THERMOCHEMISTRY Thermodynamics ENTHALPY CHANGES be able to define

THERMOCHEMISTRY Thermodynamics

ENTHALPY CHANGES be able to define enthalpy and enthalpy change (ΔH) be able to define standard enthalpy chemical reactions involve the making and breaking of bonds be able to determine bond enthalpies from data use mean bond enthalpies to estimate ΔH for reactions be able to calculate enthalpy change from experimental data use Hess’s law to calculate enthalpy changes in reactions
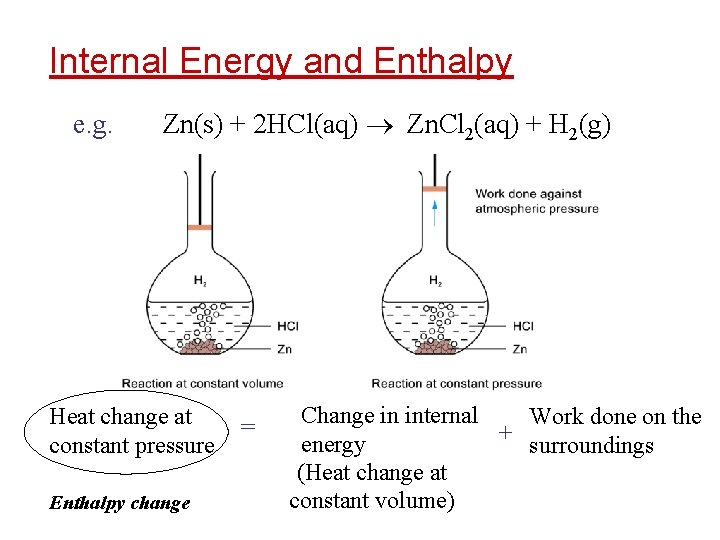
Internal Energy and Enthalpy e. g. Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2(g) Heat change at constant pressure Enthalpy change = Change in internal Work done on the + energy surroundings (Heat change at constant volume)

First Law THERMODYNAMICS Energy can be neither created nor destroyed but It can be converted from one form to another Second Law Energy flows from hot to cold Third Law A perfectly ordered crystal has zero entropy. Chemical reactions either absorb or give off energy Exothermic Energy is given out Endothermic