Thermodynamics 1 Chapter 1 Introduction and Basic Concepts

Thermodynamics 1 Chapter 1 Introduction and Basic Concepts Assoc. Prof. Dr. Sommai Priprem Reference: Cengel, Yunus A. and Michael A. Boles, Thermodynamics: An Engineering Approach, 5 th ed. , New York, Mc. Graw-Hill: 2006.
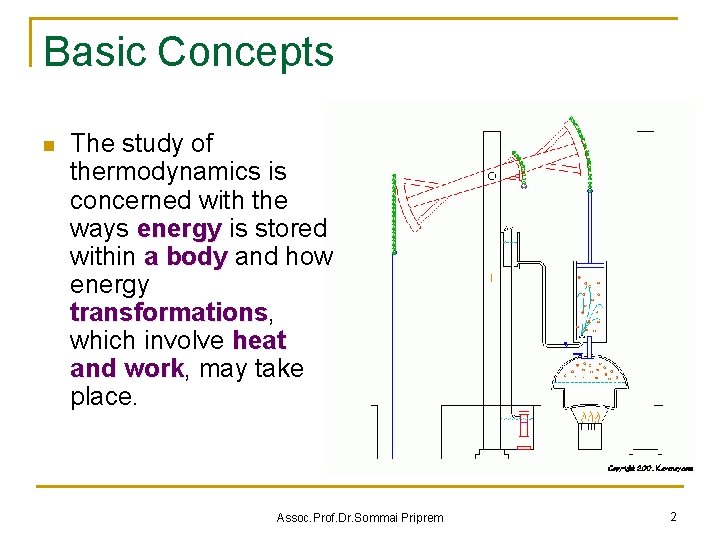
Basic Concepts n The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, transformations which involve heat and work, work may take place. Assoc. Prof. Dr. Sommai Priprem 2

The Thermodynamics Laws n n n One of the most fundamental laws of nature is the conservation of energy principle It is the FIRST LAW of Thermodynamics. It also called the principle of ENERGY BALANCE Assoc. Prof. Dr. Sommai Priprem 3

Approaches to studying thermodynamics n n Classical Thermodynamics (Macro) Statistical Thermodynamics (Micro) Assoc. Prof. Dr. Sommai Priprem 4

Applications of Thermodynamics Assoc. Prof. Dr. Sommai Priprem 5
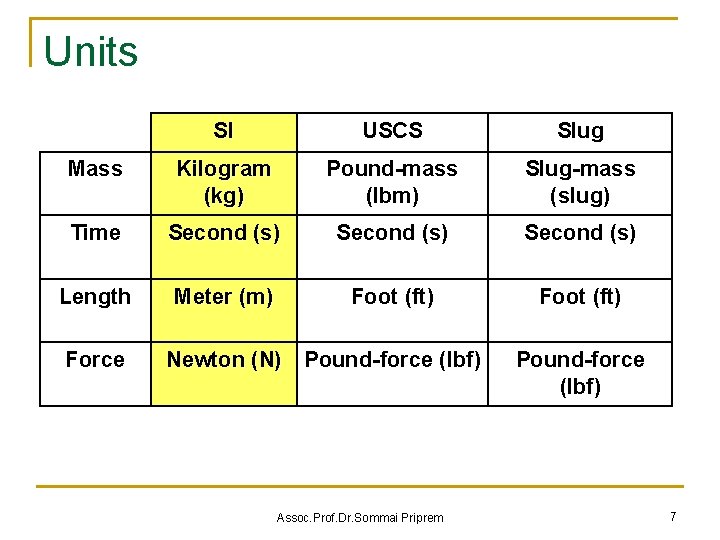
Units SI USCS Slug Mass Kilogram (kg) Pound-mass (lbm) Slug-mass (slug) Time Second (s) Length Meter (m) Foot (ft) Force Newton (N) Pound-force (lbf) Assoc. Prof. Dr. Sommai Priprem Pound-force (lbf) 7
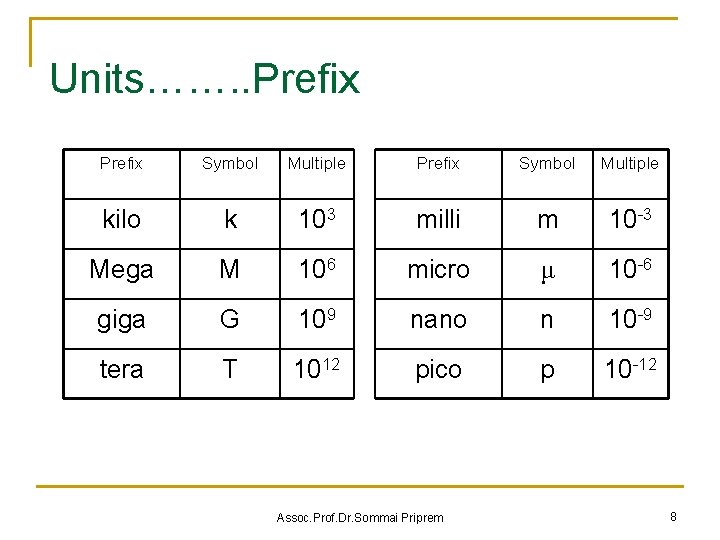
Units……. . Prefix Symbol Multiple kilo k 103 milli m 10 -3 Mega M 106 micro μ 10 -6 giga G 109 nano n 10 -9 tera T 1012 pico p 10 -12 Assoc. Prof. Dr. Sommai Priprem 8

