Thermodynamic Properties w Property Table from direct measurement

Thermodynamic Properties w Property Table -from direct measurement w Equation of State -- any equations that relates P, v, and T of a substance

Ideal -Gas Equation of State w Any relation among the pressure, temperature, and specific volume of a substance is called an equation of state. The simplest and best-known equation of state is the ideal-gas equation of state, given as where R is the gas constant. Caution should be exercised in using this relation since an ideal gas is a fictitious substance. Real gases exhibit ideal-gas behavior at relatively low pressures and high temperatures.

Universal Gas Constant Universal gas constant is given on Ru = 8. 31434 k. J/kmol-K = 8. 31434 k. Pa-m 3/kmol-k = 0. 0831434 bar-m 3/kmol-K = 82. 05 L-atm/kmol-K = 1. 9858 Btu/lbmol-R = 1545. 35 ft-lbf/lbmol-R = 10. 73 psia-ft 3/lbmol-R

Example Determine the particular gas constant for air and hydrogen.

Ideal Gas “Law” is a simple Equation of State
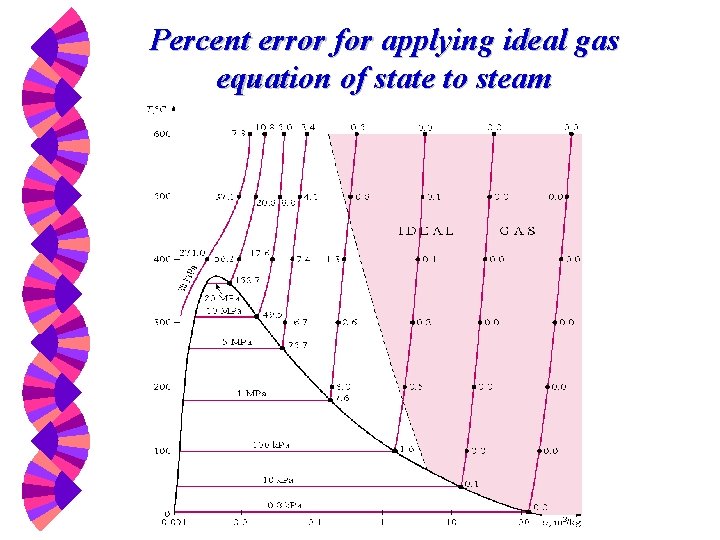
Percent error for applying ideal gas equation of state to steam

Question …. . . Under what conditions is it appropriate to apply the ideal gas equation of state?

Ideal Gas Law Good approximation for P-v-T behaviors of real gases at low densities (low pressure and high temperature). w Air, nitrogen, oxygen, hydrogen, helium, argon, neon, carbon dioxide, …. ( < 1% error). w

Compressibility Factor w The deviation from ideal-gas behavior can be properly accounted for by using the compressibility factor Z, defined as Z represents the volume ratio or compressibility.

Ideal Gas Real Gases Z=1 Z > 1 or Z<1

Real Gases w Pv = ZRT or w Pv = ZRu. T, where v is volume per unit mole. Z is known as the compressibility factor. w Real gases, Z < 1 or Z > 1. w

Compressibility factor What is it really doing? w It accounts mainly for two things w • Molecular structure • Intermolecular attractive forces

Principle of corresponding states w. The compressibility factor Z is approximately the same for all gases at the same reduced temperature and reduced pressure. Z = Z(PR, TR) for all gases