Energy is absorbed Every Day Examples Exothermic combustion of fuels respiration (oxidation of carbohydrates) Endothermic photosynthesis thermal decomposition of calcium carbonate
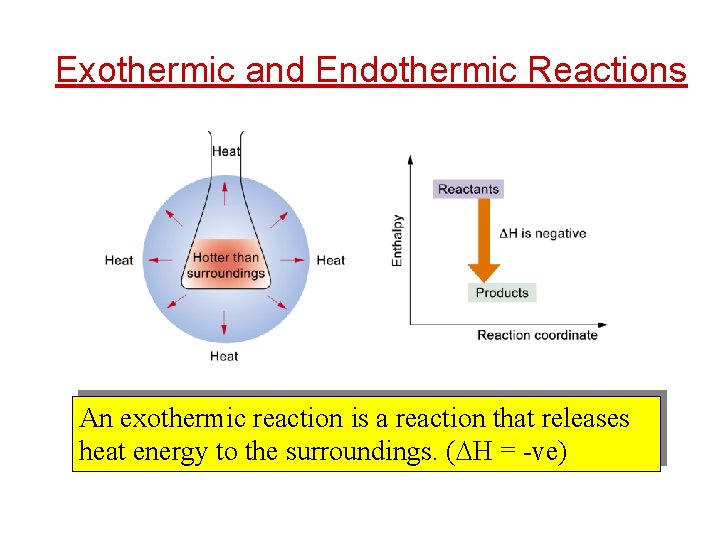
Exothermic and Endothermic Reactions An exothermic reaction is a reaction that releases heat energy to the surroundings. ( H = -ve)
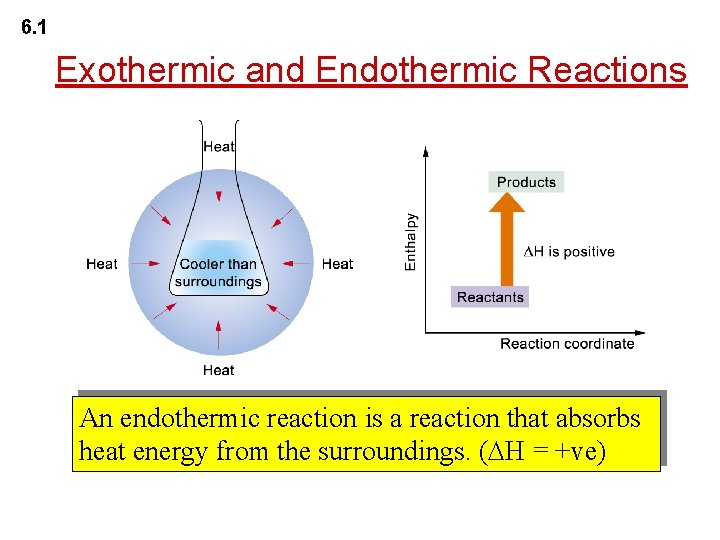
6. 1 Exothermic and Endothermic Reactions An endothermic reaction is a reaction that absorbs heat energy from the surroundings. ( H = +ve)
Bond breaking - endothermic Energy is always required to be inputted to break a bond. Bond breaking is always endothermic.

Bond making - exothermic • Energy is always released when a bond is formed. Bond making is always exothermic.
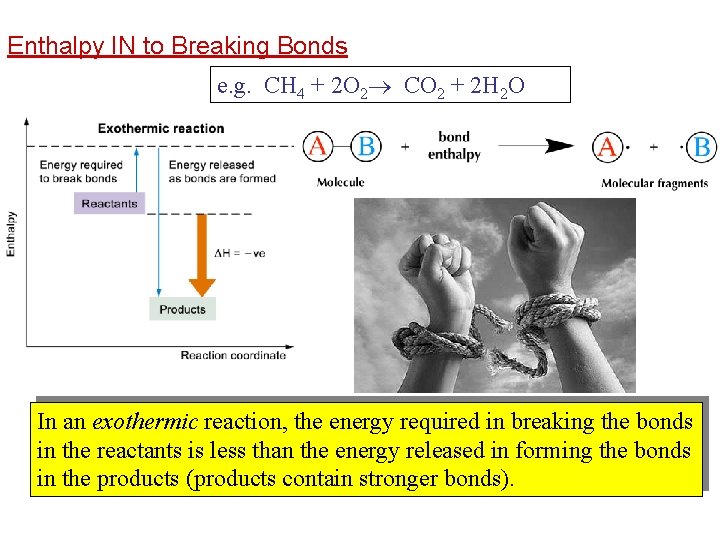
Enthalpy IN to Breaking Bonds e. g. CH 4 + 2 O 2 CO 2 + 2 H 2 O In an exothermic reaction, the energy required in breaking the bonds in the reactants is less than the energy released in forming the bonds in the products (products contain stronger bonds).
