Thermodynamic data A tutorial course Session 8 Calculation























- Slides: 57

Thermodynamic data A tutorial course Session 8: Calculation of phase equilibria from critically assessed thermodynamic data Alan Dinsdale “Thermochemistry of Materials” SRC

Outline • Describe how phase diagrams can be calculated from critically assessed thermodynamic data – What are critically assessed data ? • How thermodynamic data are modelled – Temperature – Composition – Pressure • Databases and software • Applications 2

Why calculate phase diagrams ? • Provides way to rationalise different but related sets of experimental properties • . . . and to extrapolate thermodynamic data from small systems into higher order systems in such a way that they allow prediction of multicomponent phase equilibria • Now software and data are sufficiently robust for – monitoring and controlling industrial plant – choosing materials used in everyday household appliances – understanding the way in which mankind affects the world and the environment in which we live – devising how we can perhaps make the world a safer and better place for us and our children to live in 3

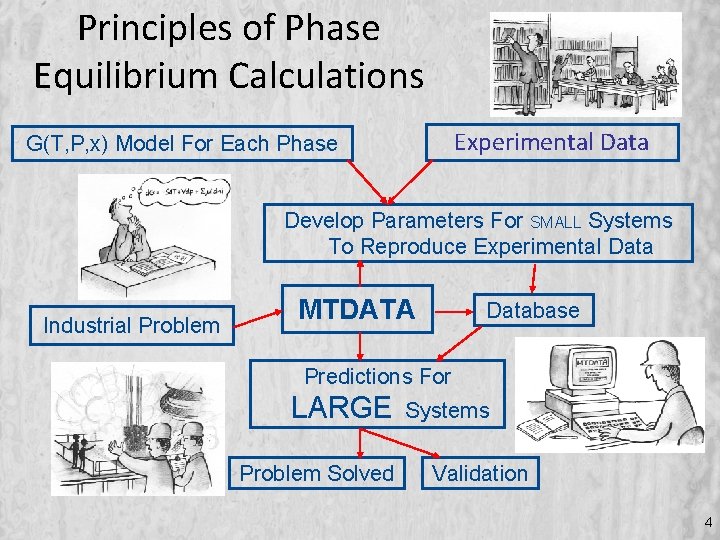
Principles of Phase Equilibrium Calculations Experimental Data G(T, P, x) Model For Each Phase Develop Parameters For SMALL Systems To Reproduce Experimental Data Industrial Problem MTDATA Database Predictions For LARGE Problem Solved Systems Validation 4

In a nutshell

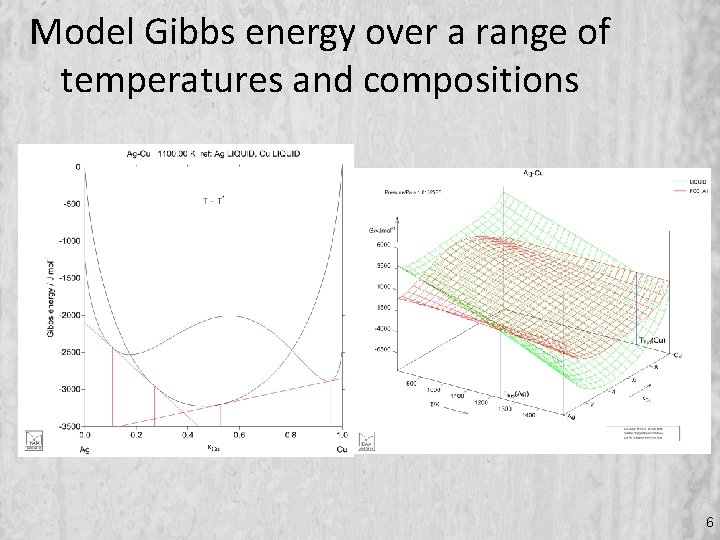
Model Gibbs energy over a range of temperatures and compositions 6

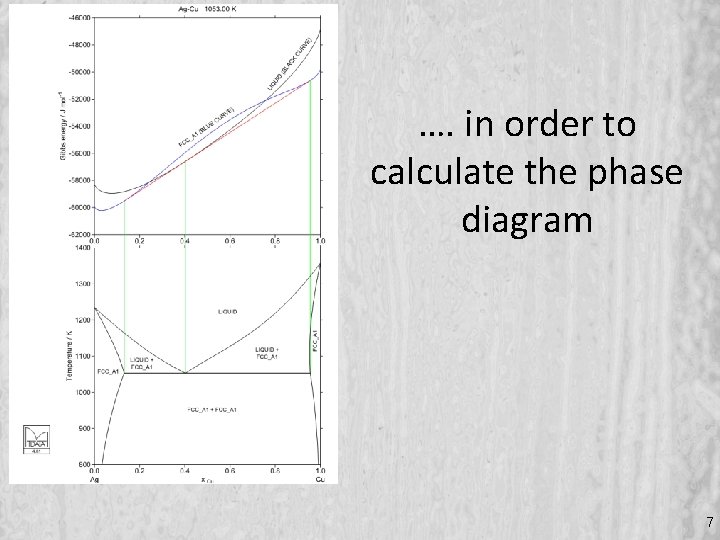
…. in order to calculate the phase diagram 7

What are critically assessed data ?

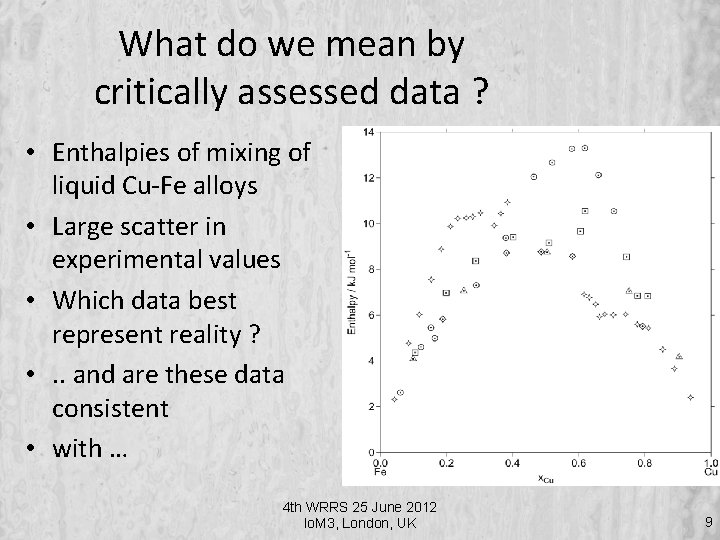
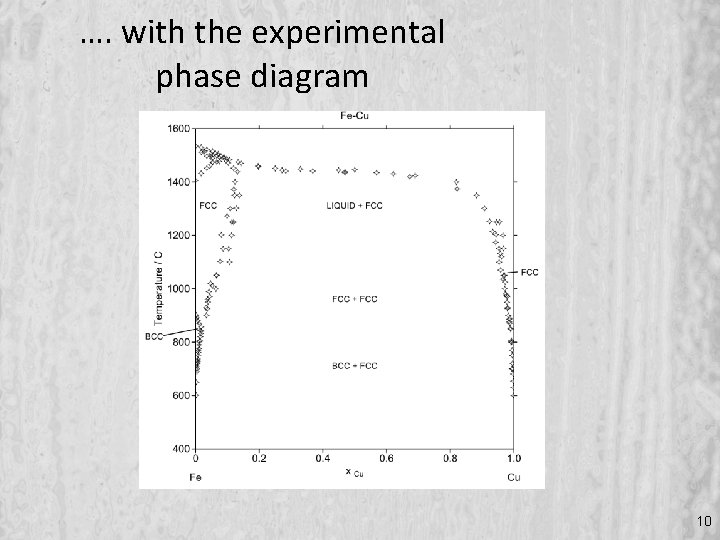
What do we mean by critically assessed data ? • Enthalpies of mixing of liquid Cu-Fe alloys • Large scatter in experimental values • Which data best represent reality ? • . . and are these data consistent • with … 4 th WRRS 25 June 2012 Io. M 3, London, UK 9