Reduced Pressure and Temperature where: PR and TR are reduced values. Pcr and Tcr are critical properties.
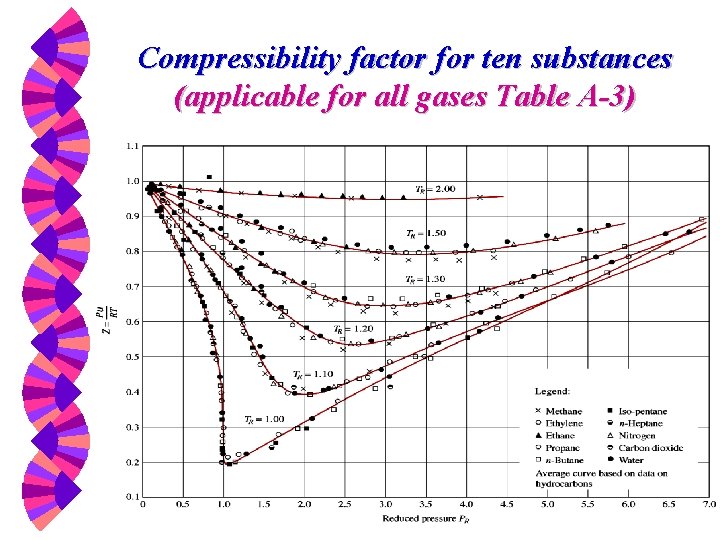
Compressibility factor for ten substances (applicable for all gases Table A-3)
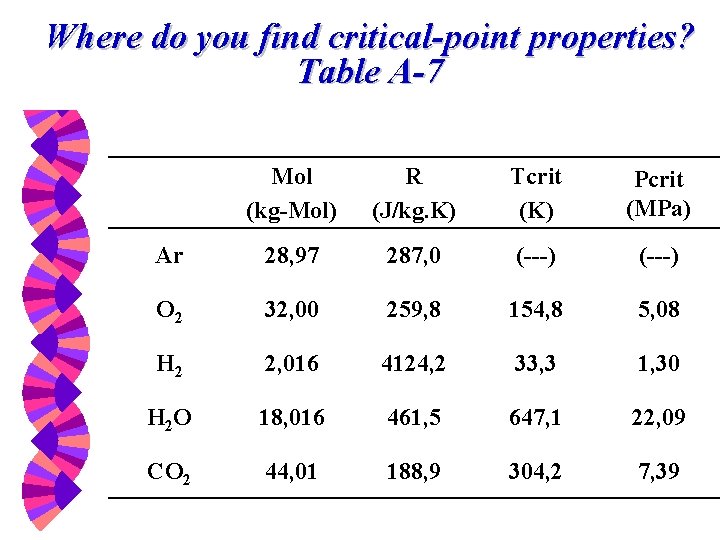
Where do you find critical-point properties? Table A-7 Mol (kg-Mol) R (J/kg. K) Tcrit (K) Pcrit (MPa) Ar 28, 97 287, 0 (---) O 2 32, 00 259, 8 154, 8 5, 08 H 2 2, 016 4124, 2 33, 3 1, 30 H 2 O 18, 016 461, 5 647, 1 22, 09 CO 2 44, 01 188, 9 304, 2 7, 39

Reduced Properties This works great if you are given a gas, a P and a T and asked to find the v. w However, if you are given P and v and asked to find T (or T and v and asked to find P), trouble lies ahead. w Use pseudo-reduced specific volume. w

Pseudo-Reduced Specific Volume w When either P or T is unknown, Z can be determined from the compressibility chart with the help of the pseudoreduced specific volume, defined as not vcr !