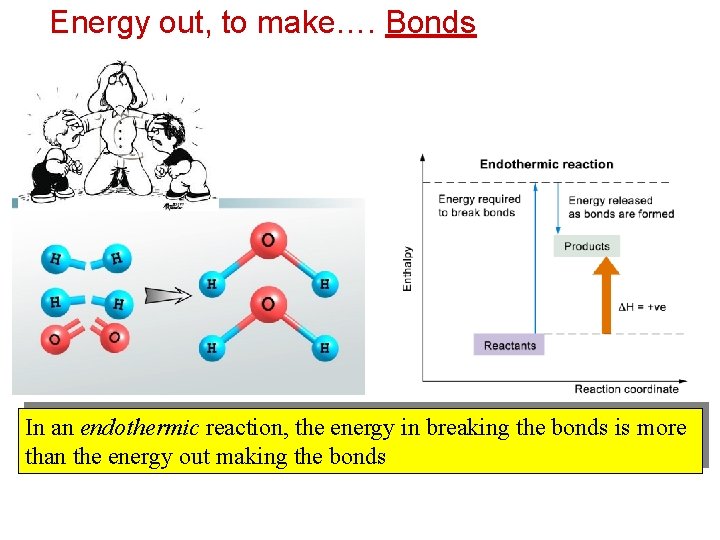
Energy out, to make…. Bonds In an endothermic reaction, the energy in breaking the bonds is more than the energy out making the bonds
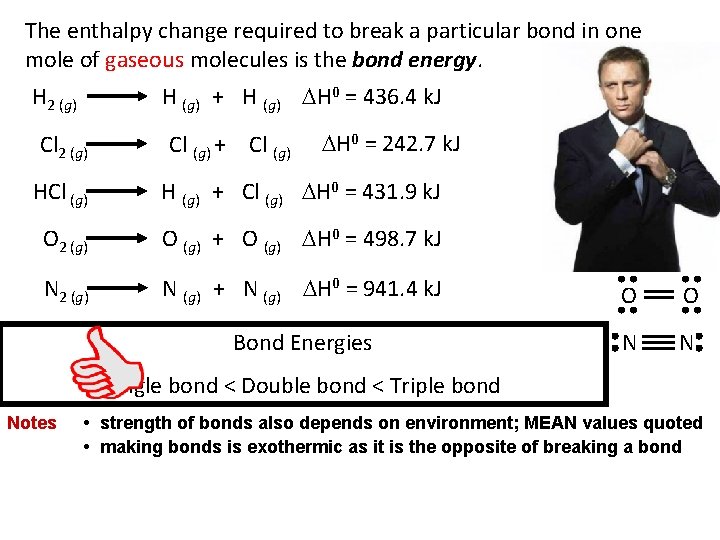
The enthalpy change required to break a particular bond in one mole of gaseous molecules is the bond energy. H (g) + H (g) H 0 = 436. 4 k. J H 2 (g) Cl (g) + Cl (g) H 0 = 242. 7 k. J HCl (g) H (g) + Cl (g) H 0 = 431. 9 k. J O 2 (g) O (g) + O (g) H 0 = 498. 7 k. J N 2 (g) N (g) + N (g) H 0 = 941. 4 k. J O O Bond Energies N N Single bond < Double bond < Triple bond Notes • strength of bonds also depends on environment; MEAN values quoted • making bonds is exothermic as it is the opposite of breaking a bond

Calculation of enthalpy of reaction from bond energies Only the net number and types of bonds broken (red bonds above) and formed (blue bonds above) are included in the calculation.

