Thermodynamics and Statistical Mechanics First Law of Thermodynamics





















- Slides: 27

Thermodynamics and Statistical Mechanics First Law of Thermodynamics Thermo & Stat Mech Spring 2006 Class 3

Review of van der Waals Critical Values Thermo & Stat Mech - Spring 2006 Class 3 2

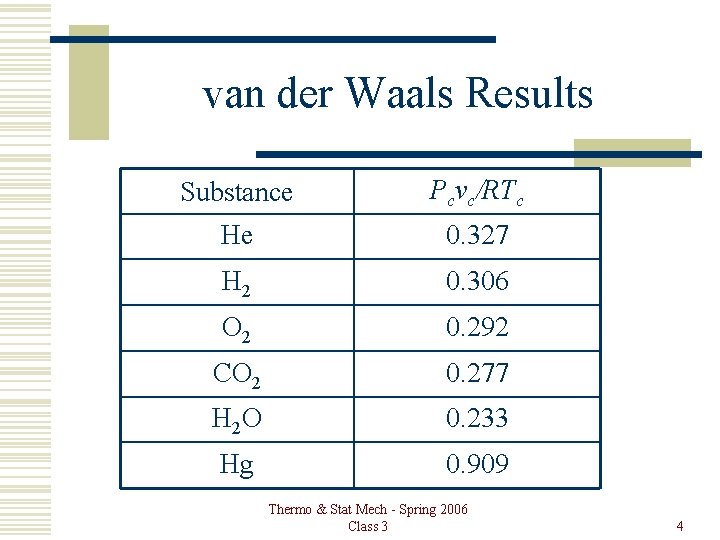
van der Waals Results Thermo & Stat Mech - Spring 2006 Class 3 3

van der Waals Results Substance Pcvc/RTc He 0. 327 H 2 0. 306 O 2 0. 292 CO 2 0. 277 H 2 O 0. 233 Hg 0. 909 Thermo & Stat Mech - Spring 2006 Class 3 4

Configuration Work đW = Pd. V Gas, Liquid, Solid: Thermo & Stat Mech - Spring 2006 Class 3 5

Kinds of Processes Often, something is held constant. Examples: d. V = 0 d. Q = 0 d. P = 0 d. T = 0 isochoric or isovolumic process adiabatic process isobaric process isothermal process Thermo & Stat Mech - Spring 2006 Class 3 6

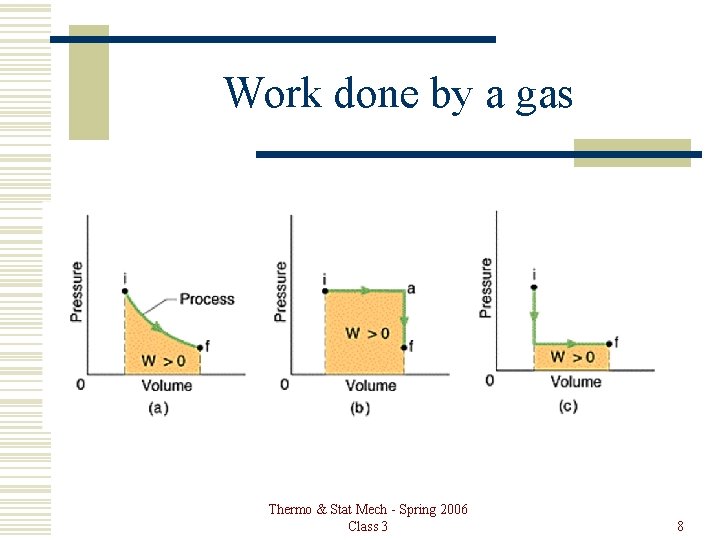
Work done by a gas đW = Pd. V For isochoric process d. V = 0, so W = 0 For isobaric process d. P = 0, so W = PDV Thermo & Stat Mech - Spring 2006 Class 3 7