Ideal-Gas Approximation w The compressibility chart shows the conditions for which Z = 1 and the gas behaves as an ideal gas: w (a) PR < 0. 1 and TR > 1
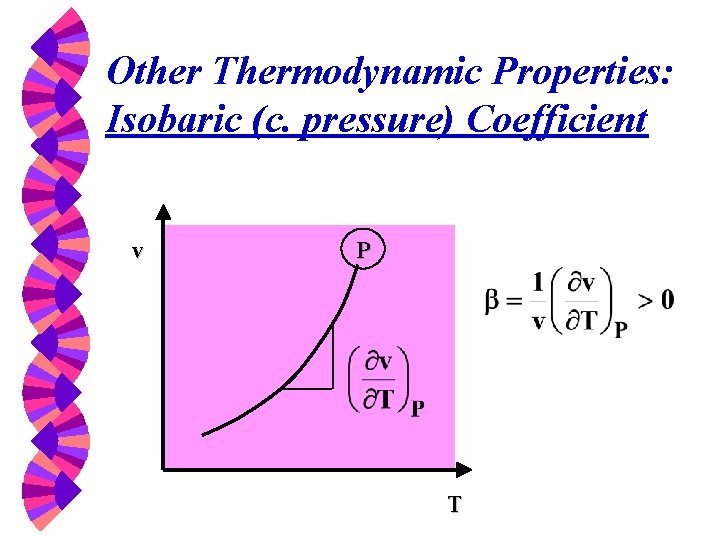
Other Thermodynamic Properties: Isobaric (c. pressure) Coefficient v P T
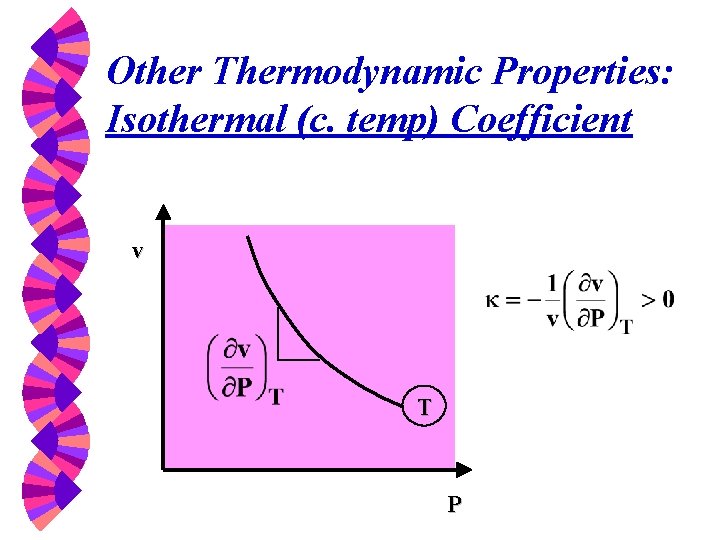
Other Thermodynamic Properties: Isothermal (c. temp) Coefficient v T P

Other Thermodynamic Properties: We can think of the volume as being a function of pressure and temperature, v = v(P, T). Hence infinitesimal differences in volume are expressed as infinitesimal differences in P and T, using k and b coefficients If k and b are constant, we can integrate for v:

Other Thermodynamic Properties: Internal Energy, Enthalpy and Entrop
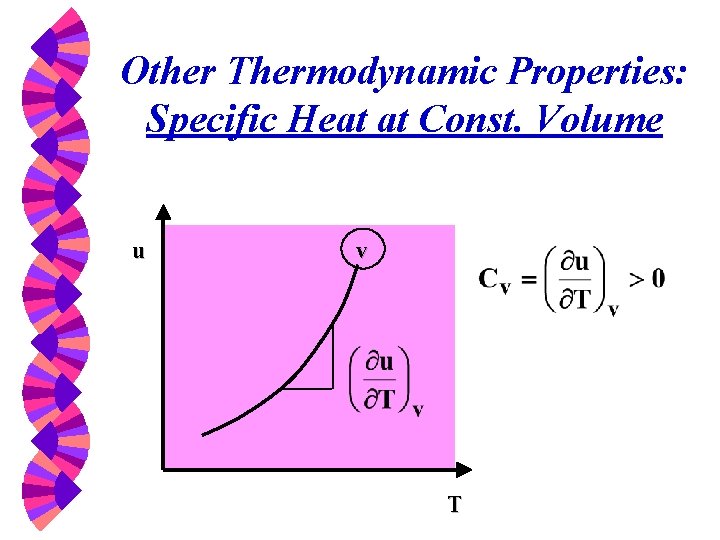
Other Thermodynamic Properties: Specific Heat at Const. Volume u v T
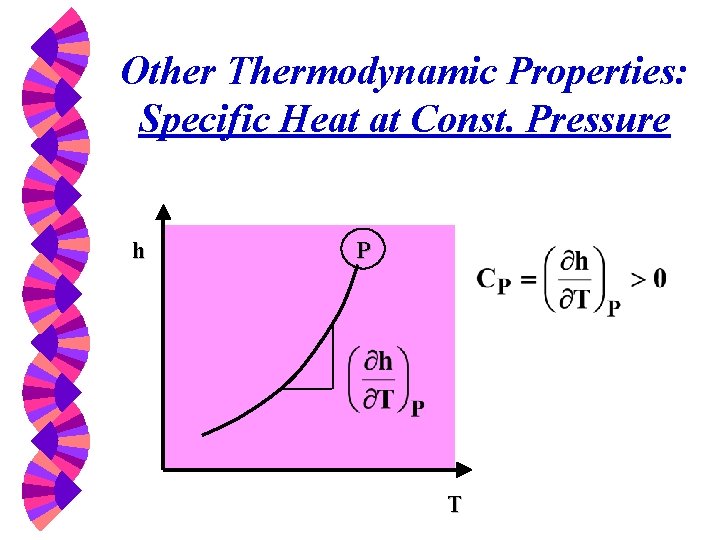
Other Thermodynamic Properties: Specific Heat at Const. Pressure h P T