…. with the experimental phase diagram 10

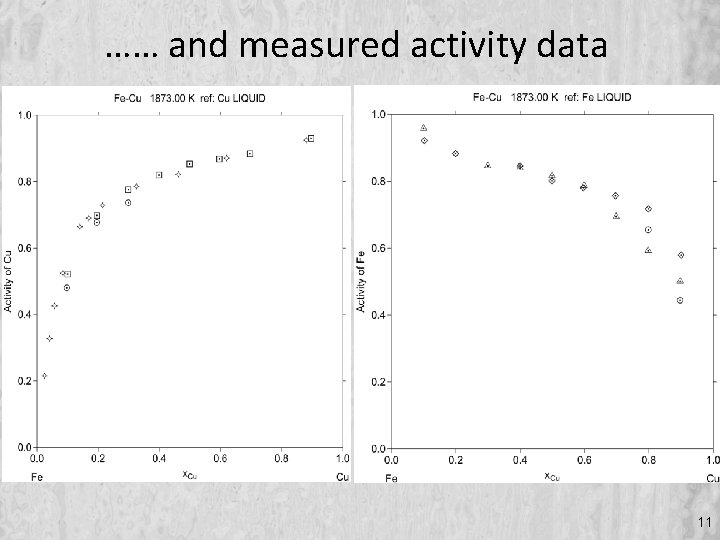
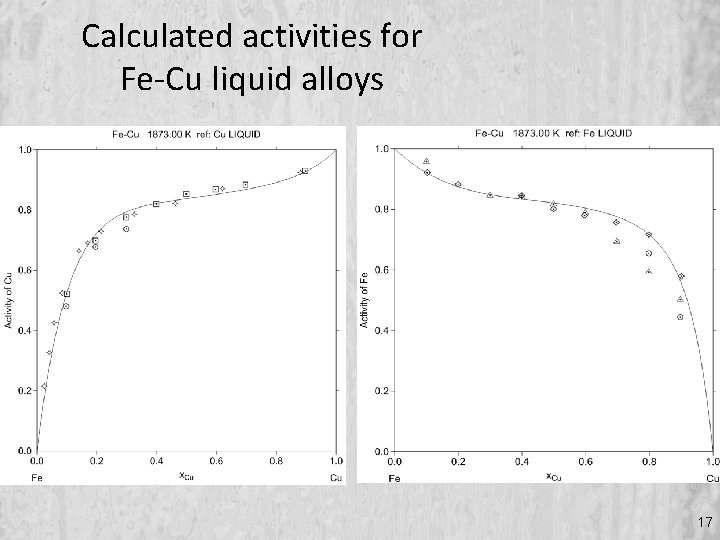
…… and measured activity data 11

What is the aim of a critical assessment ? • To generate a set of reliable data or diagrams which are self consistent and represent all the available experimental data • Involves a critical analysis of the experimental data • …. . followed by a computer based optimisation process to reduce the experimental data into a small number of model parameters • …. . using rigorous theoretical basis underlying thermodynamics 12

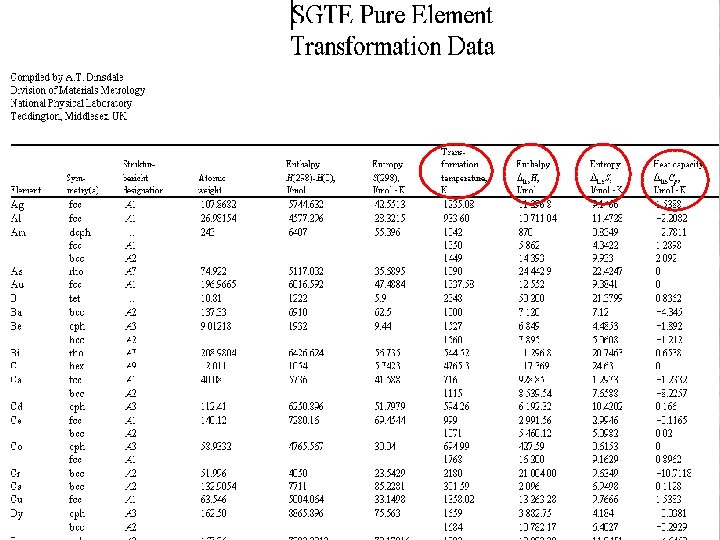
Critical analysis of experimental data • Experimental data: Search and analysis – Search through standard compilations eg Hultgren, Massalski – Use a database of references to the literature eg Cheynet – Carry out a full literature search • Which properties – Phase diagram information • Liquidus / solidus temperatures • Solubilities – Thermodynamic information • • • enthalpies of mixing vapour pressure data emf data heat capacities Enthalpies of transformation Ab-initio calculations 13

Obtaining model parameters • Aim is to determine set of coefficients which gives best agreement with experimental data – by least-squares fitting of thermodynamic functions to selected set of experimental and ab-initio data • It is usually carried out with the assistance of a computer – Using optimisation software • • MTDATA optimisation module PARROT (inside Thermo. Calc) LUKAS program BINGSS CHEMOPT 14