Work done by a gas Thermo & Stat Mech - Spring 2006 Class 3 8

Work done by an ideal gas For isothermal process d. T = 0, so T = constant. Thermo & Stat Mech - Spring 2006 Class 3 9

Isothermal Process Thermo & Stat Mech - Spring 2006 Class 3 10

Heat Capacity Heat capacity measures the amount of heat that must be added to a system to increase its temperature by a given amount. Its definition: where y is a property of the system that is kept constant as heat is added. Thermo & Stat Mech - Spring 2006 Class 3 11

Heat Capacity Properties that are usually kept constant for a hydrostatic system are volume or pressure. Then, Thermo & Stat Mech - Spring 2006 Class 3 12

Heat Capacity Clearly, the heat capacity depends on the size of the system under consideration. To get rid of that effect, and have a heat capacity that depends only on the properties of the substance being studied, two other quantities are defined: specific heat capacity, and molar heat capacity. Thermo & Stat Mech - Spring 2006 Class 3 13

Specific Heat Capacity Specific heat capacity is the heat capacity per mass of the system. A lower case letter is used to represent the specific heat capacity. Then, if m is the mass of the system, Thermo & Stat Mech - Spring 2006 Class 3 14

Molar Heat Capacity Molar heat capacity is the heat capacity per mole of the system. A lower case letter is used to represent the molar heat capacity. Then, if there are n moles in the system, Thermo & Stat Mech - Spring 2006 Class 3 15

Shorter Version Use heat per mass. Thermo & Stat Mech - Spring 2006 Class 3 16

c. P – c. V đq = du + Pdv where u(T, v) Thermo & Stat Mech - Spring 2006 Class 3 17

Constant Volume Thermo & Stat Mech - Spring 2006 Class 3 18

Constant Pressure Thermo & Stat Mech - Spring 2006 Class 3 19

Ideal Gas u is not a function of v. Thermo & Stat Mech - Spring 2006 Class 3 20

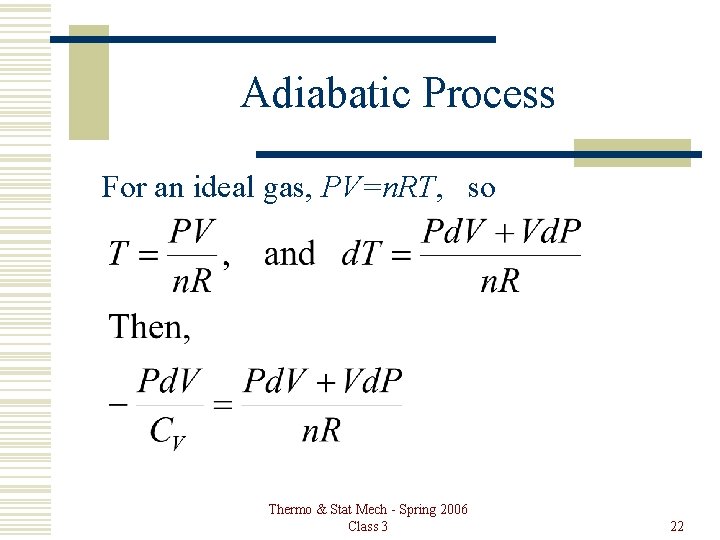
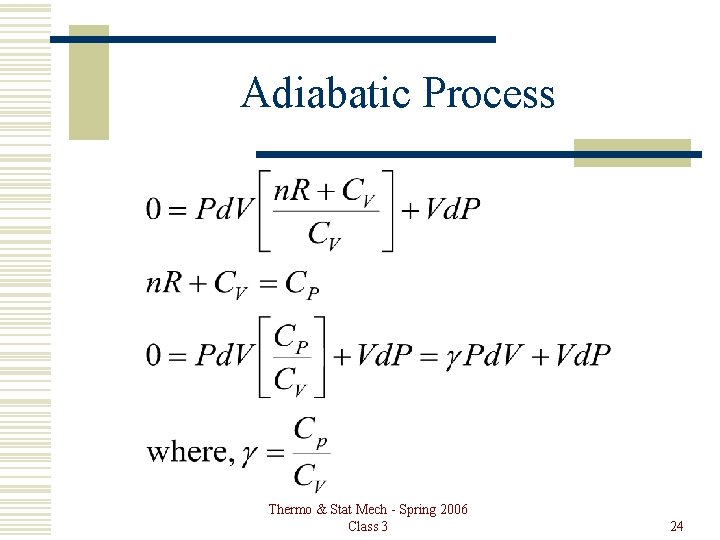
Adiabatic Process For an ideal gas, and most real gasses, đQ = d. U + Pd. V đQ = CVd. T + Pd. V, . Then, when đQ = 0, Thermo & Stat Mech - Spring 2006 Class 3 21

