Thermodynamics and Statistical Mechanics Transport Processes Thermo Stat













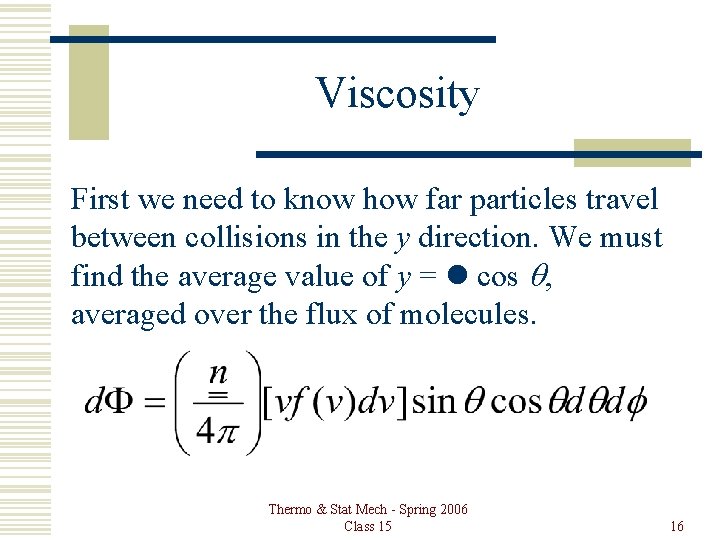
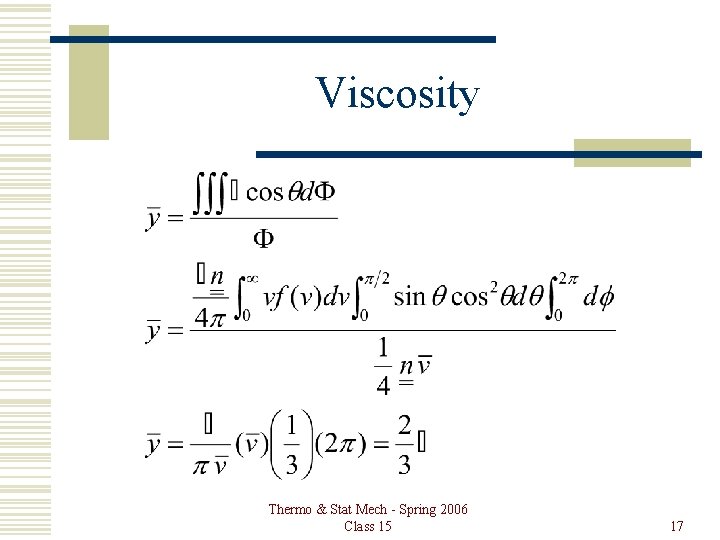
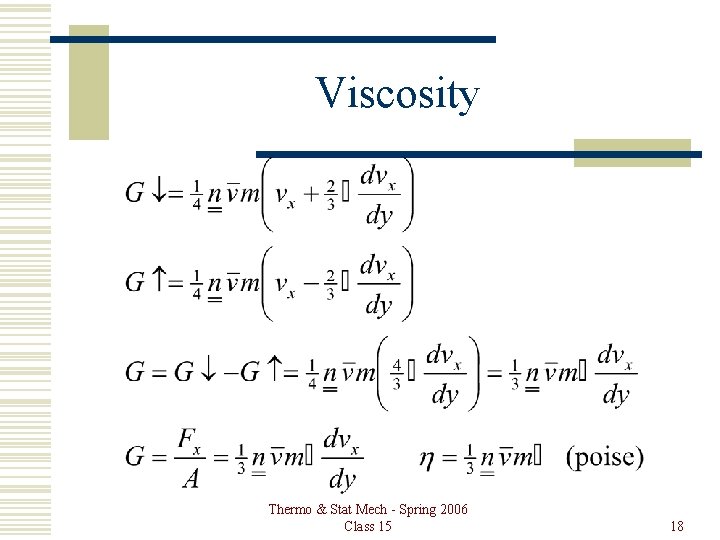





- Slides: 23

Thermodynamics and Statistical Mechanics Transport Processes Thermo & Stat Mech Spring 2006 Class 15 1

Mean Free Path The average distance that a particles travels between collisions is called the mean free path, l. In order to have a collision, the centers of two particles of radius R must approach to within a distance of 2 R = d. The picture on the next slide illustrates this point. Thermo & Stat Mech - Spring 2006 Class 15 2