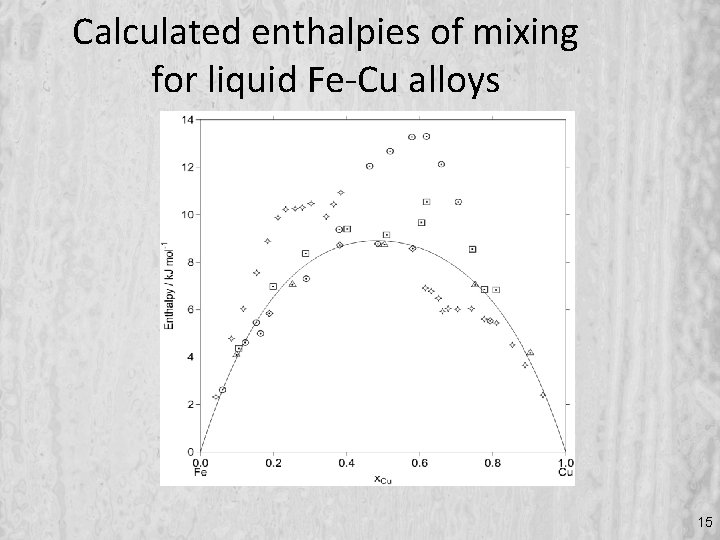
Calculated enthalpies of mixing for liquid Fe-Cu alloys 15

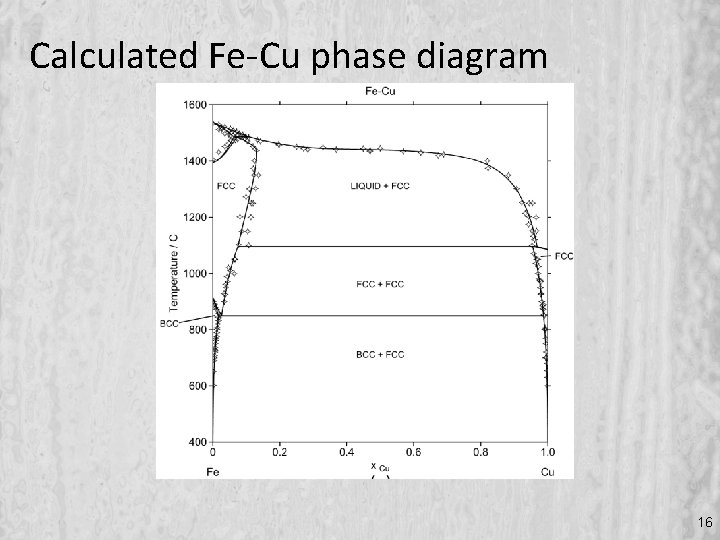
Calculated Fe-Cu phase diagram 16

Calculated activities for Fe-Cu liquid alloys 17

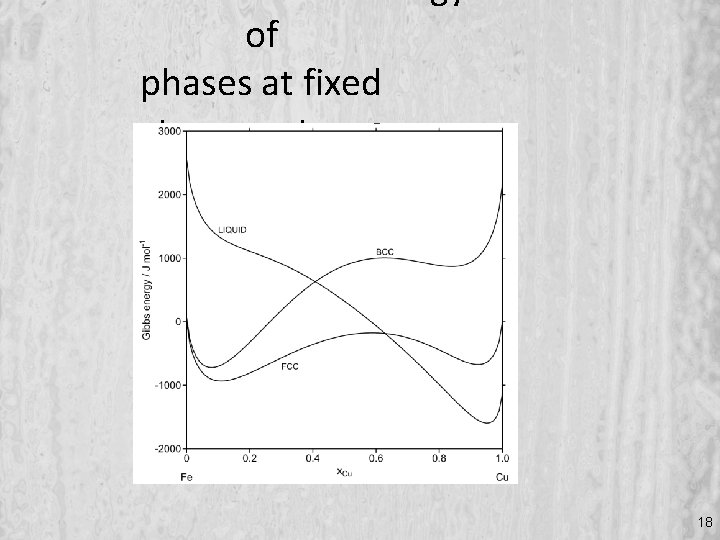
of phases at fixed temperature 18

How to model thermodynamic data need to understand how G changes with T, P and x

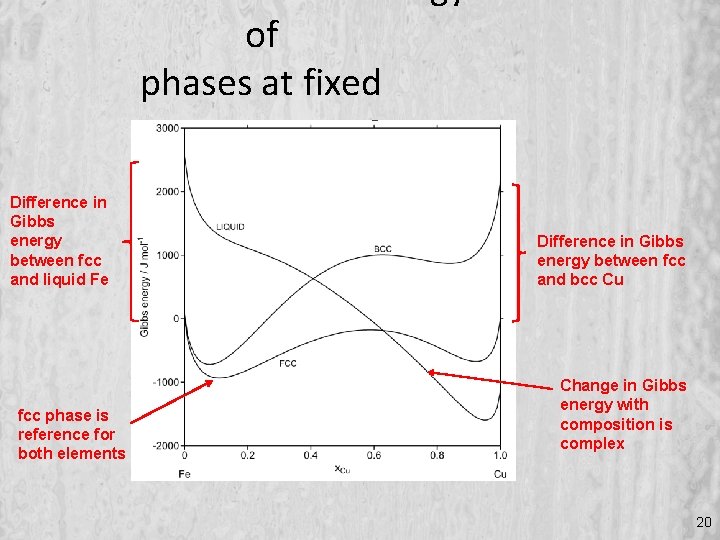
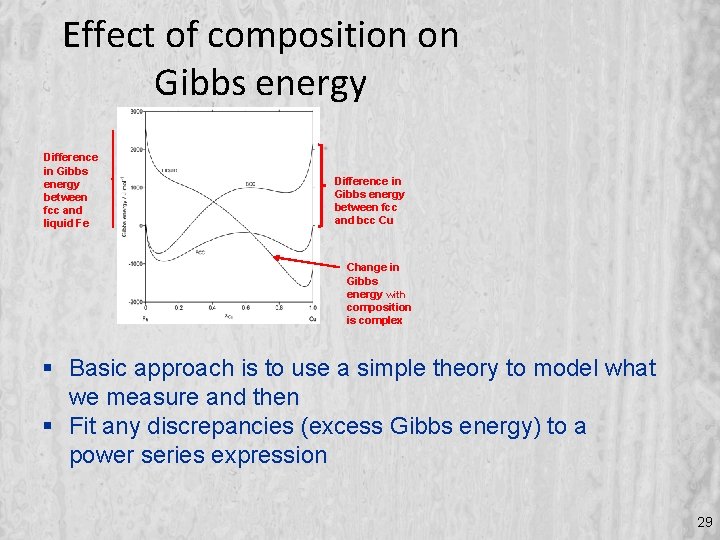
of phases at fixed temperature Difference in Gibbs energy between fcc and liquid Fe fcc phase is reference for both elements Difference in Gibbs energy between fcc and bcc Cu Change in Gibbs energy with composition is complex 20