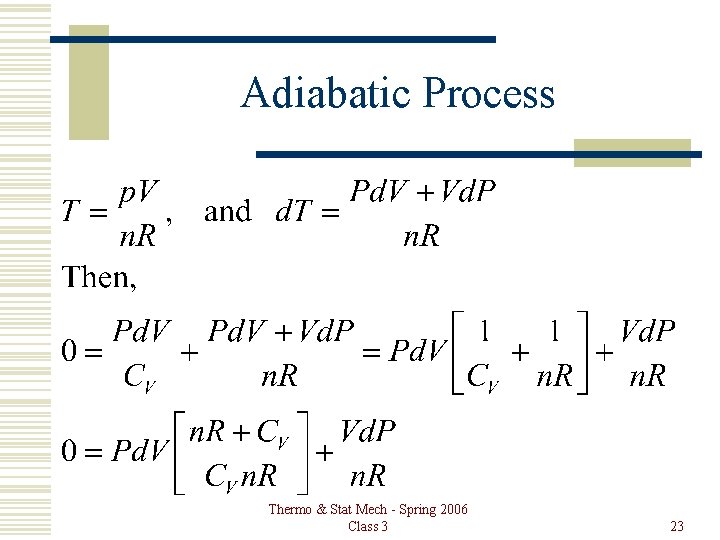
Adiabatic Process For an ideal gas, PV=n. RT, so Thermo & Stat Mech - Spring 2006 Class 3 22

Adiabatic Process Thermo & Stat Mech - Spring 2006 Class 3 23

Adiabatic Process Thermo & Stat Mech - Spring 2006 Class 3 24

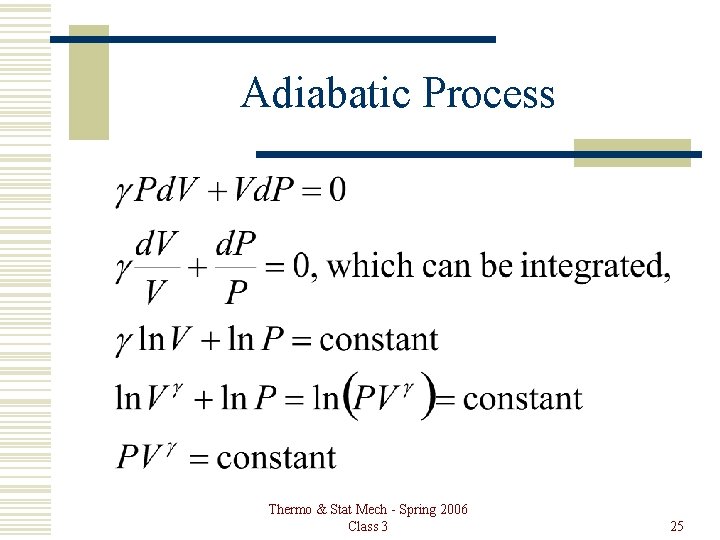
Adiabatic Process Thermo & Stat Mech - Spring 2006 Class 3 25

Adiabatic Process Thermo & Stat Mech - Spring 2006 Class 3 26

g for “Ideal Gasses” Thermo & Stat Mech - Spring 2006 Class 3 27