n n n Force, F Weight, W Pressure, P = ma, = mg, = F/A, kg-m/s 2 = N N/m 2 = Pa 1 Bar Assoc. Prof. Dr. Sommai Priprem = 105 Pa 9

Group Discussion 1. 2. 3. vs Water 1 m 3 /kg vs Water 1 m 3 /s vs Water 1 kg /s Water 1 m 3 Water 1 kg Example 1. 2 A tank is filled with oil whose density is ρ = 850 kg/m 3. If the volume of the Tank is V = 2 m 3, determind the amount of mass m in the tank. Solution m = ρV m = (850 kg/m 3)(2 m 3) m = 1, 700 kg Answer Assoc. Prof. Dr. Sommai Priprem 10

Thermodynamics ENERGY FLUID
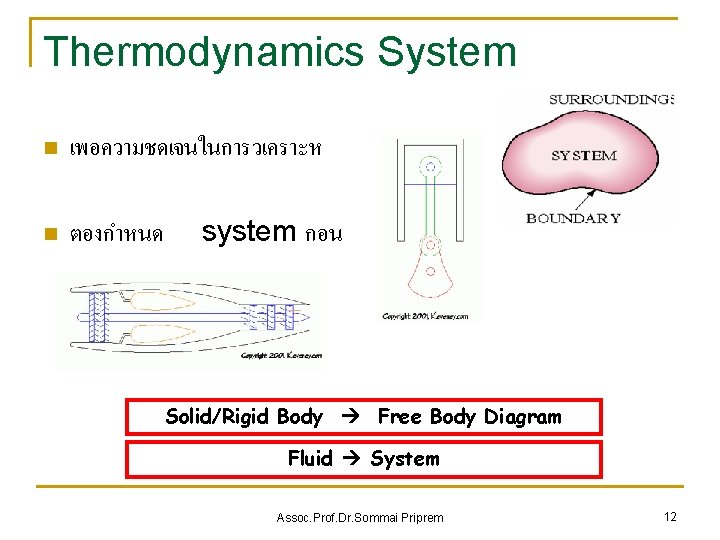
Thermodynamics System n เพอความชดเจนในการวเคราะห n ตองกำหนด system กอน Solid/Rigid Body Free Body Diagram Fluid System Assoc. Prof. Dr. Sommai Priprem 12