4 th WRRS 25 June 2012 Io. M 3, London, UK 21

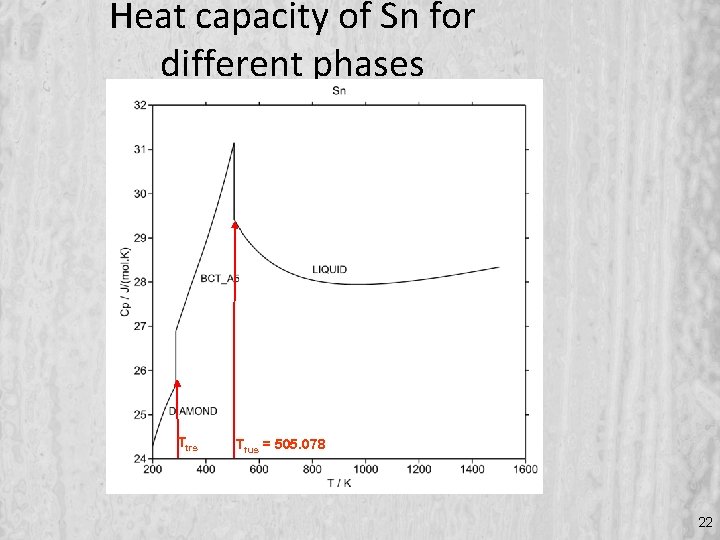
Heat capacity of Sn for different phases Ttrs Tfus = 505. 078 22

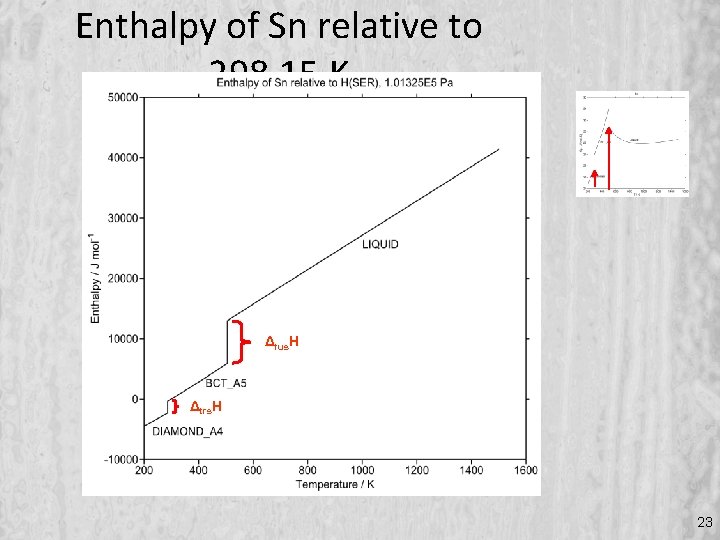
Enthalpy of Sn relative to 298. 15 K Δfus. H Δtrs. H 23

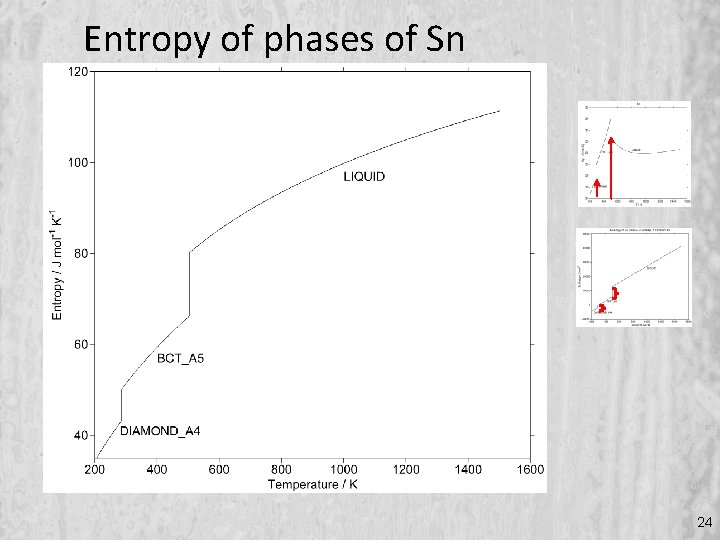
Entropy of phases of Sn 24

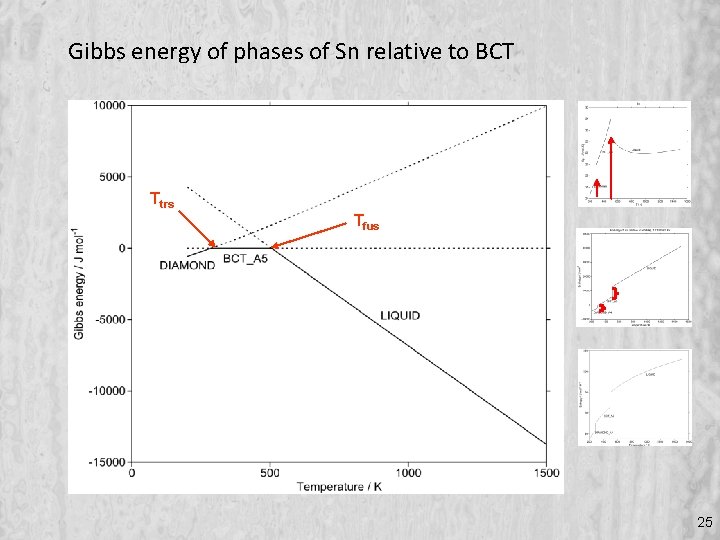
Gibbs energy of phases of Sn relative to BCT Ttrs Tfus 25

Mathematical description of Gibbs energy variation with temperature • Heat capacity generally represented by 1 or more expressions of the form: – Cp = a + b T + c T 2 + d T-2 (generally obtained from experiment) • With enthalpy of formation and entropies at 298. 15 K (or transition enthalpies and entropies) this leads to expressions for the Gibbs energy of the form: – G = A + B T + C T ln(T) + D T 2 + ET 3 + F T-1 – (relative to some defined reference state) 26

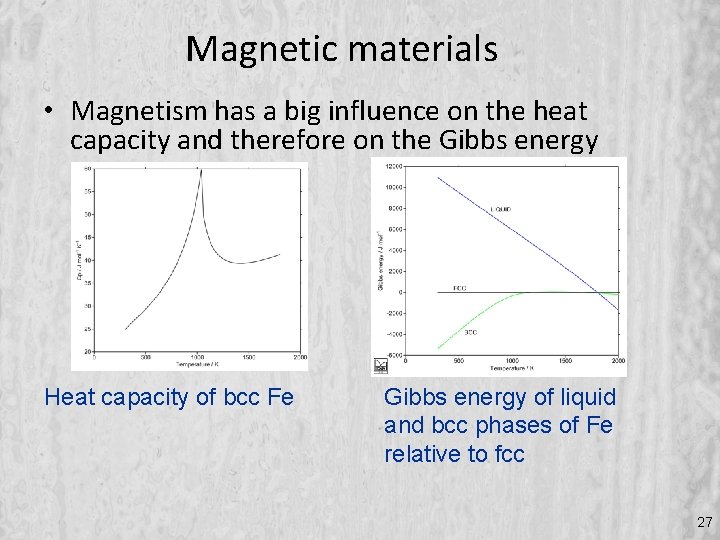
Magnetic materials • Magnetism has a big influence on the heat capacity and therefore on the Gibbs energy Heat capacity of bcc Fe Gibbs energy of liquid and bcc phases of Fe relative to fcc 27