Other Thermodynamic Properties: Ratio of Specific Heat
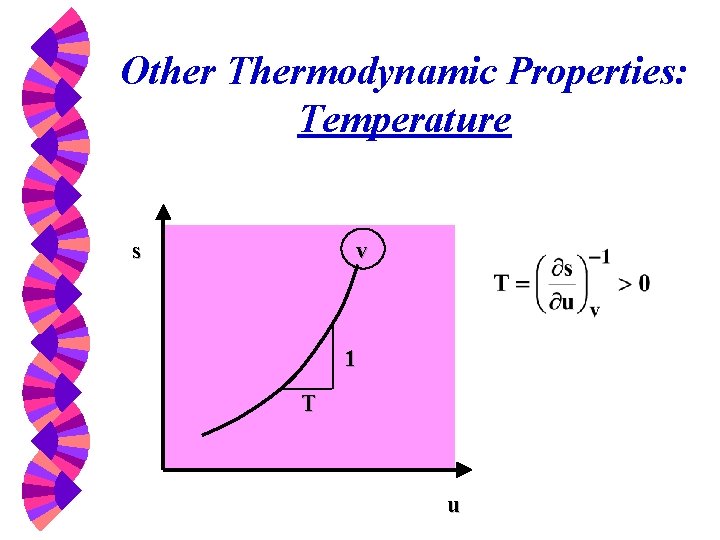
Other Thermodynamic Properties: Temperature v s 1 T u

Ideal Gases: u = u(T) 0 Therefore,

We can start with du and integrate to get the change in u: Note that Cv does change with temperature and cannot be automatically pulled from the integral.

Let’s look at enthalpy for an ideal gas: w h = u + Pv where Pv can be replaced by RT because Pv = RT. w Therefore, h = u + RT => since u is only a function of T, R is a constant, then h is also only a function of T w so h = h(T)

Similarly, for a change in enthalpy for ideal gases:

Summary: Ideal Gases w For ideal gases u, h, Cv, and Cp are functions of temperature alone. w For ideal gases, Cv and Cp are written in terms of ordinary differentials as
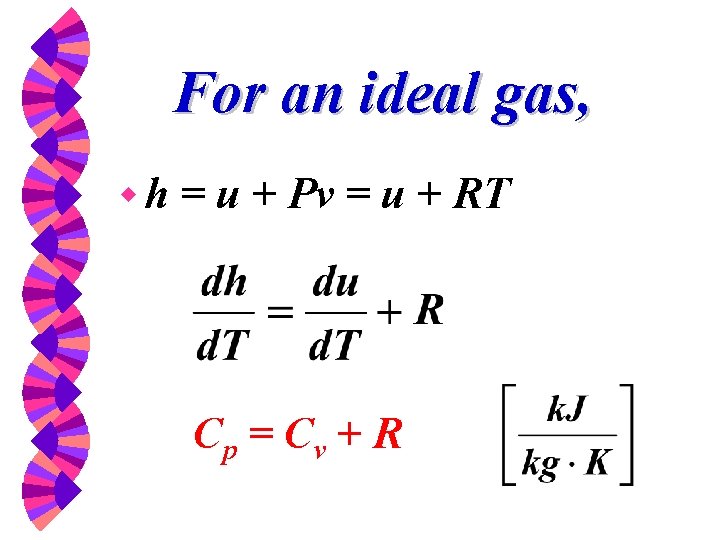
For an ideal gas, wh = u + Pv = u + RT Cp = Cv + R
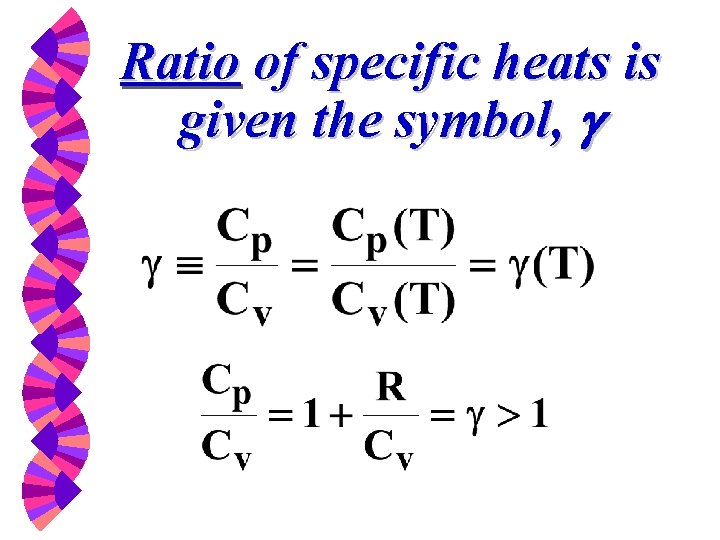
Ratio of specific heats is given the symbol, g