Collision Cross Section s = p d 2 Thermo & Stat Mech - Spring 2006 Class 15 3

Mean Free Path Thermo & Stat Mech - Spring 2006 Class 15 4

Mean Free Paths(? ) Thermo & Stat Mech - Spring 2006 Class 15 5

Mean Free Path Thermo & Stat Mech - Spring 2006 Class 15 6

Some Molecular Speeds Thermo & Stat Mech - Spring 2006 Class 15 7

Effusion If there is a hole in a container of gas, molecules will escape through it. The rate at which they pass through the hole is equal to the flux of particles hitting the hole times the area of the hole, FA. Then the rate of change of the number of molecules in the container is: Thermo & Stat Mech - Spring 2006 Class 15 8

Effusion Limit If the hole is small equilibrium conditions prevail, and F is known. Thermo & Stat Mech - Spring 2006 Class 15 9

Transport Processes 1. Molecular diffusion. The movement of the particles is of interest. This is particle transport. 2. Thermal conductivity. Particles carry energy from a high temperature region to a lower temperature region. This is energy transport. 3. Viscosity. Drag is created when particles move to a region which is moving at a different speed. This is momentum transport. Thermo & Stat Mech - Spring 2006 Class 15 10

Transport Processes Thermo & Stat Mech - Spring 2006 Class 15 11

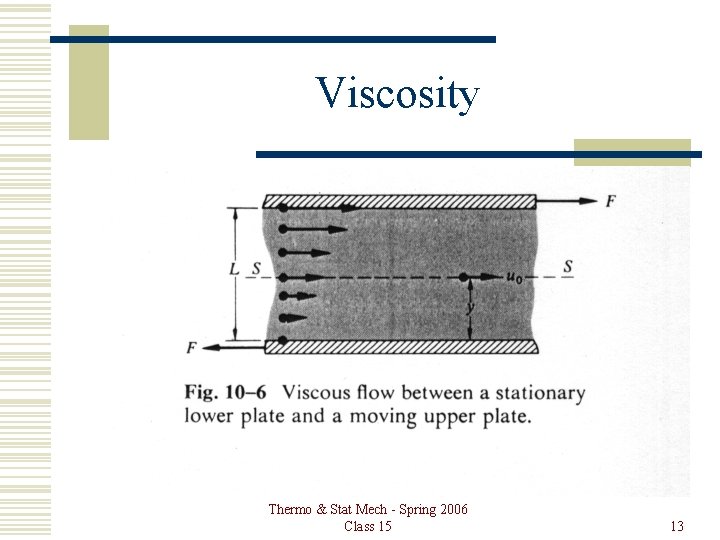
Viscosity Consider a plate moving parallel to another plate with a layer of gas between them. The gas in contact with either plate is at rest relative to the plate, so there is a velocity gradient in the gas. A force F is applied to each plate to maintain the motion, and the area of the upper plate is A. Thermo & Stat Mech - Spring 2006 Class 15 12

Viscosity Thermo & Stat Mech - Spring 2006 Class 15 13

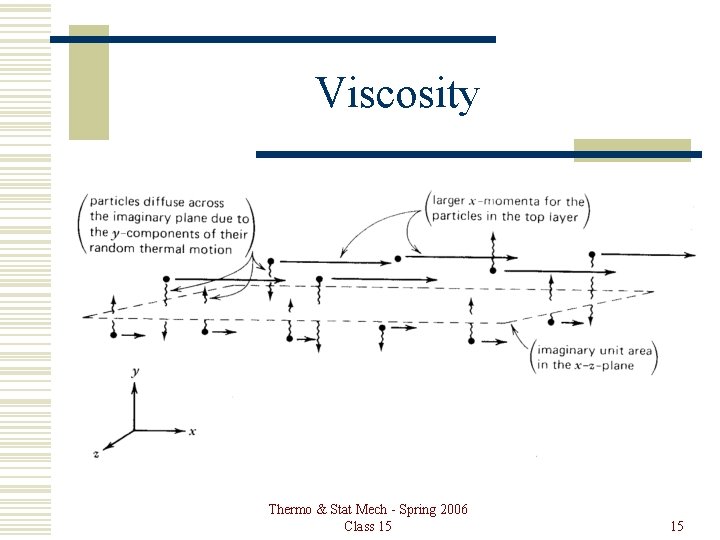
Viscosity Gas diffuses up and down, transferring x momentum from layer to layer. The gas that moves in the + y direction is going slower than the layer it moves into, and the gas that moves in the – y direction is going faster than the layer it moves into. Thermo & Stat Mech - Spring 2006 Class 15 14