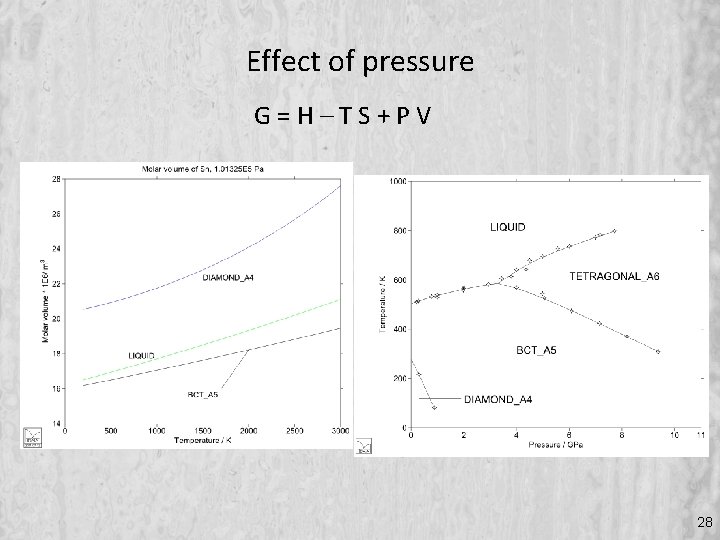
Effect of pressure G=H–TS+PV 28

Effect of composition on Gibbs energy Difference in Gibbs energy between fcc and liquid Fe Difference in Gibbs energy between fcc and bcc Cu Change in Gibbs energy with composition is complex Basic approach is to use a simple theory to model what we measure and then Fit any discrepancies (excess Gibbs energy) to a power series expression 29

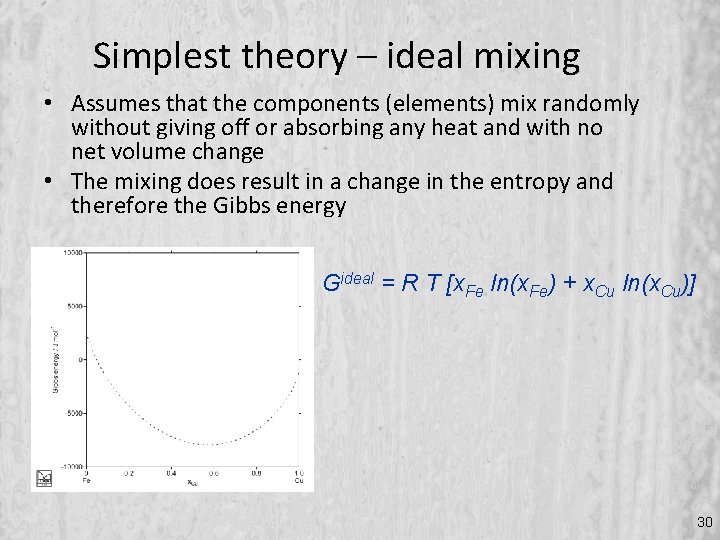
Simplest theory – ideal mixing • Assumes that the components (elements) mix randomly without giving off or absorbing any heat and with no net volume change • The mixing does result in a change in the entropy and therefore the Gibbs energy Gideal = R T [x. Fe ln(x. Fe) + x. Cu ln(x. Cu)] 30

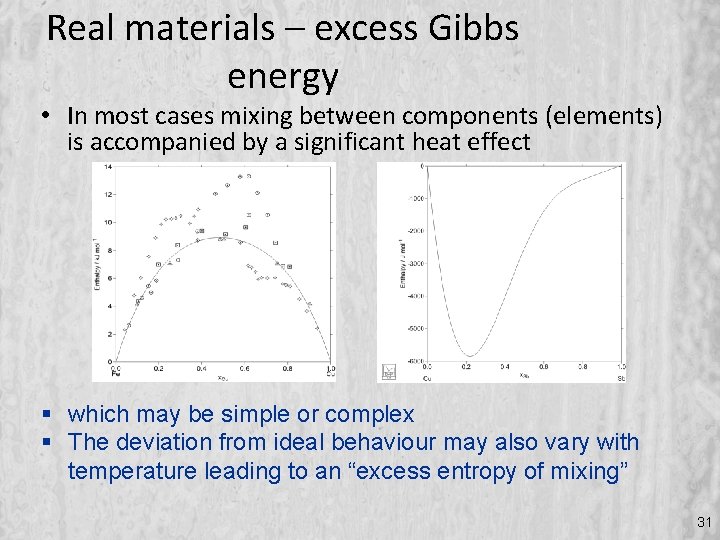
Real materials – excess Gibbs energy • In most cases mixing between components (elements) is accompanied by a significant heat effect which may be simple or complex The deviation from ideal behaviour may also vary with temperature leading to an “excess entropy of mixing” 31

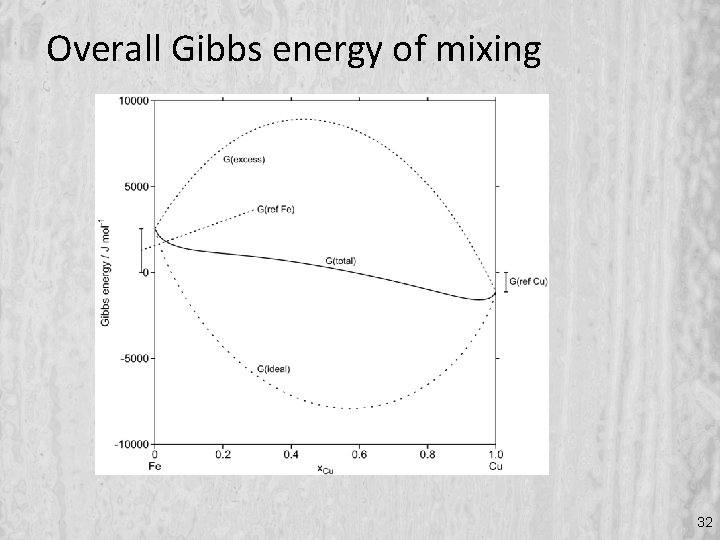
Overall Gibbs energy of mixing 32

Gibbs energy of binary solutions Pure component Gibbs energies G= x. Fe GFe + x. Cu GCu + R T [ x. Fe ln(x. Fe) + x. Cu ln(x. Cu)] + x. Cu x. Fe [ a + b (x. Cu-x. Fe) + c (x. Cu-x. Fe)2 + d (x. Cu-x. Fe)3 + …. . ] Ideal contribution to Gibbs energy Excess Gibbs energy – in this case “Redlich. Kister expression” where a, b, c, d …. could be temperature dependent (in practice for Fe-Cu we may need only one or possibly two parameters) 33

