Thermodynamics and Statistical Mechanics Entropy and the Second


















- Slides: 23

Thermodynamics and Statistical Mechanics Entropy and the Second Law of Thermodynamics Thermo & Stat Mech Spring 2006 Class 6 1

Second Law Kelvin-Planck statement: No series of processes is possible whose sole result is the absorption of heat from a thermal reservoir and the complete conversion of this energy to work. There are no perfect engines! Thermo & Stat Mech - Spring 2006 Class 6 2

Second Law Clausius statement: No series of processes is possible whose sole result is the transfer of heat from a reservoir at a given temperature to a reservoir at a higher temperature. There are no perfect refrigerators! Thermo & Stat Mech - Spring 2006 Class 6 3

Carnot Cycle is Best A Carnot cycle is the most efficient possible, operating between two reservoirs at temperatures T 1 and T 2. Proof: Assume there is a more efficient engine. Let it produce work, and use that work to run a Carnot refrigerator between the same two reservoirs. Thermo & Stat Mech - Spring 2006 Class 6 4

Carnot Cycle is Best Thermo & Stat Mech - Spring 2006 Class 6 5

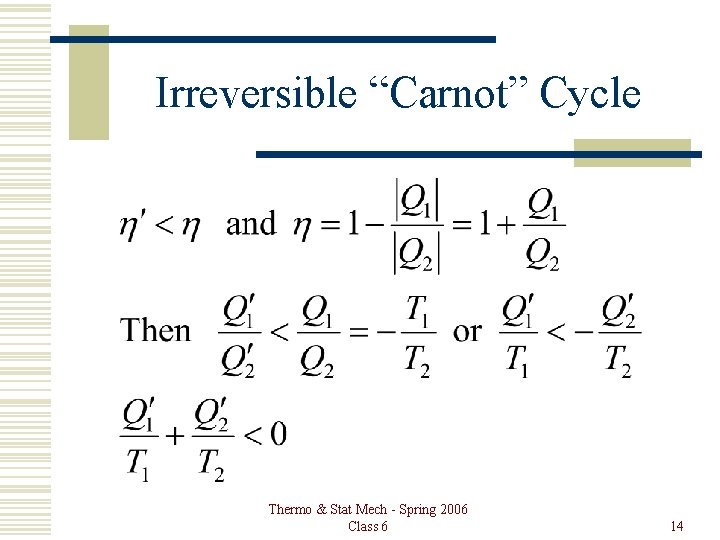
Carnot Cycle is Best Then, |Q 2¢| < |Q 2|. Also, |W| = |Q 2¢| – |Q 1¢| = |Q 2| – |Q 1|. So, |Q 2| – |Q 2¢| = |Q 1| – |Q 1¢|, and |Q 1¢| < |Q 1| also. Heat has been taken out of the low temperature reservoir and put into the high temperature reservoir with no expenditure of work! Not possible. Thermo & Stat Mech - Spring 2006 Class 6 6

For a Carnot Engine or Thermo & Stat Mech - Spring 2006 Class 6 7

For infinitesimal cycles Any cycle can be represented as a sum of infinitesimal Carnot cycles. Then, Thermo & Stat Mech - Spring 2006 Class 6 8

Carnot Cycles Thermo & Stat Mech - Spring 2006 Class 6 9

Entropy For reversible processes. Entropy is a state variable. Thermo & Stat Mech - Spring 2006 Class 6 10

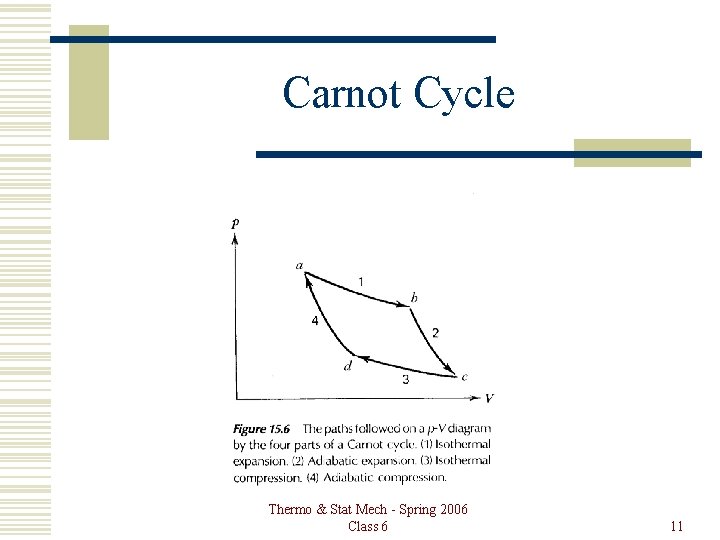
Carnot Cycle Thermo & Stat Mech - Spring 2006 Class 6 11

