Thermodynamics and Statistical Mechanics Partition Function Thermo Stat





























- Slides: 32

Thermodynamics and Statistical Mechanics Partition Function Thermo & Stat Mech Spring 2006 Class 19 1

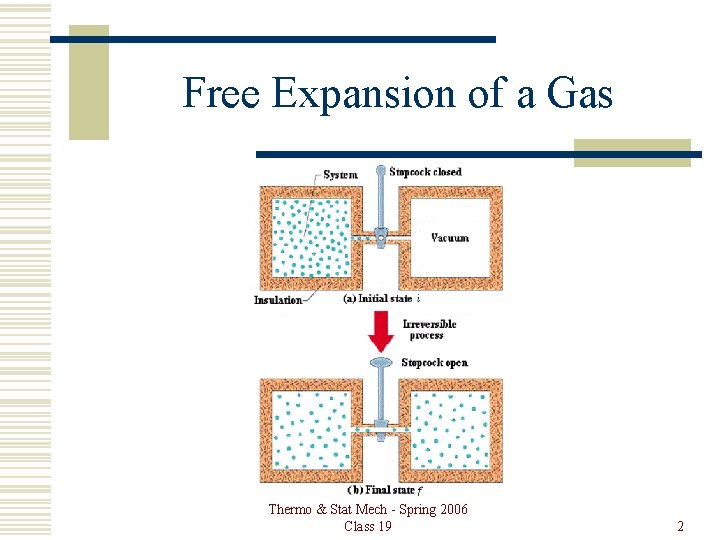
Free Expansion of a Gas Thermo & Stat Mech - Spring 2006 Class 19 2

Free Expansion Thermo & Stat Mech - Spring 2006 Class 19 3

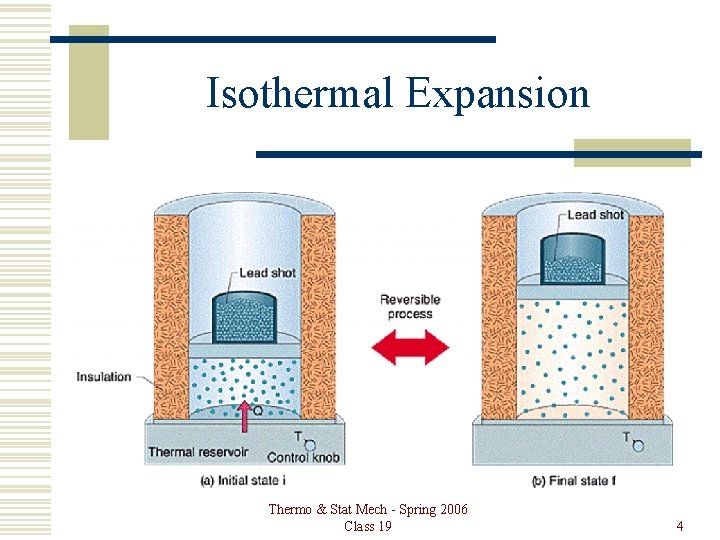
Isothermal Expansion Thermo & Stat Mech - Spring 2006 Class 19 4

Isothermal Expansion Reversible route between same states. đQ = đW + d. U Since T is constant, d. U = 0. Then, đQ = đW. Thermo & Stat Mech - Spring 2006 Class 19 5

Entropy Change The entropy of the gas increased. For the isothermal expansion, the entropy of the Reservoir decreased by the same amount. So for the system plus reservoir, DS = 0 For the free expansion, there was no reservoir. Thermo & Stat Mech - Spring 2006 Class 19 6

Statistical Approach Thermo & Stat Mech - Spring 2006 Class 19 7

Statistical Approach Thermo & Stat Mech - Spring 2006 Class 19 8

Partition Function Thermo & Stat Mech - Spring 2006 Class 19 9

Boltzmann Distribution Thermo & Stat Mech - Spring 2006 Class 19 10

Maxwell-Boltzmann Distribution Correct classical limit of quantum statistics is Maxwell-Boltzmann distribution, not Boltzmann. What is the difference? Thermo & Stat Mech - Spring 2006 Class 19 11