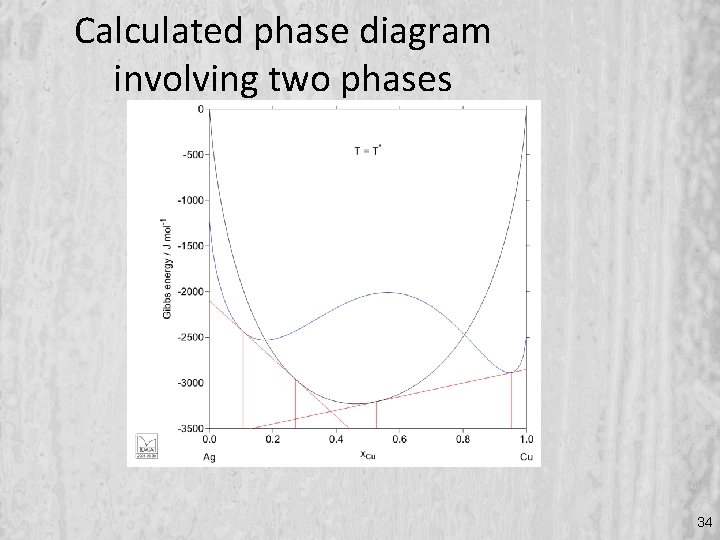
Calculated phase diagram involving two phases 34


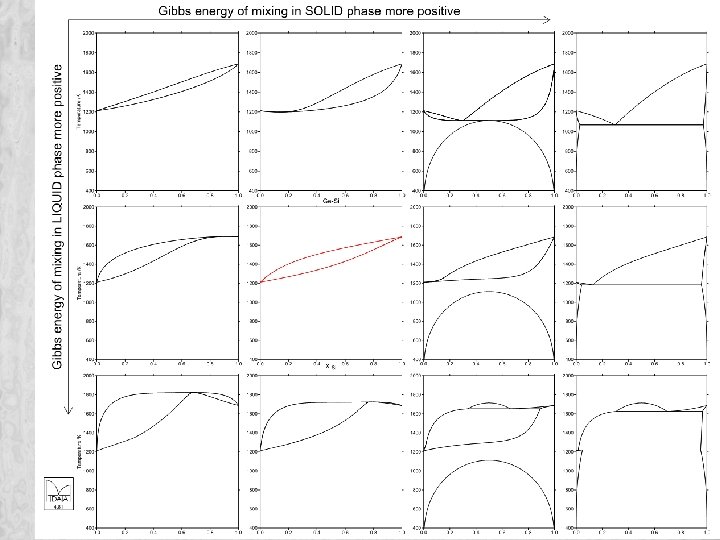
From binary to multicomponent • Multicomponent Gibbs energy given by G= Σ xi Gi + R T Σ xi ln(xi) + Gexcess Various models used to extrapolate excess Gibbs energy into ternary and higher order systems from data for binary systems. Extra ternary terms used if required Kohler Muggianu Toop 36

Compound phases • Stoichiometric phases: variation of Gibbs energy with T similar to that for phases of elements • Many important compound phases are stable over ranges of homogeneity. Crystal structure indicates sublattices with preferred occupancy. eg: sigma, mu, gamma brass – • Use compound energy formalism to allow mixing on different sites – – – (C, Va)1 Laves phases: (Cu, Mg)2 (Cu, Mg)1 Interstitial solution of carbon: (Cr, Fe)1 Spinels: (Fe 2+, Fe 3+)1 (Fe 2+, Fe 3+)2 (O 2 -)4 37

Gibbs energy using compound energy formalism eg (Cu, Mg) 2 1 • Gibbs energy again has 3 contributions • Pure compounds with element from each sublattice Cu: Cu, Cu: Mg, Mg: Cu, Mg: Mg • Ideal mixing of elements on each sublattice • Cu and Mg on first and on second sublattices • Non-ideal interaction between the elements on each sublattice but with a specific element on the other sublattice – Cu, Mg: Cu Cu, Mg: Mg Cu: Cu, Mg Mg: Cu, Mg • 38

Thermodynamic databases and software

Thermodynamic databases • Two types of databases – Collections of datasets suitable for potentially diverse materials eg SGSUB, SGSOL – Application specific databases: comprehensive set of data for particular material types • All databases need to be based upon a set of standards – Consistent data for the elements – Consistent models to represent thermodynamic data based (where possible) on crystallographic information 40

Database providers • Software developers – MTDATA (NPL) – Thermo. Calc (TCSAB) – Fact. Sage (GTT, Ecole Polytechnique Montreal – PANDAT (Compu. Therm) – JMat. Pro (Thermo. Tech) • Specialist centres eg SGTE, CEA, Tohoku, Claustal • International Collaborative Projects – COST 507, COST 531, COST MP 0602 – Superdata, III-V semiconductor project 41

Software packages General phase diagram calculations • Thermo. Calc: TCSAB, Stockholm, Sweden • Fact. Sage: GTT, Herzogenrath, Germany • Pandat: Compu. Therm, Madison, USA • MTDATA: NPL, Teddington, UK Calculation of materials properties • JMat. Pro: Sente Software Ltd, Guildford, UK 42

General facilities available • Single calculation of phase equilibria for defined temperature, pressure and composition • Calculations over a range of temperatures for fixed composition • Mass of phases in equilibrium (and compositions) • Partial pressures • Enthalpy changes and heat capacity • Transformation temperatures • Phase diagram calculations (binary, ternary, isopleths, liquidus projections) • Database management • Assessment and optimisation capability • Simple simulations (eg Scheil solidification) 43