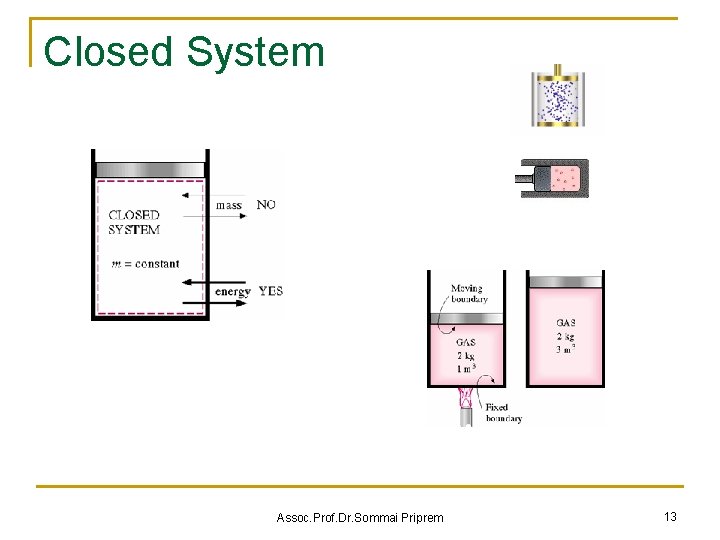
Closed System Assoc. Prof. Dr. Sommai Priprem 13
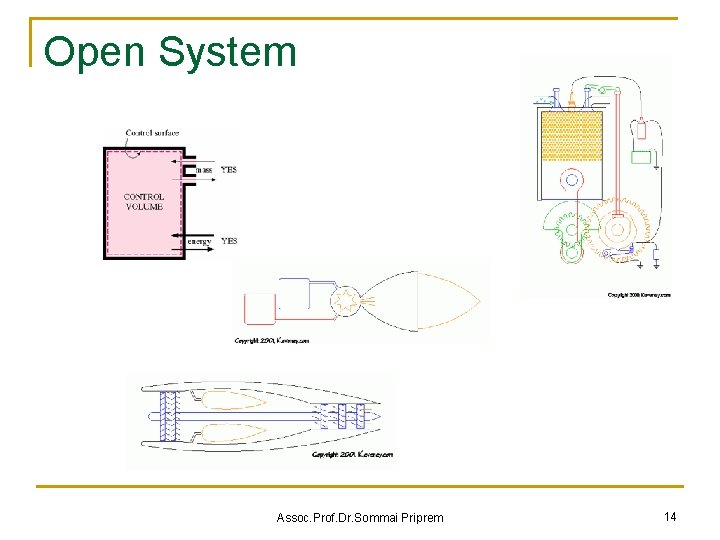
Open System Assoc. Prof. Dr. Sommai Priprem 14
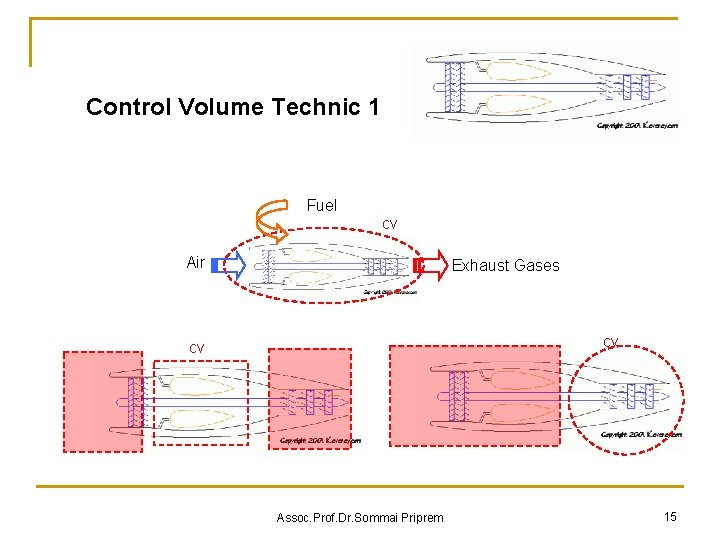
Control Volume Technic 1 Fuel cv Air Exhaust Gases cv cv Assoc. Prof. Dr. Sommai Priprem 15
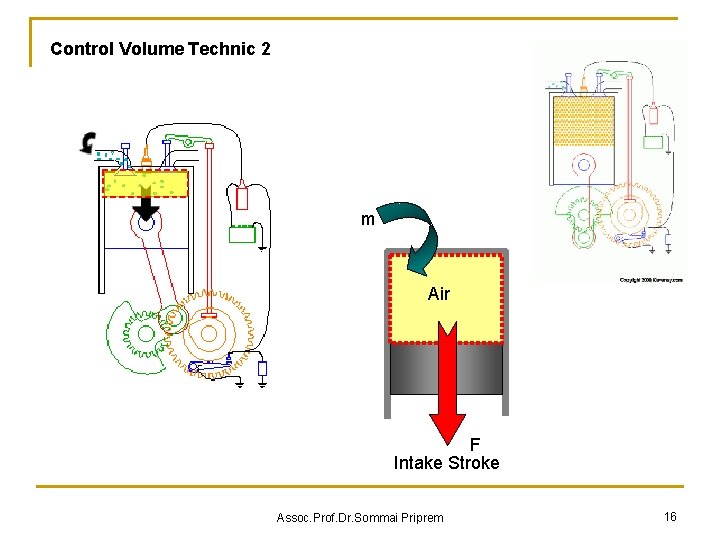
Control Volume Technic 2 m Air F Intake Stroke Assoc. Prof. Dr. Sommai Priprem 16
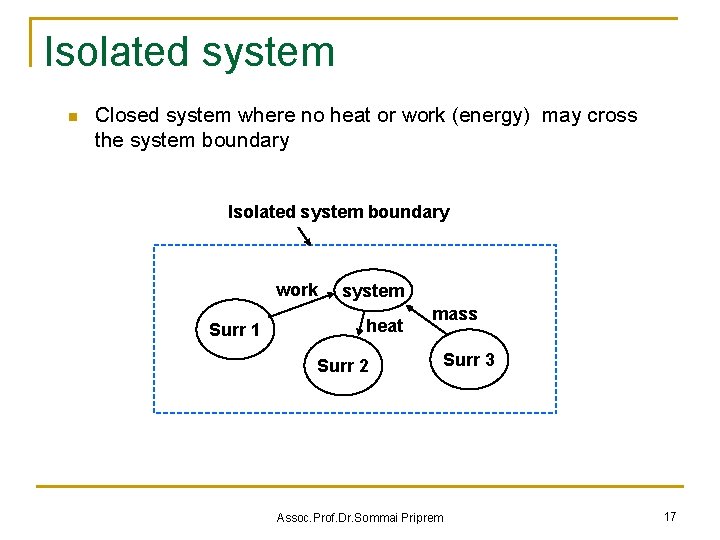
Isolated system n Closed system where no heat or work (energy) may cross the system boundary Isolated system boundary work Surr 1 system heat mass Surr 2 Assoc. Prof. Dr. Sommai Priprem Surr 3 17

