Thermodynamics And Relationships between heat and work What

Thermodynamics And Relationships between heat and work

What is Internal Energy? • Internal energy is defined as the energy associated with the random, disordered motion of molecules.

Heat, Work and Internal Energy • Internal energy can be used for work. Example #1: Friction forces generated through pulling a nail from wood, increase the nail’s temperature. The energy can be transferred to the surrounding air. (The work is done by the friction forces. ) Serway/Faughn Physics – pg 332

Heat, Work and Internal Energy Internal energy can be used for work. Example #2: Consider a flask of water with a balloon placed over the opening. Heating the water cause it to boil. The water vapor expands the balloon. The balloon expansion provides a force that does work on the atmosphere. The steam does the work.

Heat & Work are Energy Transferred to or from a System • Objects contain internal energy, but are not said to have heat or work. The heat or work is transferred to or from a substance. Serway/Faughn Physics – pg 332 • The coffee cup feels hot as it is transferring heat energy to your hand.

Heat energy is transferred from a warmer object to a cooler object.

Energy transfer to or from a system • A balloon, flask, water, steam can be thought of as a system. A burner transfers energy to this system. The system internal energy is increased. • When the expanding balloon does work on the surroundings, the system’s internal energy is decreased. • Some of the energy is transferred into the system asheat is transferred to the surroundings. Serway/Faughn Physics – pg 332

For Thermodynamic systems work is defined in terms of pressure and volume change. • Thermo – thermal energy – heat • Dynamic – changing • Therefore thermodynamic involves changes in heat/energy.

Example – Gas expanding and pushing a piston within a cylinder does positive work the piston. As the gas is compressed, the work done on the piston is negative. Serway/Faughn Physics – pg 332

First Law of Thermodynamics • Energy cannot be created or destroyed, but transferred or converted from one form to another.
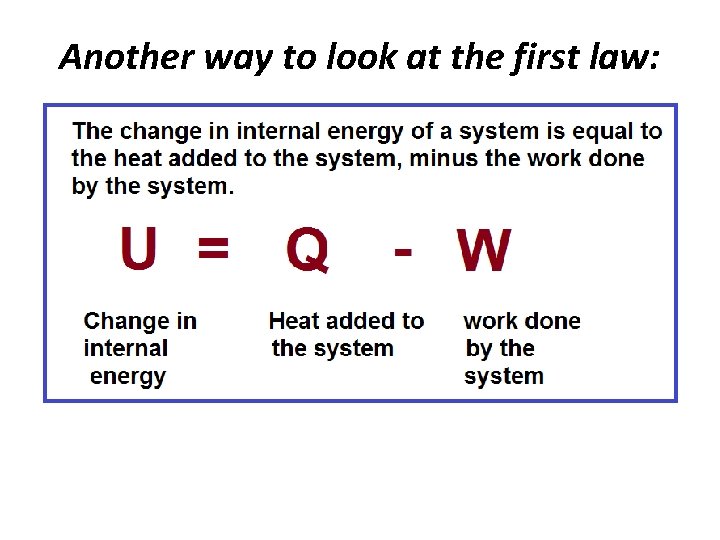
Another way to look at the first law:
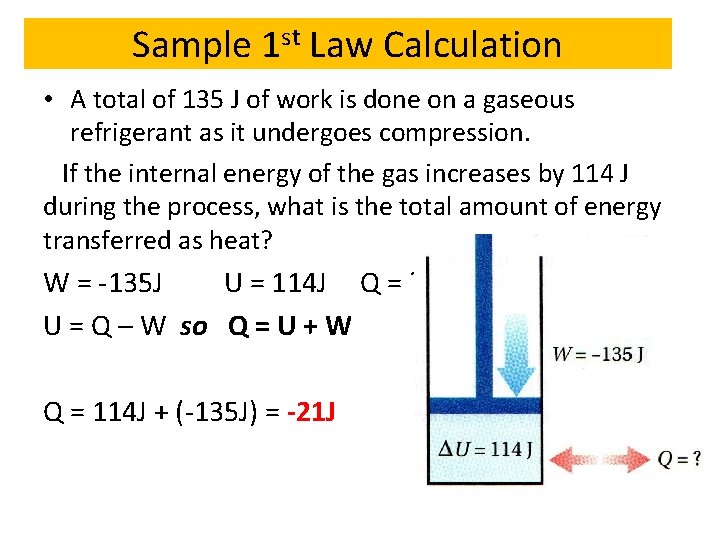
Sample 1 st Law Calculation • A total of 135 J of work is done on a gaseous refrigerant as it undergoes compression. If the internal energy of the gas increases by 114 J during the process, what is the total amount of energy transferred as heat? W = -135 J U = 114 J Q = ? U = Q – W so Q = U + W Q = 114 J + (-135 J) = -21 J
Refrigerators and Heat engines (Cyclical processes) • A refrigerator performs mechanical work to create temperature difference between its closed interior and its environment (the air in the room). • This is accomplished in a cyclical process of compression and expansion or refrigerant, and transferring thermal energy.

Heat engines • A heat engine is a device that uses heat to do mechanical work. • A heat engine does work by transferring energy from a hightemperature substance to a lowertemperature substance.

Internal Combustion Engines • Internal combustion engines are examples of heat engines. Potential energy of chemical bonds in fuel is converted to kinetic energy of particle products from combustion. • These gaseous products push against a piston to do work. Only part of the internal energy leaves the engine as work done on the environment (pistons). Most of the energy is removed as heat.

Four-Cycle Gasoline Engine Intake stroke – An air-fuel mixture is drawn into the cylinder through the intake valve as the piston moves downward. (The exhaust valve stays closed. )

• Compression stroke – work is done by the piston as the air-fuel mixture is compressed in the cylinder. (Both valves are closed at this time. )

• Power stroke – The compressed, hot gases are ignited and combustion takes place. The combustion of gases cause the piston to move downward in the cylinder. A great deal of this energy, in the form of heat, is transferred to the surrounding environment. (Both valves remain closed)

• Exhaust Stroke – The piston moves up through the cylinder and pushes the combustion products back out the cylinder through an exhaust valve.

The Second Law of Thermodynamics • With regards to a heat engine: “No cyclic process that converts heat entirely into work is possible. • Some energy is always transferred as heat into the surroundings. ” Serway/Faugh Physics pg 348 • How much heat energy is converted to work, instead of being lost to the surroundings refers to the efficiency of that engine.

Entropy – tendency toward disorder • “In thermodynamics, a system left to itself tends to go from a state with a very ordered set of energies to one where there is less order. ” Serway/Faugh Physics pg 351 • All systems tend toward more disorder and randomness.

• The measure of a system’s disorder is called the entropy of the system. The greater the entropy, the greater the disorder. Greater disorder means there is less energy to do work.
- Slides: 22