Applications

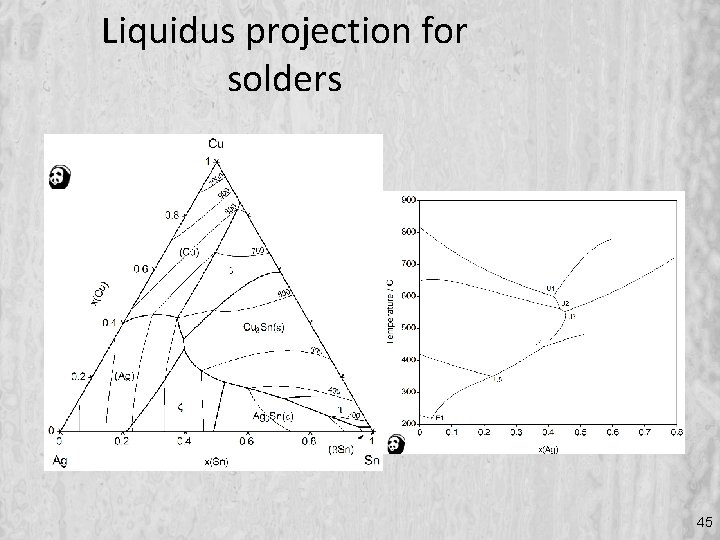
Liquidus projection for solders 45

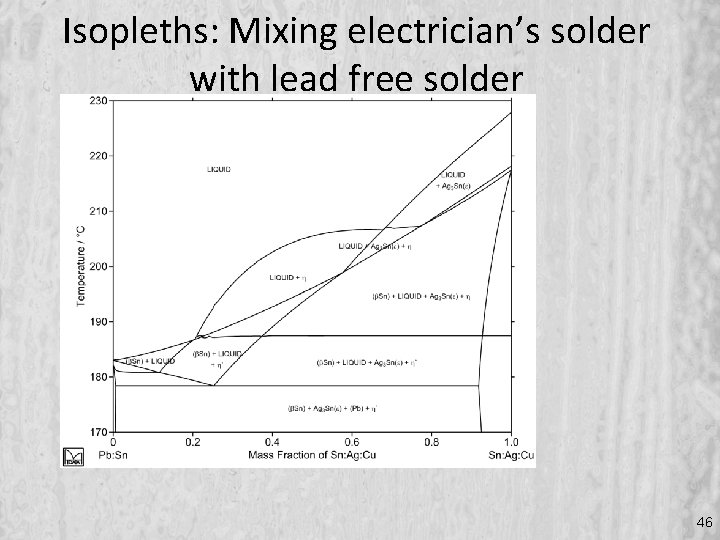
Isopleths: Mixing electrician’s solder with lead free solder 46

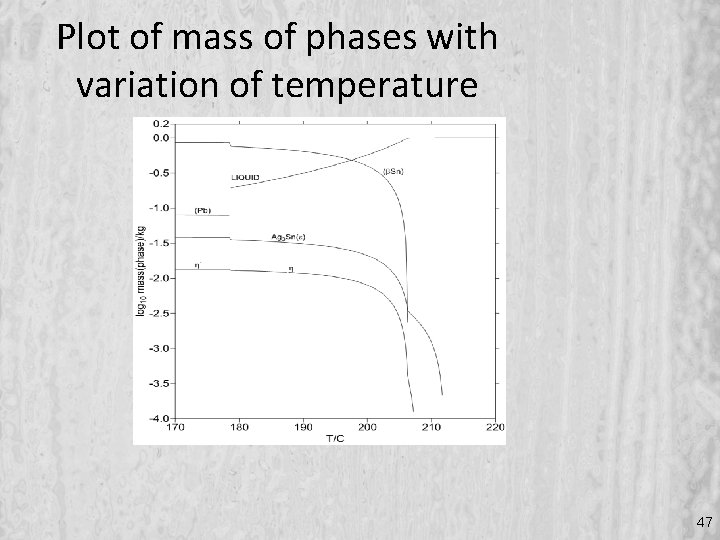
Plot of mass of phases with variation of temperature 47

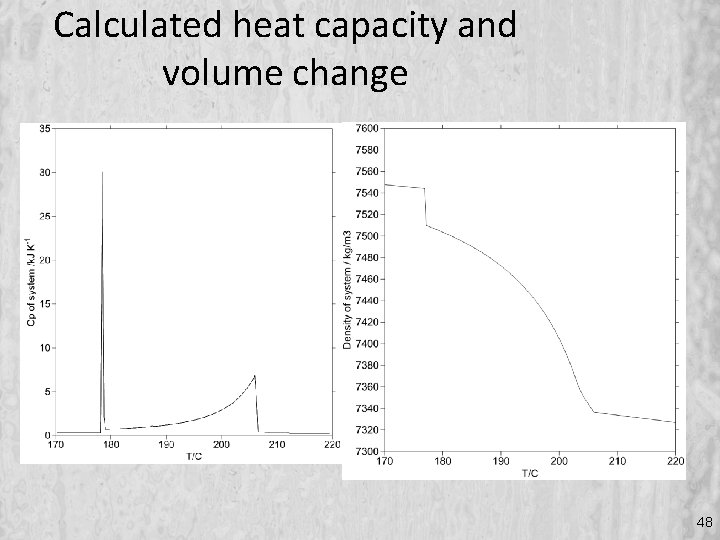
Calculated heat capacity and volume change 48

Use of thermodynamic data to predict thermophysical properties

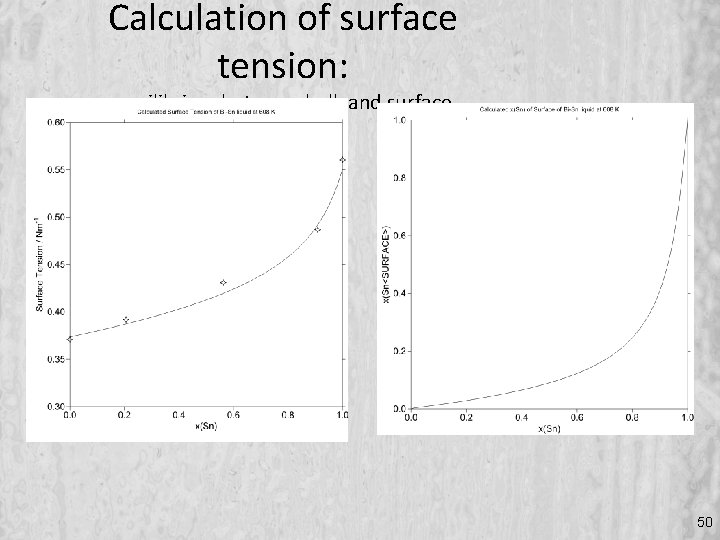
Calculation of surface tension: equilibrium between bulk and surface 50

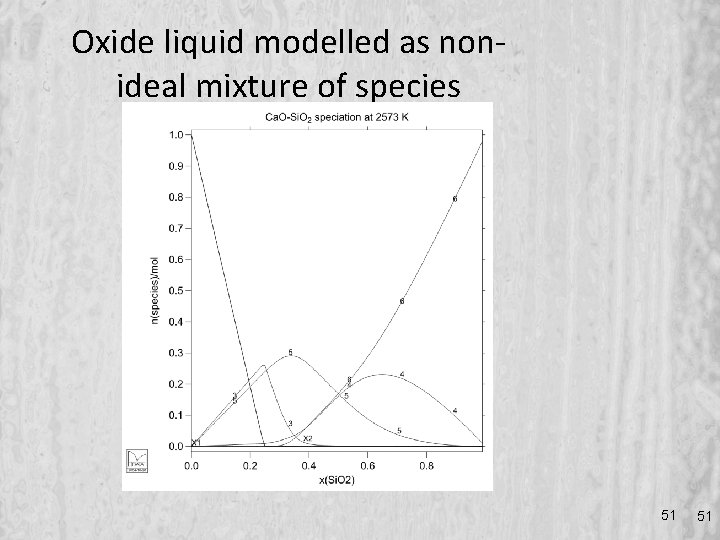
Oxide liquid modelled as nonideal mixture of species 51 51

