Thermodynamics and Metabolism Metabolism all the chemical reactions

Thermodynamics and Metabolism

Metabolism: all the chemical reactions occurring in an organism that change or transform matter and energy in cells Anabolism: chemical reactions that expend energy to make new chemical bonds (build larger molecules) Catabolism: chemical reactions that harvest energy when bonds are broken (breaking down compounds) C 3, C 2. 1

Flow of Energy: the capacity to do work -kinetic energy: the energy of motion -potential energy: stored energy Energy can take many forms: mechanical chemical thermal light C 3, C 2. 1

Bond Energy = the amount of energy needed to break or form a chemical bond Energy is always released when a bond forms, therefore free atoms have more chemical energy than compounds (Fig. 3. 2 p 115) C 3, C 2. 1

• In the lab, energy from chemical reactions is usually in the form of heat energy • In biological systems, too much heat is damaging, so some energy may be released in the form of: – movement (of materials across the cell), – contraction of muscles, – phosphorescence (light), – or may be used to make another reaction occur in a metabolic pathway. C 3, C 2. 1

Laws of Thermodynamics: Study of the transfer and transformation of thermal energy First Law of Thermodynamics – energy cannot be created or destroyed, but can be converted from one form to another. Amount of energy in the universe is constant E. g. : sunlight energy chemical energy photosynthesis C 3, C 2. 1

Second Law of Thermodynamics: during any process, the universe tends to move towards disorder. entropy: disorder in the universe The 2 nd Law of Thermodynamics states that entropy is always increasing. Fig. 3. 4 p 117 C 3, C 2. 1

C 3, C 2. 1

Free energy: the energy from a chemical reaction available to do work -denoted by the symbol G (Gibb’s free energy) enthalpy: energy contained in a molecule’s chemical bonds free energy = enthalpy – (temp. x entropy) G = H - TS C 3, C 2. 1

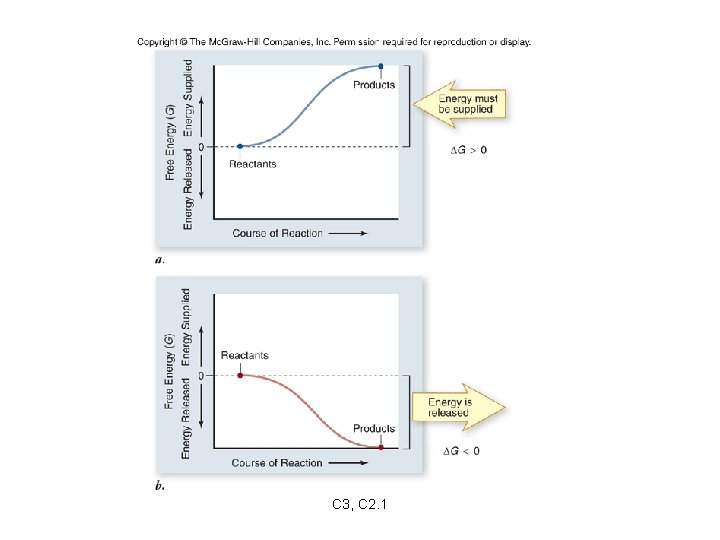
• Chemical reactions can create changes in free energy. • When products contain more free energy than reactants DG is positive. • When reactants contain more free energy than products DG is negative. C 3, C 2. 1

Chemical reactions can be described by the transfer of energy that occurs: endergonic reaction: a reaction requiring an input of energy - DG is positive exergonic reaction: a reaction that releases free energy - DG is negative C 3, C 2. 1
C 3, C 2. 1