Other relations with the ratio of specific heats which can be easily developed:

For monatomic gases, Argon, Helium, and Neon

For all other gases, w Cp is a function of temperature and it may be calculated from equations such as those in Table A-5(c) in the Appendice w Cv may be calculated from Cp=Cv+R. w Next figure shows the temperature behavior …. specific heats go up with temperature.
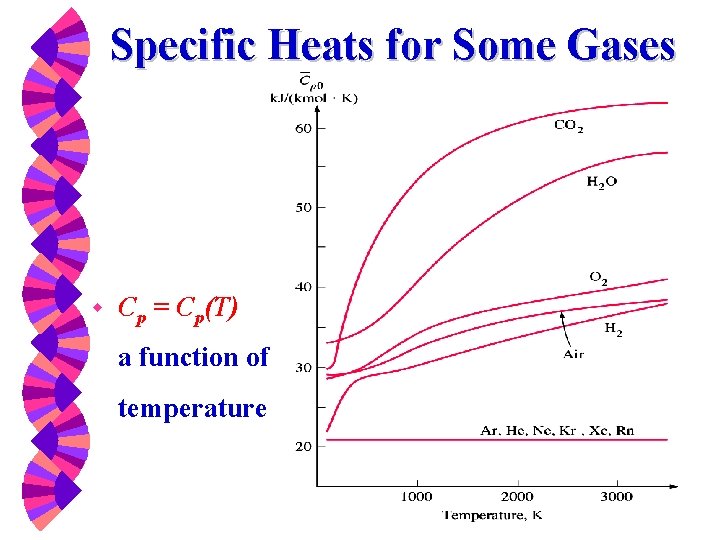
Specific Heats for Some Gases w Cp = Cp(T) a function of temperature