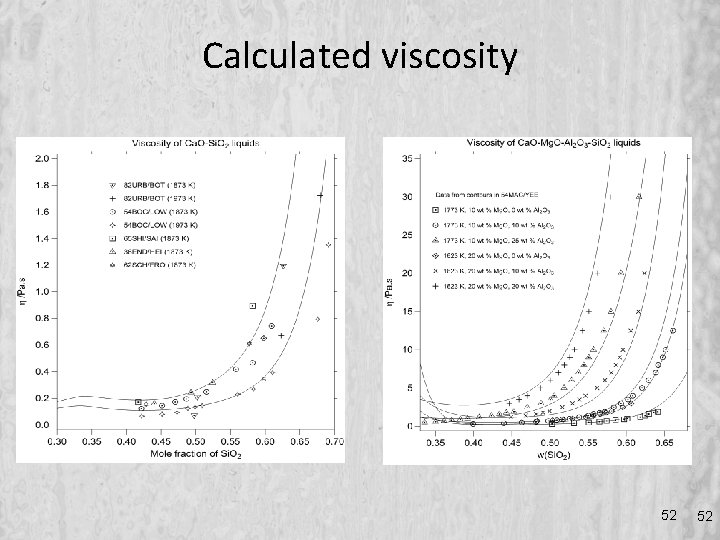
Calculated viscosity 52 52

Engineering toolkits

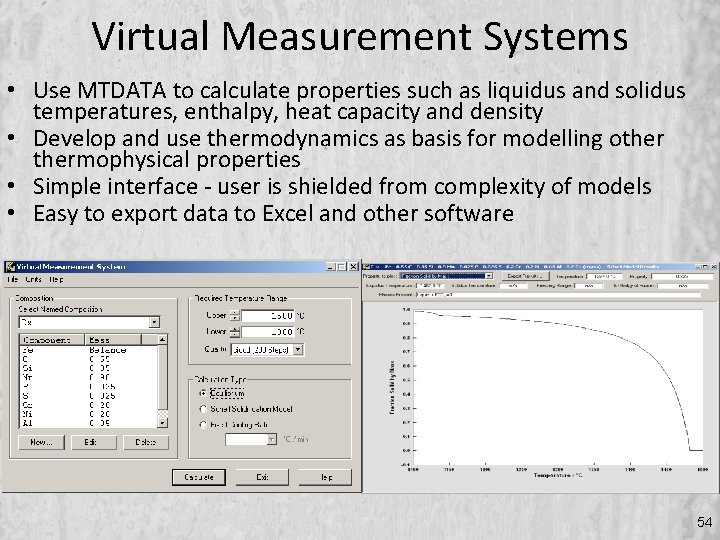
Virtual Measurement Systems • Use MTDATA to calculate properties such as liquidus and solidus temperatures, enthalpy, heat capacity and density • Develop and use thermodynamics as basis for modelling othermophysical properties • Simple interface - user is shielded from complexity of models • Easy to export data to Excel and other software 54

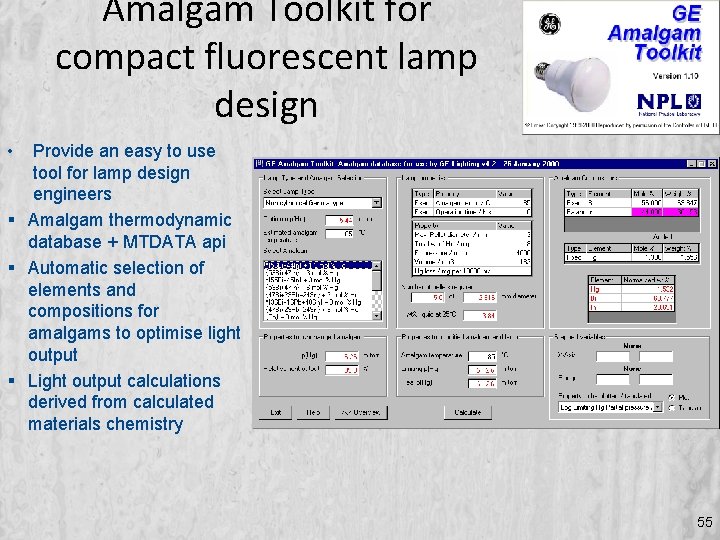
Amalgam Toolkit for compact fluorescent lamp design • Provide an easy to use tool for lamp design engineers Amalgam thermodynamic database + MTDATA api Automatic selection of elements and compositions for amalgams to optimise light output Light output calculations derived from calculated materials chemistry 55

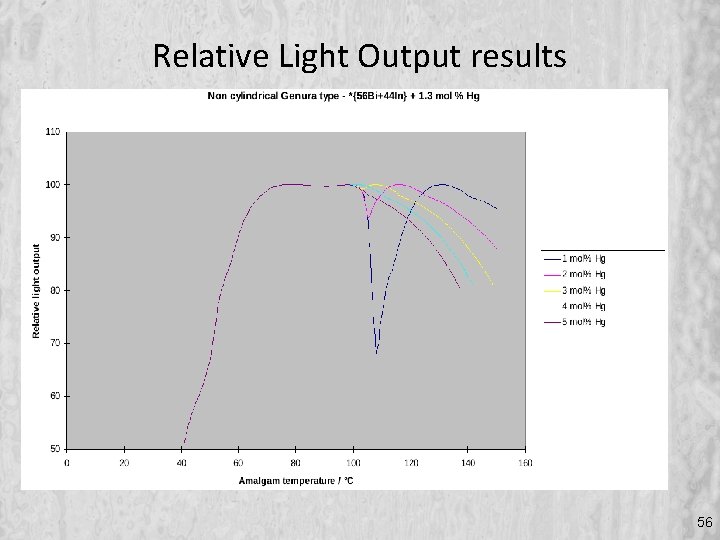
Relative Light Output results 56

Final thoughts • Now possible to calculate phase equilibria for a wide range of materials • Rely on good quality critically assessed data compiled into comprehensive databases • A number of organisations are involved in developing database • Other sorts of properties can be modelled from a thermodynamic basis • Engineering toolkits offer potential to bring phase diagram calculations to the non specialist 57