Do now! Let, play, Mr. Bond Before you have a cow
Combustion of methane CH 4(g) + 2 O 2(g) 2 H 2 O(l) + CO 2(g) + ENERGY
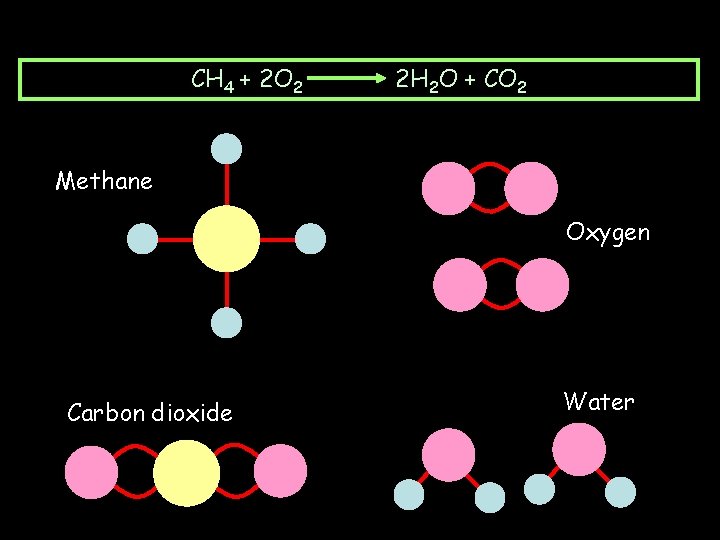
Burning Methane CH 4 + 2 O 2 2 H 2 O + CO 2 Methane Oxygen Carbon dioxide Water
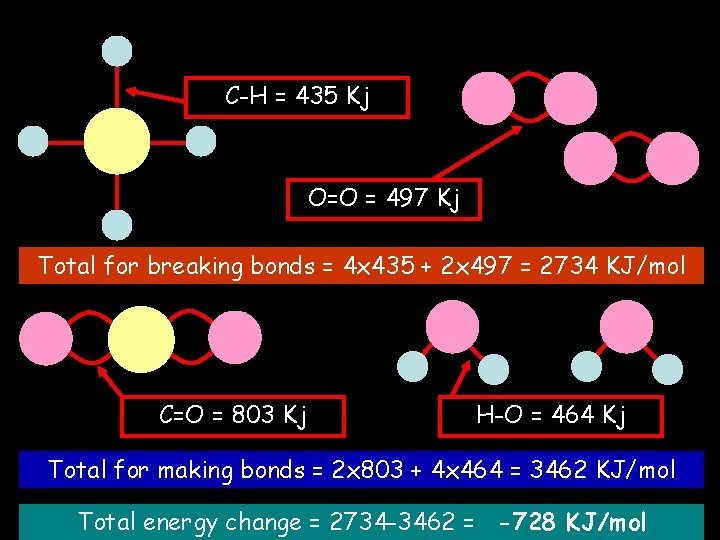
Bond energies C-H = 435 Kj O=O = 497 Kj Total for breaking bonds = 4 x 435 + 2 x 497 = 2734 KJ/mol C=O = 803 Kj H-O = 464 Kj Total for making bonds = 2 x 803 + 4 x 464 = 3462 KJ/mol Total energy change = 2734 -3462 = -728 KJ/mol
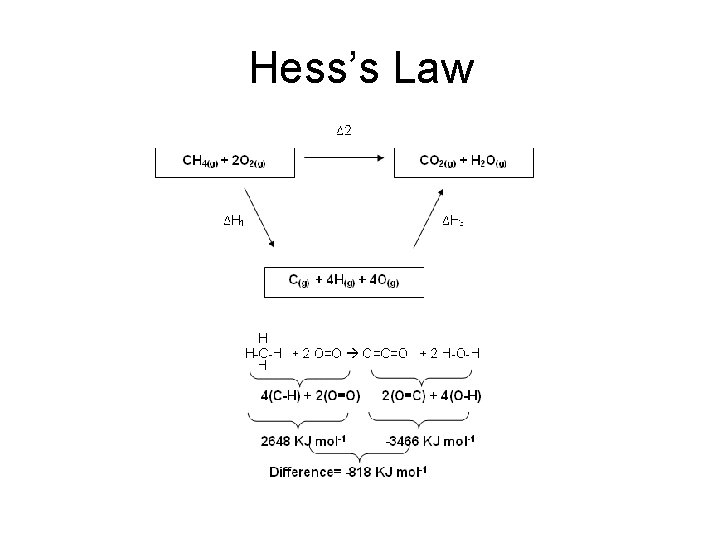
Hess’s Law
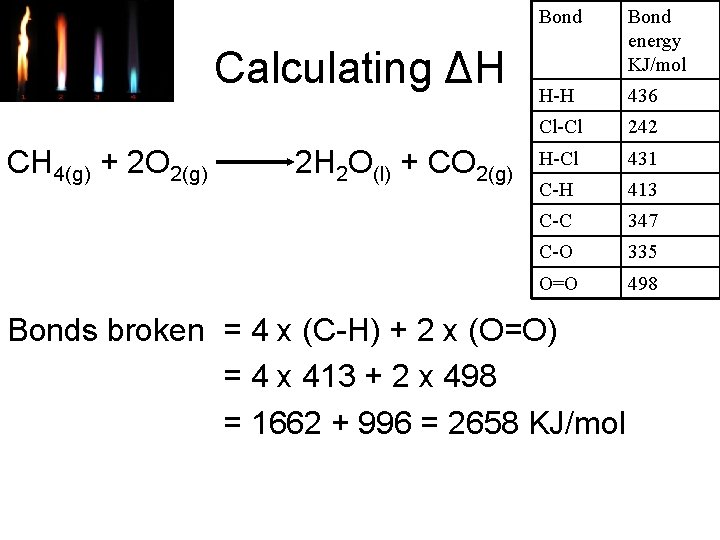
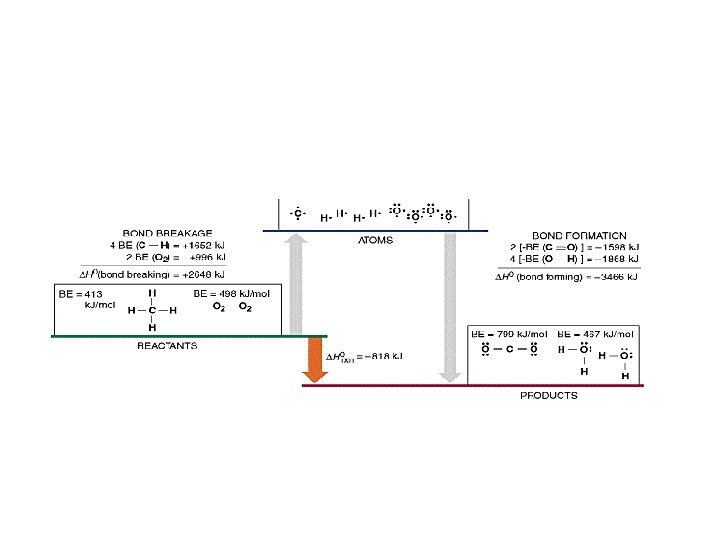
Calculating ΔH CH 4(g) + 2 O 2(g) 2 H 2 O(l) + CO 2(g) Bond energy KJ/mol H-H 436 Cl-Cl 242 H-Cl 431 C-H 413 C-C 347 C-O 335 O=O 498 Bonds broken = 4 x (C-H) + 2 x (O=O) = 4 x 413 + 2 x 498 = 1662 + 996 = 2658 KJ/mol
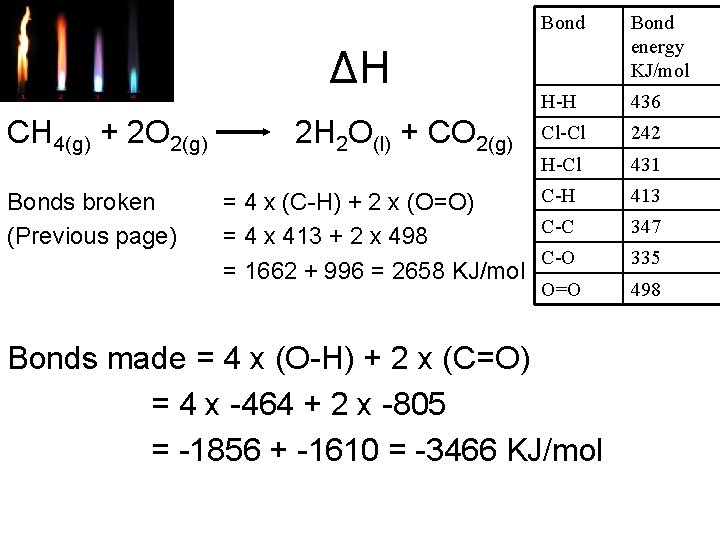
Bond energy KJ/mol H-H 436 Cl-Cl 242 H-Cl 431 C-H = 4 x (C-H) + 2 x (O=O) C-C = 4 x 413 + 2 x 498 C-O = 1662 + 996 = 2658 KJ/mol 413 ΔH CH 4(g) + 2 O 2(g) Bonds broken (Previous page) 2 H 2 O(l) + CO 2(g) O=O Bonds made = 4 x (O-H) + 2 x (C=O) = 4 x -464 + 2 x -805 = -1856 + -1610 = -3466 KJ/mol 347 335 498