Total Energy of a System n n n Sum of all forms of energy (i. e. , thermal, mechanical, kinetic, potential, electrical, magnetic, chemical, and nuclear) that can exist in a system For systems we typically deal with in this course, sum of internal, kinetic, and potential energies E = U + KE + PE q E = Total energy of system q U = internal energy q KE = kinetic energy = m. V 2/2 q PE = potential energy = mgz Assoc. Prof. Dr. Sommai Priprem 18
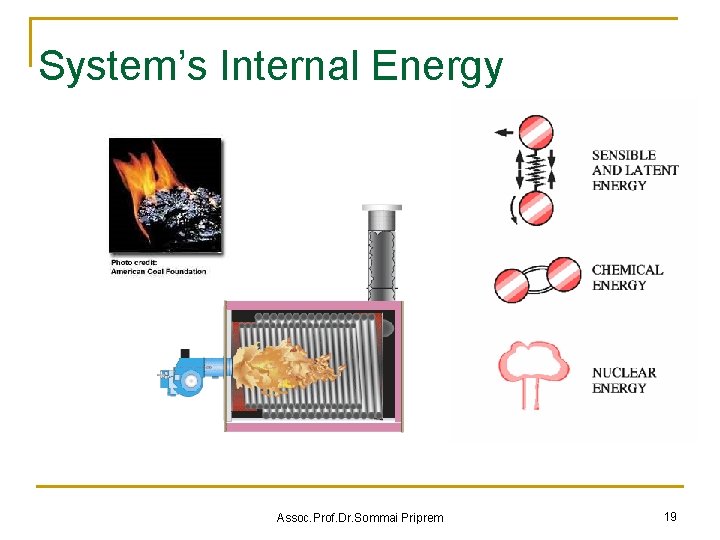
System’s Internal Energy = Sum of Microscopic Energies Assoc. Prof. Dr. Sommai Priprem 19
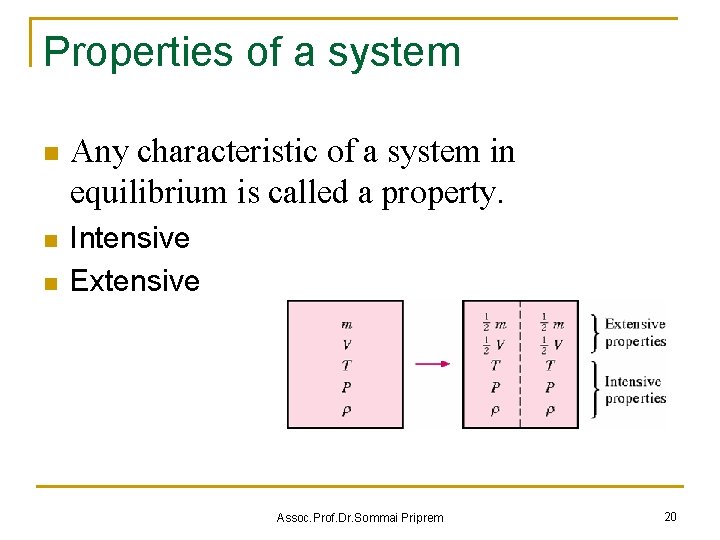
Properties of a system n Any characteristic of a system in equilibrium is called a property. n Intensive Extensive n Assoc. Prof. Dr. Sommai Priprem 20

