a. A + b. B ↔ c. C + d. D �Where A and B are the reactants, C and D are the products, and a, b, c, and d are the coefficients in a balanced equation. �Keq= [C]c[D]d [A]a[B]b Do not put SOLIDS and LIQUIDS in the keq equation!!!!!

Equilibrium �Keq > 1 : More products than reactants at equilibrium. �Keq < 1 : More reactants than products at equilibrium.

Equilibrium �Find the Equilibrium Expression for the following: 1. N 2 O 4(g) ↔ 2 NO 2(g) 2. CO(g) + 3 H 2(g) ↔ CH 4(g) + H 2 O(g) 3. Ca(OH)2(s) + H 2 O(l) Ca 2+(aq) + 2 OH-(aq) 4. Ca. CO 3(s) ↔ Ca. O(s) + CO 2(g) 5. 2 H 2(g) + O 2(g) 2 H 2 O (l)

Le Chatelier

Le Chatelier’s Principle: �A principle stating that if a stress is applied to a system in equilibrium, the equilibrium will shift to alleviate the effect of the stress.

A “stress” can be: �Change in concentration of product or reactant �Change in temperature �Change in pressure (just gases)

Changing Concentration �Equilibrium will shift AWAY from an increase in concentration and TOWARD a decrease in concentration.

This reaction is at equilibrium N 2 H 2 NH 3 heat What happens to the reaction if you increase the concentration of Nitrogen?

N 2 Increase nitrogen concentration NH 3 H 2 heat 1. How does the reaction need to shift to make it go back to equilibrium? Right 2. What will happen to all of the substances as a result? H 2 will decrease, NH 3 will increase
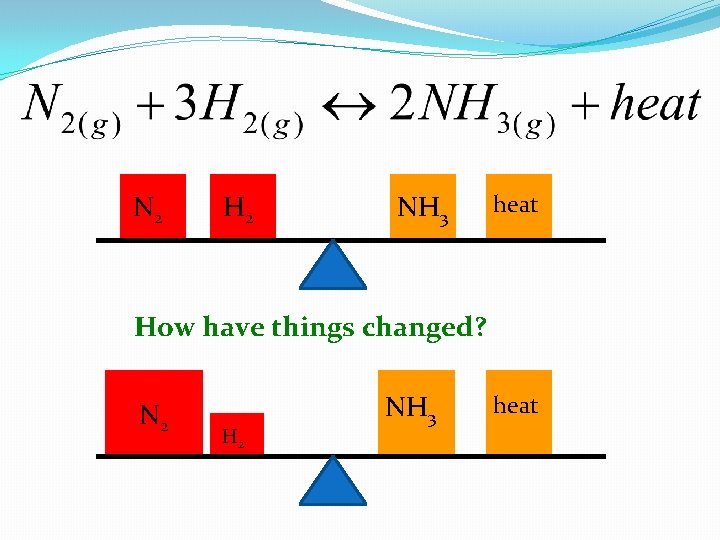
N 2 H 2 NH 3 heat How have things changed? N 2 H 2 NH 3 heat

Example 2 � 2 SO 3(g) ↔ 2 SO 2(g) + O 2(g) �Describe what happens when: �You decrease SO 2 �You increase O 2

https: //www. youtube. com/watch? v=7 zu. UV 455 z. Fs

Changing Temperature �If endothermic, heat is added to the product side �If exothermic, heat is added to the reactant side �Keq (equilbrium constant): is only affected by a temperature change. Concentration and pressure do NOT change Keq �Treat heat the same way you do concentration changes. If temperature increases, heat increases!
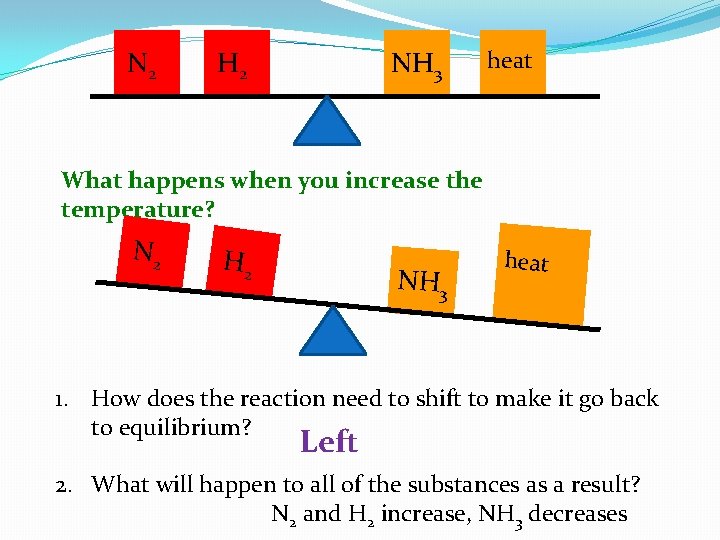
N 2 H 2 NH 3 heat What happens when you increase the temperature? N 2 H 2 NH 3 heat 1. How does the reaction need to shift to make it go back to equilibrium? Left 2. What will happen to all of the substances as a result? N 2 and H 2 increase, NH 3 decreases