Three Ways to Calculate Δu and Δh w Δu = u 2 - u 1 (table) w Δh = h 2 - h 1 (table) w Δu = w Δh = w Δu = Cv, av ΔT w Δh = Cp, av ΔT
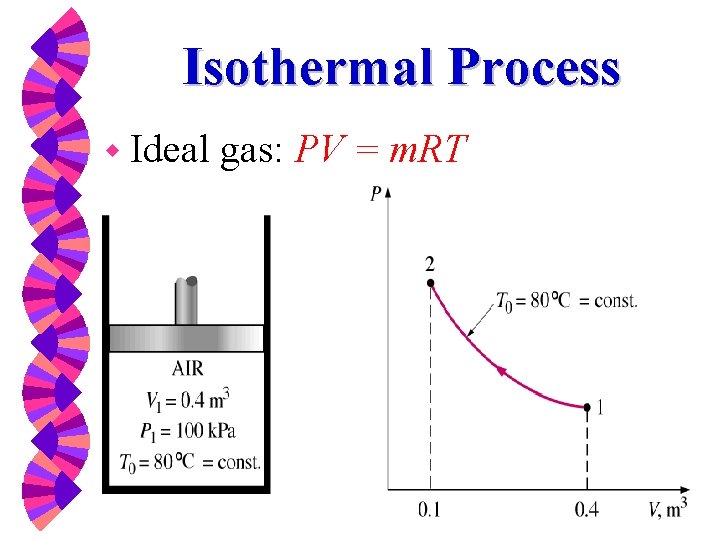
Isothermal Process w Ideal gas: PV = m. RT
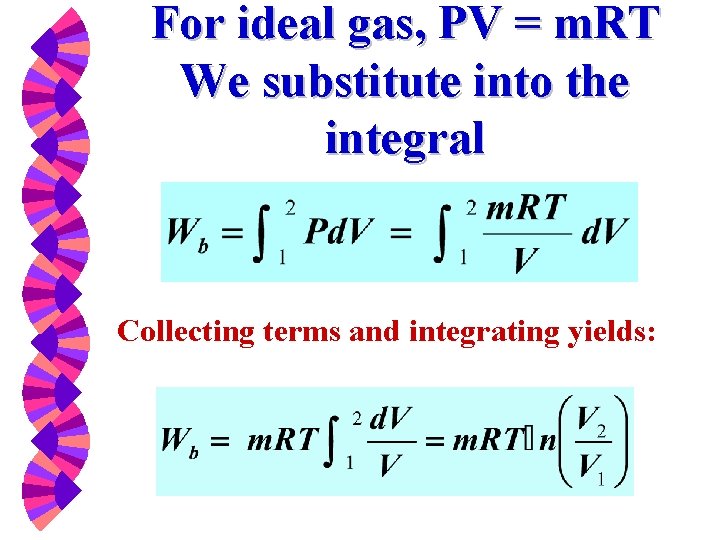
For ideal gas, PV = m. RT We substitute into the integral Collecting terms and integrating yields:
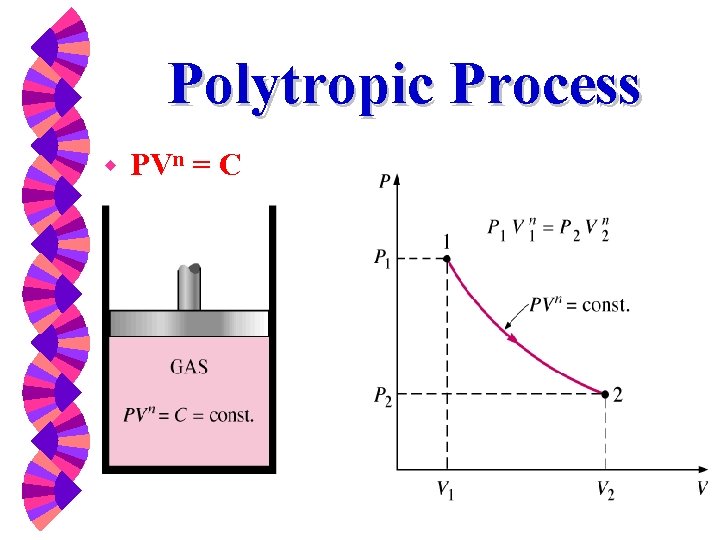
Polytropic Process w PVn = C

Ideal Gas Adiabatic Process and Reversible Work w w 1. 2. 3. 4. 5. What is the path for process with expand or contract without heat flux? How P, v and T behavior when Q = 0? To develop an expression to the adiabatic process is necessary employ: Reversible work mode: d. W = Pd. V Adiabatic hypothesis: d. Q =0 Ideal Gas Law: Pv=RT Specific Heat Relationships First Law Thermodynamics: d. Q-d. W=d. U