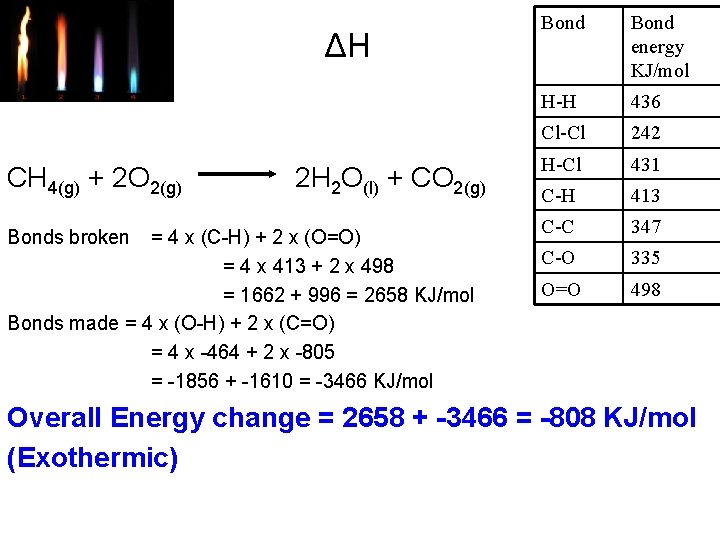
ΔH CH 4(g) + 2 O 2(g) Bonds broken 2 H 2 O(l) + CO 2(g) = 4 x (C-H) + 2 x (O=O) = 4 x 413 + 2 x 498 = 1662 + 996 = 2658 KJ/mol Bonds made = 4 x (O-H) + 2 x (C=O) = 4 x -464 + 2 x -805 = -1856 + -1610 = -3466 KJ/mol Bond energy KJ/mol H-H 436 Cl-Cl 242 H-Cl 431 C-H 413 C-C 347 C-O 335 O=O 498 Overall Energy change = 2658 + -3466 = -808 KJ/mol (Exothermic)

Standard Enthalpy Changes CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g) H = -808 k. J mol-1 CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) H = -890 k. J mol-1 As enthalpy changes depend on temperature and pressure, chemists find it convenient to report enthalpy changes based on an internationally agreed set standard conditions: 1. elements or compounds in their normal physical states; 2. a pressure of 1 atm (101. 325 KPa); and 3. a temperature of 250 C (298 K) ø Enthalpy change under standard conditions denoted by symbol: H
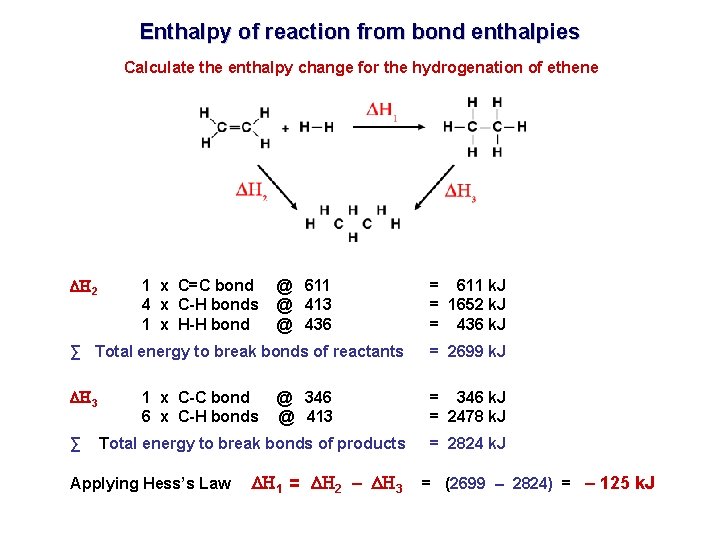