State and Equilibrium n State of a system q q q n system that is not undergoing any change all properties of system are known & are not changing if one property changes then the state of the system changes Thermodynamic equilibrium q q “equilibrium” - state of balance A system is in equilibrium if it maintains thermal (uniform temperature), mechanical (uniform pressure), phase (mass of two phases), and chemical equilibrium Assoc. Prof. Dr. Sommai Priprem 21
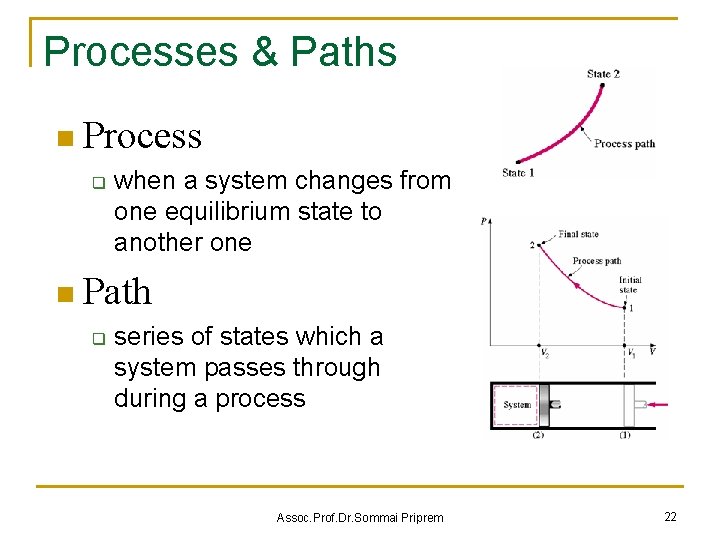
Processes & Paths n Process q when a system changes from one equilibrium state to another one n Path q series of states which a system passes through during a process Assoc. Prof. Dr. Sommai Priprem 22
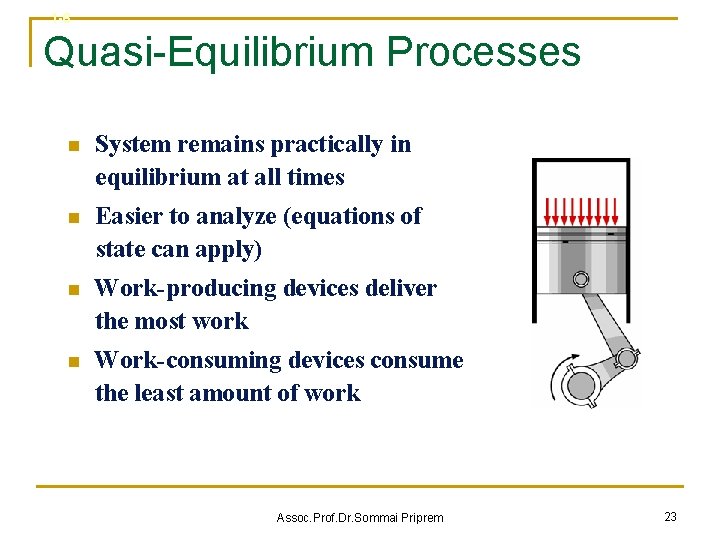
1 -6 Quasi-Equilibrium Processes n System remains practically in equilibrium at all times n Easier to analyze (equations of state can apply) n Work-producing devices deliver the most work n Work-consuming devices consume the least amount of work Assoc. Prof. Dr. Sommai Priprem 23
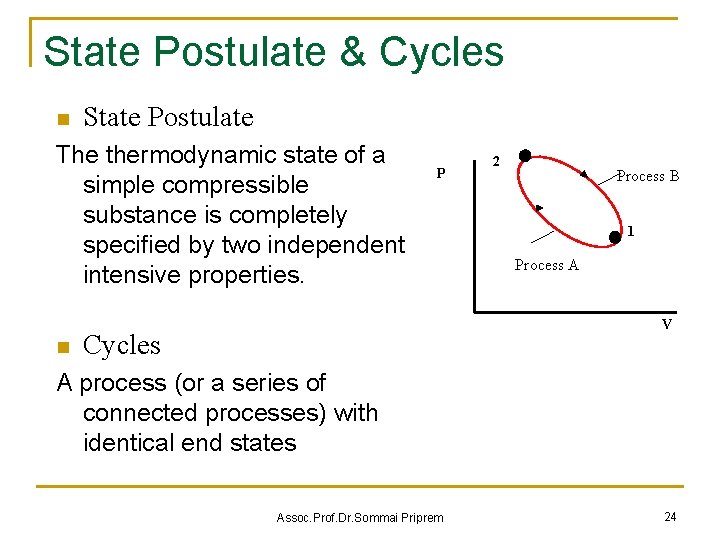
State Postulate & Cycles n State Postulate The thermodynamic state of a simple compressible substance is completely specified by two independent intensive properties. n P 2 Process B 1 Process A V Cycles A process (or a series of connected processes) with identical end states Assoc. Prof. Dr. Sommai Priprem 24