eal Gas Adiabatic Process and Reversible Wo First Law: Using P = MRT/V Integrating from (1) to (2)
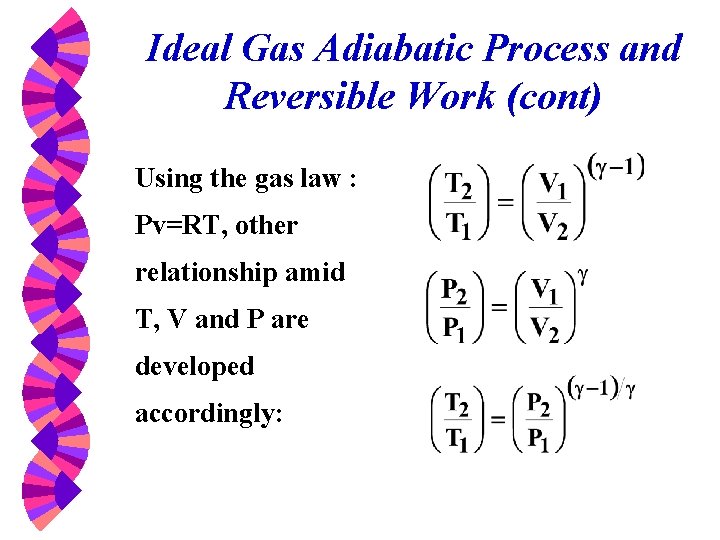
Ideal Gas Adiabatic Process and Reversible Work (cont) Using the gas law : Pv=RT, other relationship amid T, V and P are developed accordingly:
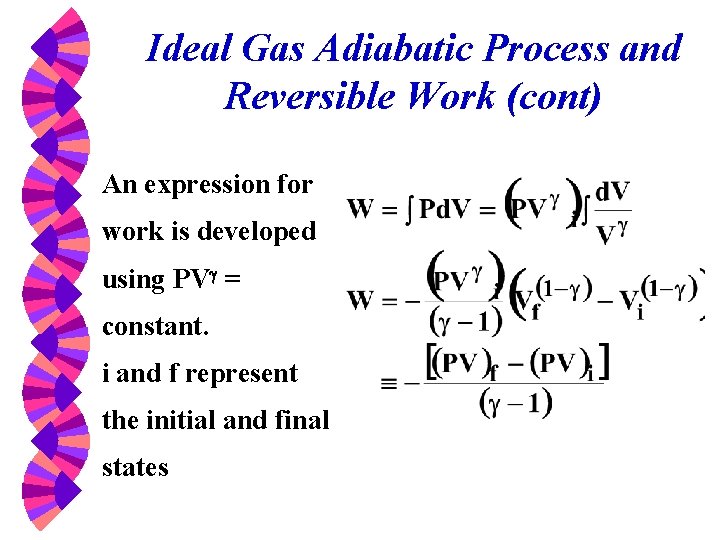
Ideal Gas Adiabatic Process and Reversible Work (cont) An expression for work is developed using PVg = constant. i and f represent the initial and final states
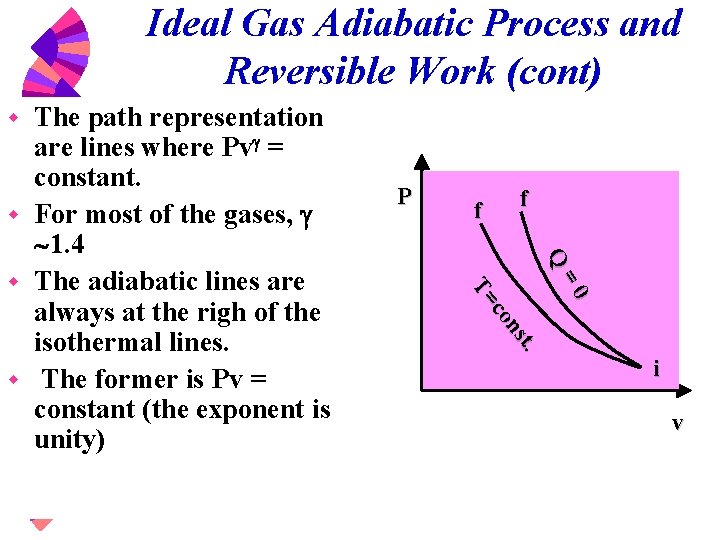
Ideal Gas Adiabatic Process and Reversible Work (cont) The path representation are lines where Pvg = constant. w For most of the gases, g 1. 4 w The adiabatic lines are always at the righ of the isothermal lines. w The former is Pv = constant (the exponent is unity) w P f f Q =0 t. ns co T= i v

Polytropic Process A frequently encountered process for gases is the polytropic process: PVn = constant Since this expression relates P & V, we can calculate the work for this path.

Polytropic Process Constant pressure w Constant volume w Isothermal & ideal gas w Adiabatic & ideal gas w Exponent n 0 1 k=Cp/Cv

Boundary work for a gas which obeys the polytropic equation

We can simplify it further n The constant c = P 1 V 1 = P 2 V 2 n

Polytropic Process
- Slides: 52