Enthalpy of reaction from bond enthalpies Calculate the enthalpy change for the hydrogenation of ethene H 2 1 x C=C bond 4 x C-H bonds 1 x H-H bond @ 611 @ 413 @ 436 = 611 k. J = 1652 k. J = 436 k. J ∑ Total energy to break bonds of reactants = 2699 k. J H 3 = 346 k. J = 2478 k. J ∑ 1 x C-C bond 6 x C-H bonds @ 346 @ 413 Total energy to break bonds of products Applying Hess’s Law H 1 = H 2 – H 3 = 2824 k. J = (2699 – 2824) = – 125 k. J
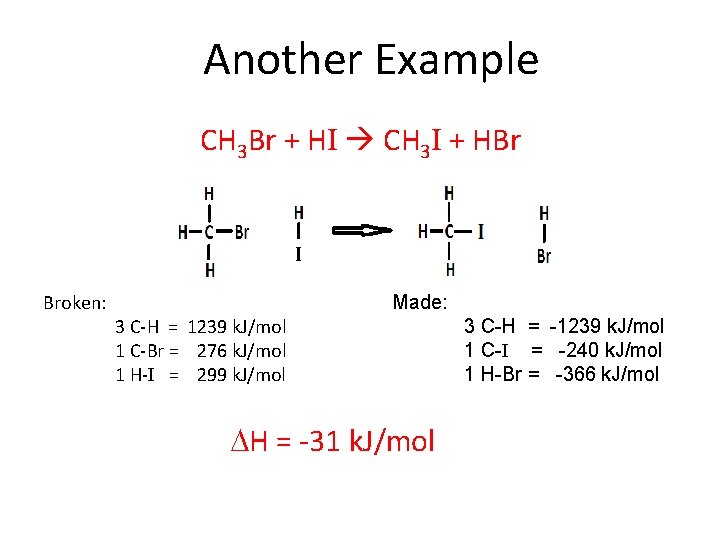
Another Example CH 3 Br + HI CH 3 I + HBr Broken: Made: 3 C-H = 1239 k. J/mol 1 C-Br = 276 k. J/mol 1 H-I = 299 k. J/mol H = -31 k. J/mol 3 C-H = -1239 k. J/mol 1 C-I = -240 k. J/mol 1 H-Br = -366 k. J/mol

HESS’S LAW “The enthalpy change is independent of the path taken” How The enthalpy change going from A to B can be found by adding the values of the enthalpy changes for the reactions A to X, X to Y and Y to B. Hr = H 1 + H 2 + H 3 If you go in the opposite direction of an arrow, you subtract the value of the enthalpy change. e. g. H 2 = - H 1 + Hr - H 3

Standard Enthalpy Changes of Reactions Standard Enthalpy Change of Neutralization ø The standard enthalpy change of neutralization ( Hneut) is the enthalpy change when one mole of water is formed from the neutralization of an acid by an alkali under standard conditions. Hneut = -57. 