State point identification n n n Cold Water Cooler Water Warmer Water Hot Water Boiled Water Boiling Water • How can we identified the different • How can we tell the EXACT condition of a fluid Assoc. Prof. Dr. Sommai Priprem 25
Process & Cycle Assoc. Prof. Dr. Sommai Priprem 26
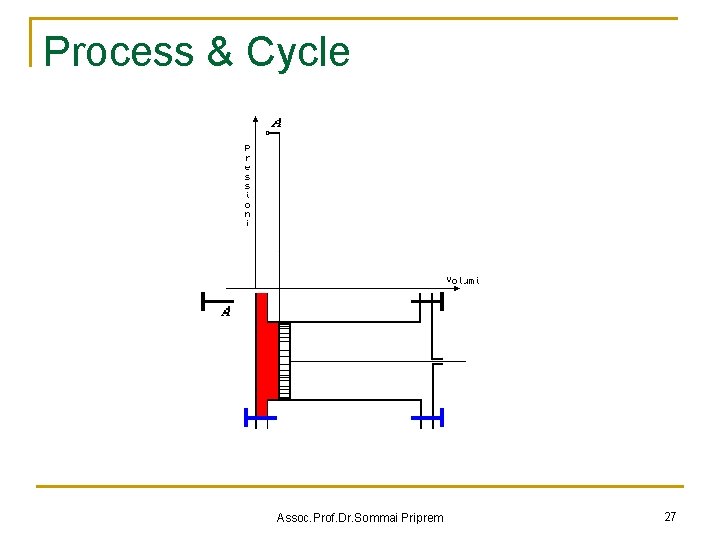
Process & Cycle Assoc. Prof. Dr. Sommai Priprem 27