Viscosity Thermo & Stat Mech - Spring 2006 Class 15 15
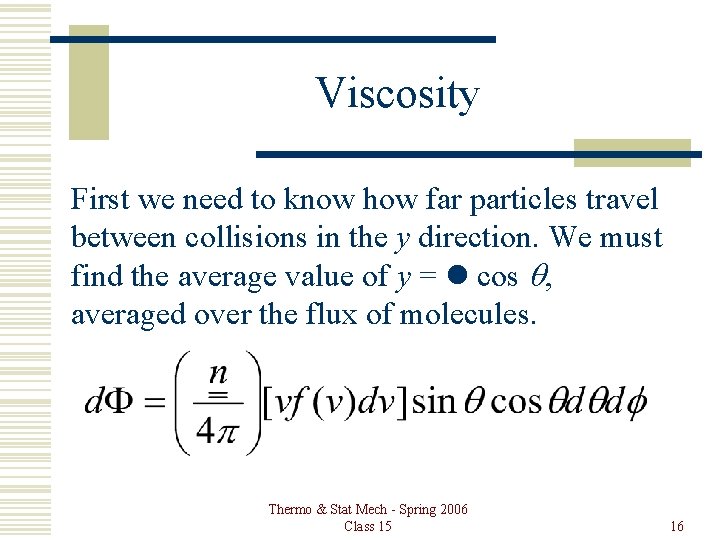
Viscosity First we need to know how far particles travel between collisions in the y direction. We must find the average value of y = l cos q, averaged over the flux of molecules. Thermo & Stat Mech - Spring 2006 Class 15 16
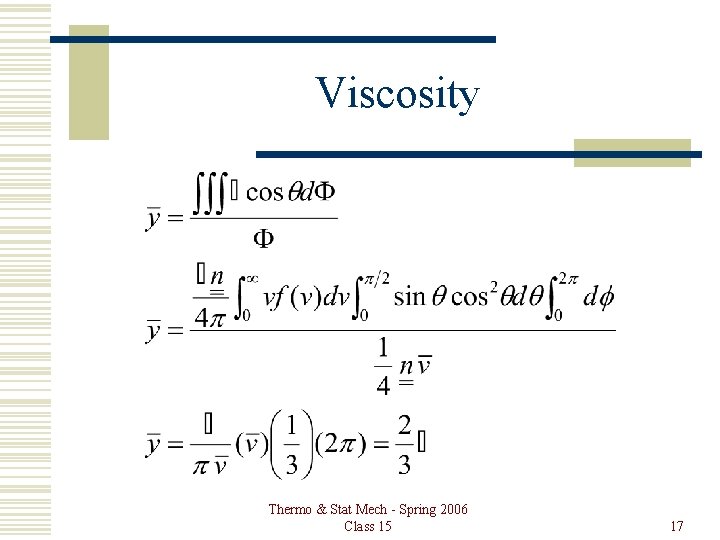
Viscosity Thermo & Stat Mech - Spring 2006 Class 15 17
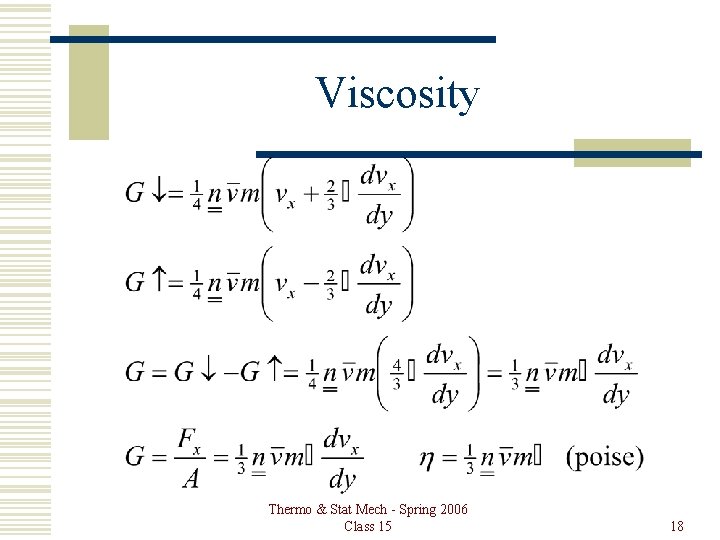
Viscosity Thermo & Stat Mech - Spring 2006 Class 15 18

Viscosity Thermo & Stat Mech - Spring 2006 Class 15 19

Diffusion Thermo & Stat Mech - Spring 2006 Class 15 20

Thermal Conductivity Thermo & Stat Mech - Spring 2006 Class 15 21

Thermal Conductivity Thermo & Stat Mech - Spring 2006 Class 15 22

Transport Processes Thermo & Stat Mech - Spring 2006 Class 15 23