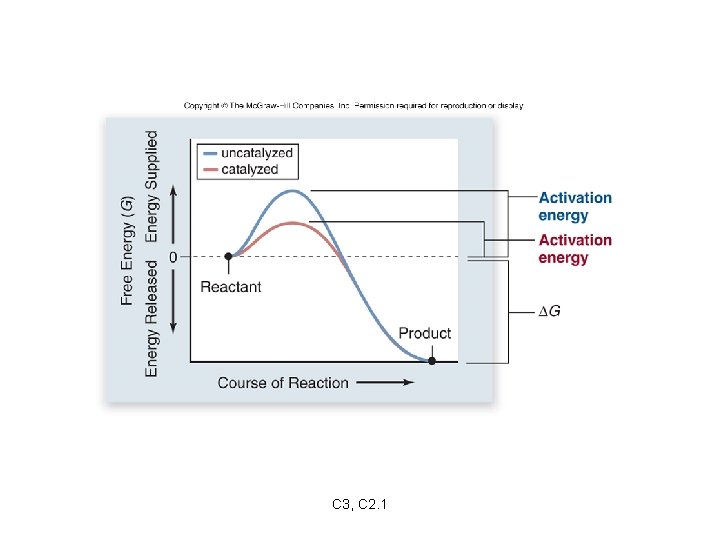
Most reactions require some energy to get started. activation energy: extra energy needed to get a reaction started -destabilizes existing chemical bonds -required even for exergonic reactions catalysts: substances that lower the activation energy of a reaction C 3, C 2. 1
C 3, C 2. 1

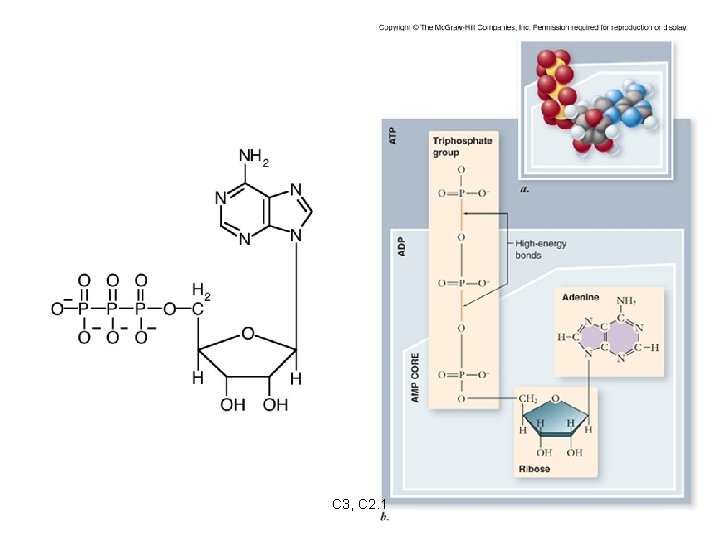
Energy Currency of Cells ATP = adenosine triphosphate -the energy “currency” of cells ATP structure: -ribose, a 5 -carbon sugar -adenine -three phosphates C 3, C 2. 1
C 3, C 2. 1

ATP stores energy in the bonds between phosphates. Phosphates are highly negative, therefore: -the phosphates repel each other -much energy is required to keep the phosphates bound to each other -much energy is released when the bond between two phosphates is broken C 3, C 2. 1

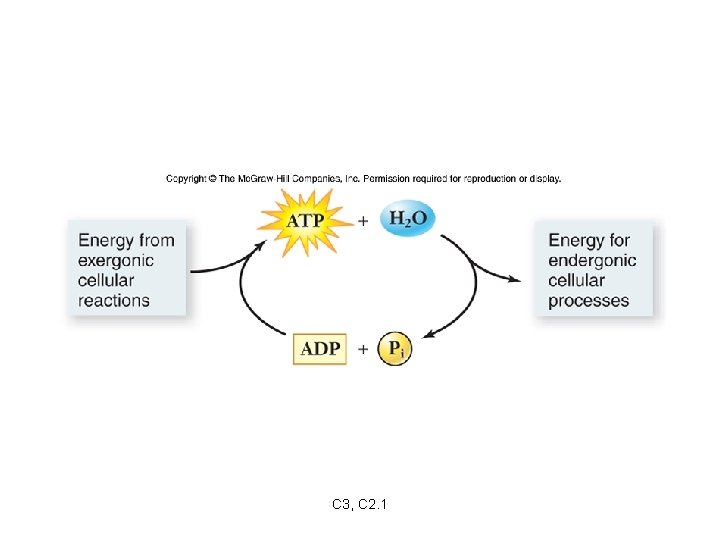
When the bond between phosphates is broken: ATP ADP + Pi energy is released ADP = adenosine diphosphate Pi = inorganic phosphate This reaction is reversible. Fig. 3. 6 p. 119 C 3, C 2. 1
C 3, C 2. 1