N 2 H 2 NH 3 heat How have things changed? N 2 H 2 heat NH 3

Try it 2 SO 3(g) + heat ↔ 2 SO 2(g) + O 2(g) a) Increase the temperature b) Decrease the temperature

Changing Pressure �Only affects equilibrium reactions with gaseous substances �Think of your reaction in a closed container. �When you increase pressure (decrease volume) you want to have less gas in your system �When you decrease pressure (increase volume) you want to produce more gas in your system

Changing Pressure Rule Thumb �When you INCREASE pressure: always shift toward fewer moles of gas! �When you DECREASE pressure: always shift toward more moles of gas!
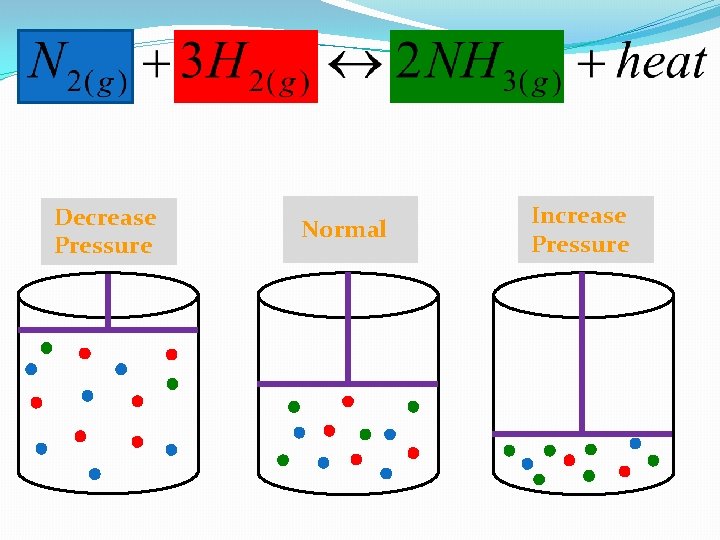
Decrease Pressure Normal Increase Pressure

Try It! Ex 1: N 2(g) + 3 H 2(g) 2 NH 3(g) a) Increase the pressure b) Decrease the pressure �Ex 2: 2 SO 3(g) ↔ 2 SO 2(g) + O 2(g) a) Increase the pressure b) Decrease the pressure
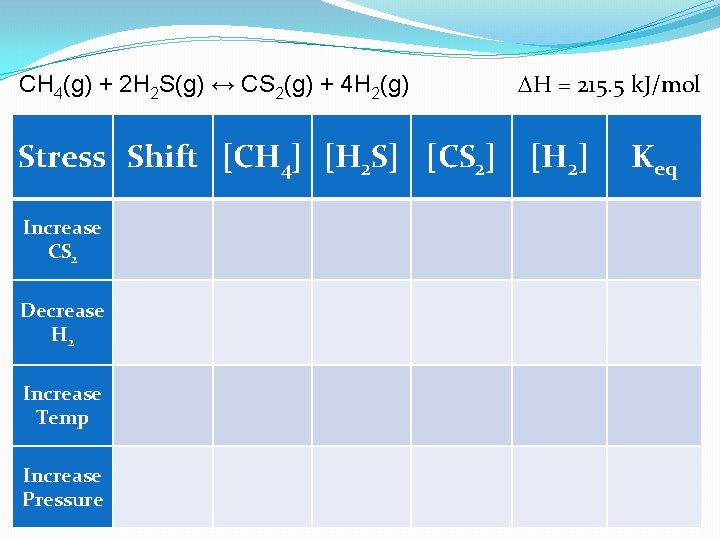
H = 215. 5 k. J/mol CH 4(g) + 2 H 2 S(g) ↔ CS 2(g) + 4 H 2(g) Stress Shift [CH 4] [H 2 S] [CS 2] [H 2] Keq Increase CS 2 Decrease H 2 Increase Temp Increase Pressure

H = 215. 5 k. J/mol CH 4(g) + 2 H 2 S(g) ↔ CS 2(g) + 4 H 2(g) Stress Shift [CH 4] [H 2 S] [CS 2] [H 2] Keq Increase CS 2 Left Increase ----- Decrease No Change Decrease H 2 Right Decrease Increase ---- No Change Increase Temp Right Decrease Increase Changes! Increase Pressure Left Increase Decrease Increase No Change

REVIEW QUESTIONS!

Practice! �How much heat is required to heat up 22 g of gold from 15˚C to 79˚C?

Answer! �

Practice! �How much heat is released to freeze 179 g of water?

Answer! �

Practice! �How much heat is released to decrease the temperature of 13. 6 g water by 13. 2˚C?