3 k. J mol-1 ø e. g. H+(aq) + OH-(aq) H 2 O(l) e. g. The standard enthalpy change of neutralization between HNO 3 and Na. OH is -57. 3 k. J mol-1 e. g. The standard enthalpy change of neutralization between HCl and Na. OH is -57. 1 k. J mol-1
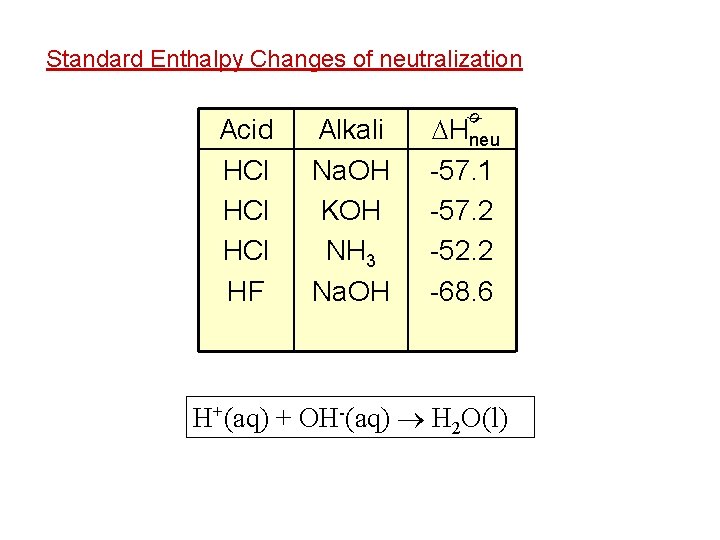
Standard Enthalpy Changes of neutralization Alkali Na. OH KOH NH 3 Na. OH Hneu -57. 1 -57. 2 -52. 2 -68. 6 ø Acid HCl HCl HF H+(aq) + OH-(aq) H 2 O(l)

Standard Enthalpy Changes of Reactions Standard Enthalpy Change of Solution ø The standard enthalpy change of solution ( Hsoln) is the enthalpy change when one mole of a solute is dissolved to form an infinitely dilute solution under standard conditions. ø e. g. Na. Cl(s) + water Na+(aq)+Cl-(aq) Hsoln=+ 3. 9 k. J mol-1 ø e. g. Li. Cl(s) + water Li+(aq) + Cl-(aq) Hsoln= -37. 2 k. J mol-1 Note that enthalpy changes of solution associate with physical changes.

Standard Enthalpy Changes of solution Hsoln(k. J mol-1) -44. 7 +3. 9 -57. 8 +20. 0 ø Salt Na. OH Na. Cl KOH KBr