The energy released when ATP is broken down to ADP can be used to fuel endergonic reactions. The energy released from an exergonic reaction can be used to fuel the production of ATP from ADP + Pi. C 3, C 2. 1

Electron Carriers • Are compounds that pick up electrons from energy-rich compounds and donate them to low-energy compounds • These are REDOX reactions • Electrons carry energy with them • Two main electron carriers: – NAD+ C 3, C 2. 1
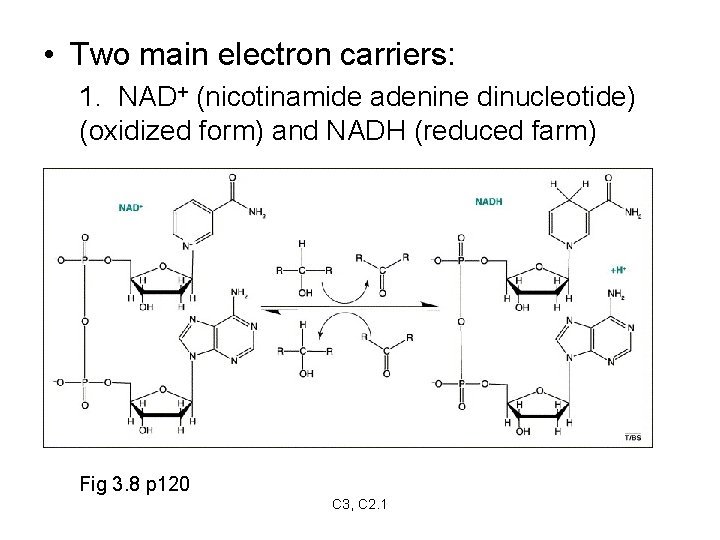
• Two main electron carriers: 1. NAD+ (nicotinamide adenine dinucleotide) (oxidized form) and NADH (reduced farm) Fig 3. 8 p 120 C 3, C 2. 1
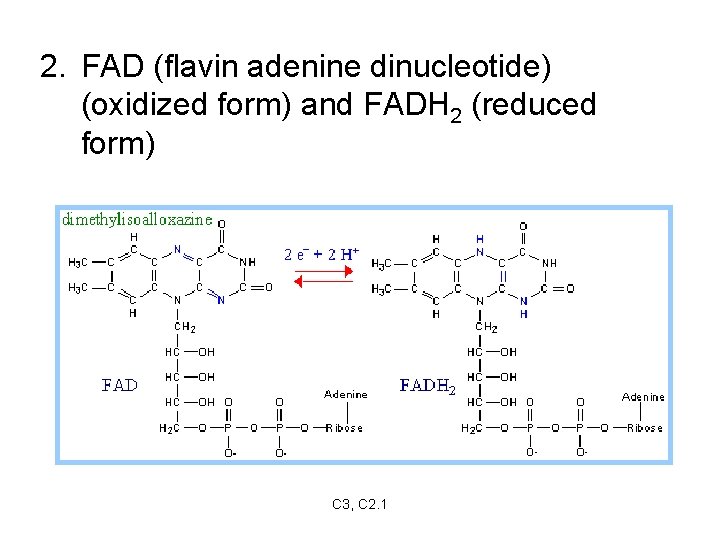
2. FAD (flavin adenine dinucleotide) (oxidized form) and FADH 2 (reduced form) C 3, C 2. 1

• HMK: – P 121 #1, 2, 5 -8, 11, 13 C 3, C 2. 1
- Slides: 24