Answer! �

Practice! �How much heat is required to boil 58 g of water?
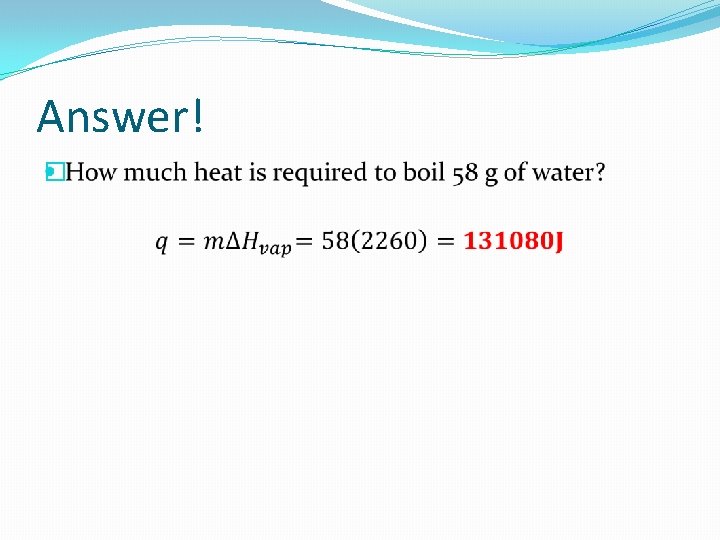
Answer! �

Practice! �Exothermic or Endothermic? �Boiling �Freezing �ΔH = - 25. 6 k. J �ΔH = + 312 k. J �C 2 H 5 OH + 3 O 2 2 CO 2 + 3 H 2 O + 1235 k. J � 2 Na. HCO 3 + 129 k. J Na 2 CO 3 + H 2 O +CO 2

Answer! �Exothermic or Endothermic? �Boiling Endothermic �Freezing Exothermic �ΔH = - 25. 6 k. J Exothermic �ΔH = + 312 k. J Endothermic �C 2 H 5 OH + 3 O 2 2 CO 2 + 3 H 2 O + 1235 k. J Exothermic � 2 Na. HCO 3 + 129 k. J Na 2 CO 3 + H 2 O +CO 2 Endothermic

Practice! �Write the equilibrium expression for this reaction: �Ca(OH)2(s) Ca 2+(aq) + 2 OH-(aq)

Answer! �Write the equilibrium expression for this reaction: �Ca(OH)2(s) Ca 2+(aq) + 2 OH-(aq) Keq = [Ca 2+][OH-]2

Practice! �Write the equilibrium expression for this reaction: � 2 SO 2(g) + O 2(g) 2 SO 3(g)

Answer! �

Practice! �What happens when SO 2 increases? � 2 SO 2(g) + O 2(g) 2 SO 3(g)

Answer! �What happens when SO 2 increases? � 2 SO 2(g) + O 2(g) 2 SO 3(g) Shift is to the right O 2 decreases SO 3 increases

Practice! �What happens when SO 3 increases? � 2 SO 2(g) + O 2(g) 2 SO 3(g)

Answer! �What happens when SO 3 increases? � 2 SO 2(g) + O 2(g) 2 SO 3(g) Shift is to the left SO 2 and O 2 increase

Practice! �What happens when pressure increases? � 2 SO 2(g) + O 2(g) 2 SO 3(g)

Answer! �What happens when pressure increases? � 2 SO 2(g) + O 2(g) 2 SO 3(g) Shift is to the right SO 2 and O 2 decrease SO 3 increase

Problem! Draw an exothermic potential energy diagram and label the energy of the products, energy of the reactants, energy of the activated complex, activation energy (of the forward and reverse reactions), and enthalpy.
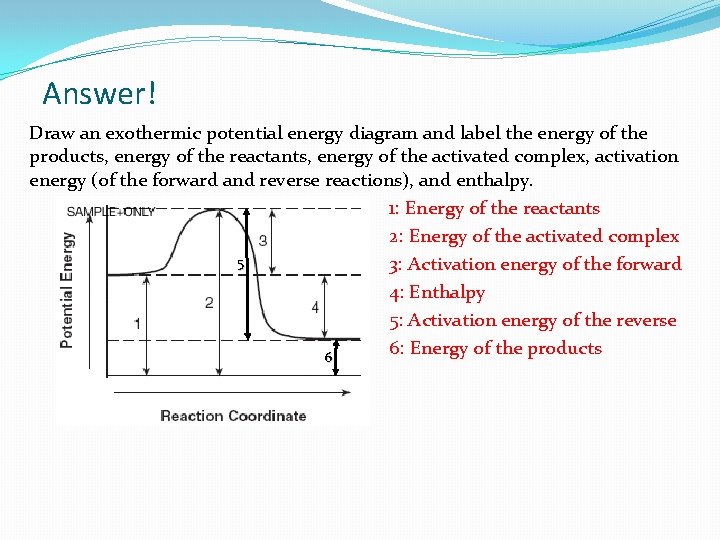
Answer! Draw an exothermic potential energy diagram and label the energy of the products, energy of the reactants, energy of the activated complex, activation energy (of the forward and reverse reactions), and enthalpy. 1: Energy of the reactants 2: Energy of the activated complex 5 3: Activation energy of the forward 4: Enthalpy 5: Activation energy of the reverse 6: Energy of the products 6
- Slides: 96