The Zero Law of Thermodynamics n If two bodies are in thermal equilibrium with a third body, they are also in thermal equilibrium with each other. B A if TA = TC and TB = TC then TA = TB C Assoc. Prof. Dr. Sommai Priprem 31
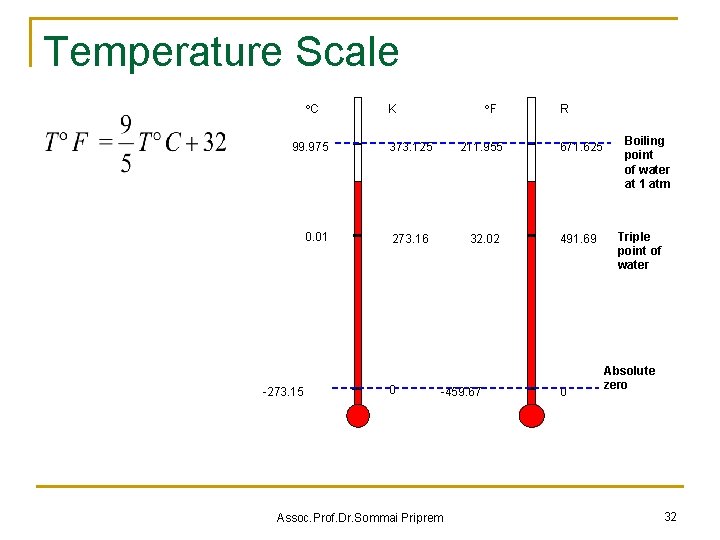
Temperature Scale C 99. 975 0. 01 -273. 15 F K R 373. 125 211. 955 671. 625 273. 16 32. 02 491. 69 0 -459. 67 Assoc. Prof. Dr. Sommai Priprem 0 Boiling point of water at 1 atm Triple point of water Absolute zero 32
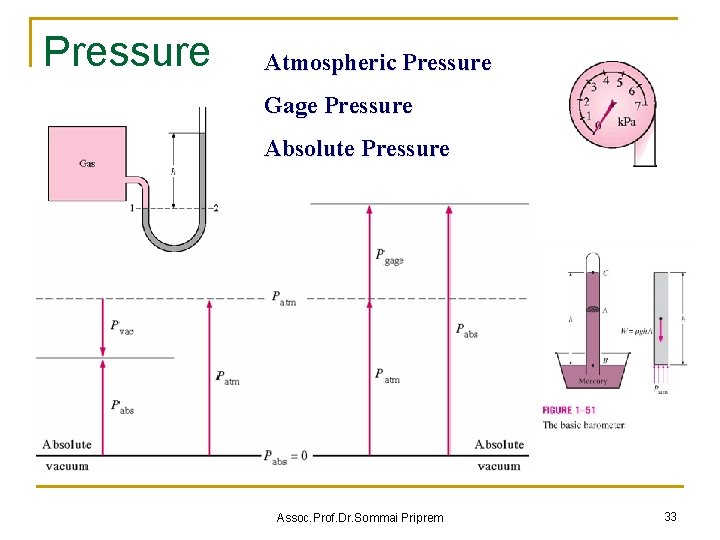
Pressure Atmospheric Pressure Gage Pressure Absolute Pressure Assoc. Prof. Dr. Sommai Priprem 33
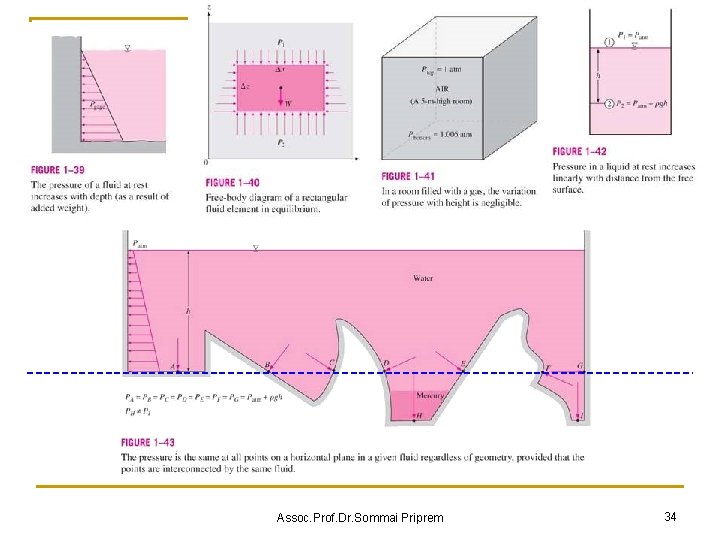
Assoc. Prof. Dr. Sommai Priprem 34