Maxwell-Boltzmann Probability w. B and w. MB yield the same distribution. Thermo & Stat Mech - Spring 2006 Class 19 12

Relation to Thermodynamics Thermo & Stat Mech - Spring 2006 Class 19 13

Relation to Thermodynamics Thermo & Stat Mech - Spring 2006 Class 19 14

Chemical Potential d. U = Td. S – Pd. V + md. N In this equation, m is the chemical energy per molecule, and d. N is the change in the number of molecules. Thermo & Stat Mech - Spring 2006 Class 19 15

Chemical Potential Thermo & Stat Mech - Spring 2006 Class 19 16

Entropy Thermo & Stat Mech - Spring 2006 Class 19 17

Entropy Thermo & Stat Mech - Spring 2006 Class 19 18

Helmholtz Function Thermo & Stat Mech - Spring 2006 Class 19 19

Chemical Potential Thermo & Stat Mech - Spring 2006 Class 19 20

Chemical Potential Thermo & Stat Mech - Spring 2006 Class 19 21

Boltzmann Distribution Thermo & Stat Mech - Spring 2006 Class 19 22

Distributions Thermo & Stat Mech - Spring 2006 Class 19 23

Distributions Thermo & Stat Mech - Spring 2006 Class 19 24

Ideal Gas Thermo & Stat Mech - Spring 2006 Class 19 25

Ideal Gas Thermo & Stat Mech - Spring 2006 Class 19 26

Ideal Gas Thermo & Stat Mech - Spring 2006 Class 19 27

Entropy Thermo & Stat Mech - Spring 2006 Class 19 28

Math Tricks For a system with levels that have a constant spacing (e. g. harmonic oscillator) the partition function can be evaluated easily. In that case, en = ne, so, Thermo & Stat Mech - Spring 2006 Class 19 29

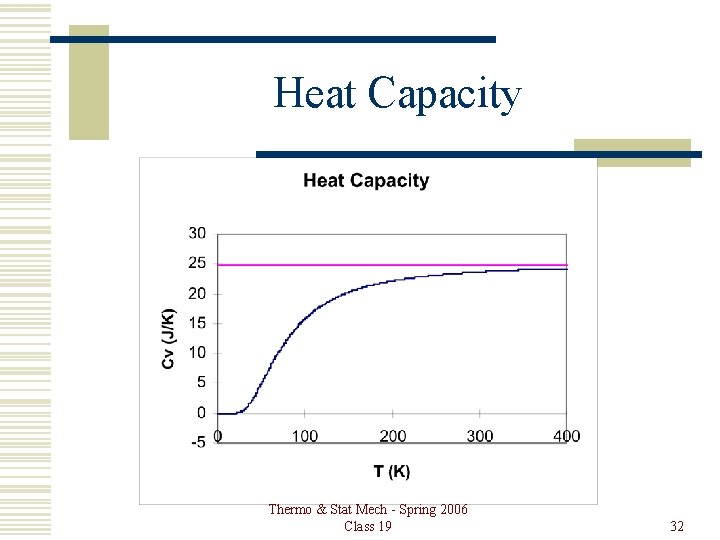
Heat Capacity of Solids Each atom has 6 degrees of freedom, so based on equipartition, each atom should have an average energy of 3 k. T. The energy per mole would be 3 RT. The heat capacity at constant volume would be the derivative of this with respect to T, or 3 R. That works at high enough temperatures, but approaches zero at low temperature. Thermo & Stat Mech - Spring 2006 Class 19 30

Heat Capacity Einstein found a solution by treating the solid as a collection of harmonic oscillators all of the same frequency. The number of oscillators was equal to three times the number of atoms, and the frequency was chosen to fit experimental data for each solid. Your class assignment is to treat the problem as Einstein did. Thermo & Stat Mech - Spring 2006 Class 19 31

Heat Capacity Thermo & Stat Mech - Spring 2006 Class 19 32