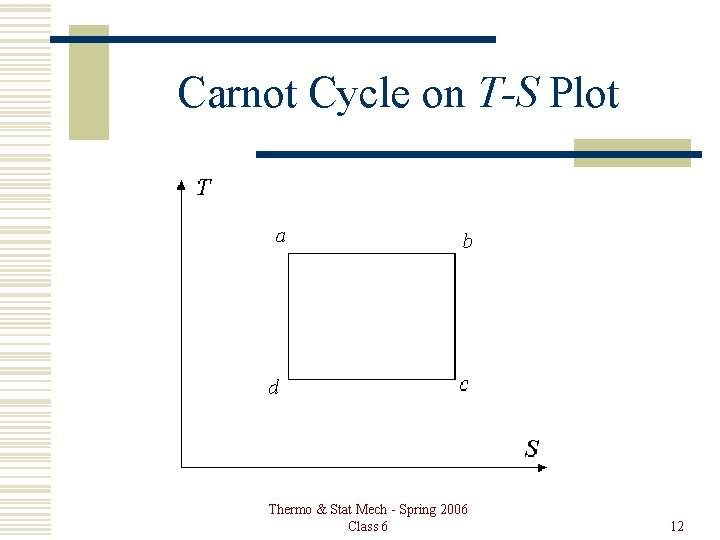
Carnot Cycle on T-S Plot Thermo & Stat Mech - Spring 2006 Class 6 12

Carnot Cycle The area enclosed by the cycle on a P-V plot is the net work done per cycle. (đW = Pd. V) The area enclosed by the cycle on a T-S plot is the net heat added per cycle. (đQ = Td. S for any reversible process. ) These two quantities are equal. Thermo & Stat Mech - Spring 2006 Class 6 13

Irreversible “Carnot” Cycle Thermo & Stat Mech - Spring 2006 Class 6 14

Clausius Inequality Irreversible cycle In general Thermo & Stat Mech - Spring 2006 Class 6 15

Entropy Change The equal sign applies for reversible processes. Thermo & Stat Mech - Spring 2006 Class 6 16

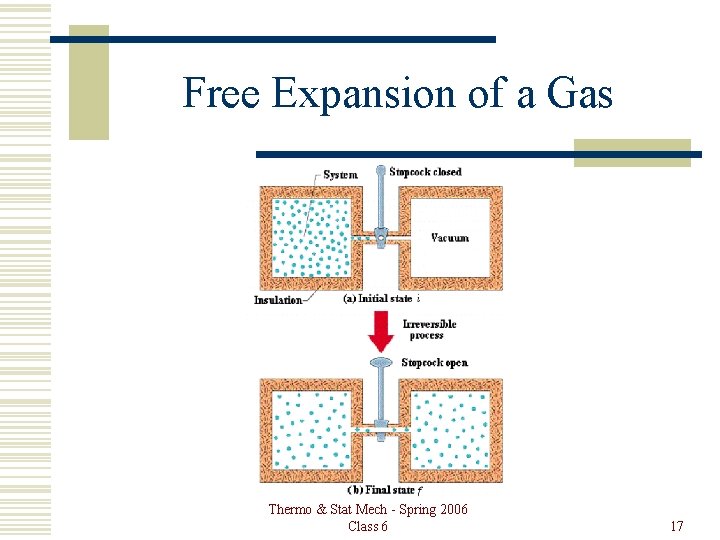
Free Expansion of a Gas Thermo & Stat Mech - Spring 2006 Class 6 17

Free Expansion Thermo & Stat Mech - Spring 2006 Class 6 18

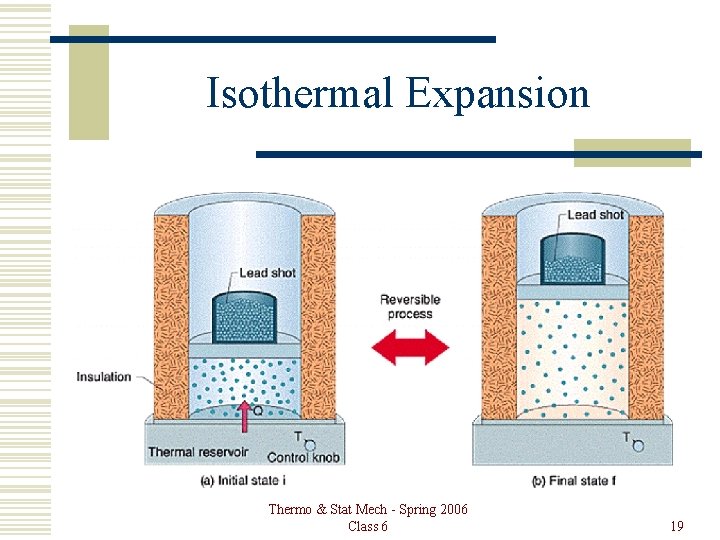
Isothermal Expansion Thermo & Stat Mech - Spring 2006 Class 6 19

Isothermal Expansion Reversible route between same states. đQ = đW + d. U Since T is constant, d. U = 0. Then, đQ = đW. Thermo & Stat Mech - Spring 2006 Class 6 20

Entropy Change The entropy of the gas increased. For the isothermal expansion, the entropy of the Reservoir decreased by the same amount. So for the system plus reservoir, DS = 0 For the free expansion, there was no reservoir. Thermo & Stat Mech - Spring 2006 Class 6 21

Second Law of Thermodynamics The entropy of an isolated system increases in any irreversible process and is unaltered in any reversible process. This is the principle of increasing entropy. DS ³ 0 Thermo & Stat Mech - Spring 2006 Class 6 22

First and Second Laws First Law: d. U = đQ – đW First law, combined with the second law: (for reversible processes) d. U = Td. S – Pd. V Thermo & Stat Mech - Spring 2006 Class 6 23