n Example 1. 6 Measuring Pressure with a Manometer A manometer is used to measure the pressure in a tank. The fluid used has a specific gravity of 0. 85, and the manometer column height is 55 cm, as show in fig 1 -46. If the local atmospheric pressure is 96 k. Pa, determine the absolute pressure within the tank. Pabs= Patm+ Pg Pg = ρgh ans Assoc. Prof. Dr. Sommai Priprem 100. 6 k. Pa 35

n Example 1. 8 Measuring Atmospheric Pressure with a Barometer Determine the pressure at a location where the barometric reading is 740 mm. Hg. and the gravitational acceleration is g = 9. 81 m/s 2. Assume the temperature of mercury to be 10 o. C, at which its density is 13, 570 kg/m 3. Pg = ρgh ans Assoc. Prof. Dr. Sommai Priprem 98. 5 k. Pa 36

n Example 1. 9 Effect og Piston Weight on Pressure in a Cylinder The piston of a vertical piston-cylinder device containing a gas has a mass of 60 kg and a cross-sectional area of 0. 04 m 2. , as shown in Fif 1 -54. The local atmospheric pressure is 0. 97 bar, and the gravitational acceleration is 9. 81 m/s 2. (a) Determine the pressure inside the cylinder. (b) If some heat is transferred to the gas and its volume is doubled, do you expect the pressure inside the cylinder change? Pabs= Patm+ Ppiston = W/A ans Assoc. Prof. Dr. Sommai Priprem 1. 12 bar 37

Chapter Outline 1 -1 Thermodynamics and Energy 1 -2 A Note on Dimensions and Units 1 -3 Closed and Open Systems 1 -4 Properties of a System 1 -5 State and Equilibrium 1 -6 Processes and Cycles 1 -7 Forms of Energy 1 -8 Energy and Environment 1 -9 Temperature and the Zeroth Law of Thermodynamics 1 -10 Pressure 1 -11 The Manometer 1 -12 Barometer and the Atmospheric Pressure Assoc. Prof. Dr. Sommai Priprem 38





That’s for today Don’t forget to review by reading a TEXT BOOK. Next lecture “Properties. . ” please bring along a Thermodynamics TABLE
- Slides: 43