Standard Enthalpy Changes of Reactions Standard Enthalpy Change of Formation ø The standard enthalpy change of formation ( Hf) is the enthalpy change of the reaction when one mole of the compound in its standard state is formed from its constituent elements under standard conditions. 2 Na(s) + Cl 2(g) 2 Na. Cl(s) 1 mole Hf = -411 k. J mol-1 ø Na(s) + ½Cl 2(g) Na. Cl(s) H = -822 k. J mol-1 ø e. g. Standard enthalpy change of formation of Na. Cl is -411 k. J mol-1.
![Standard Enthalpy Changes of Reactions ø What is Hf [N 2(g)] ? N 2(g) Standard Enthalpy Changes of Reactions ø What is Hf [N 2(g)] ? N 2(g)](http://slidetodoc.com/presentation_image_h2/870da92f7e54884425ce0586b71fdeb2/image-31.jpg)
Standard Enthalpy Changes of Reactions ø What is Hf [N 2(g)] ? N 2(g) ø Hf [N 2(g)] = 0 The enthalpy change of formation of an element is always zero.

STANDARD ENTHALPY OF FORMATION Definition The enthalpy change when ONE MOLE of a compound is formed in its standard state from its elements in their standard states. Symbol H°f Example(s) This little f is VERY important C(graphite) + O 2(g) ———> CO 2(g) H 2(g) + ½O 2(g) ———> 2 C(graphite) + ½O 2(g) + Notes H 2 O(l) 3 H 2(g) ———> C 2 H 5 OH(l) Only ONE MOLE of product on the RHS of the equation Elements In their standard states have zero enthalpy of formation.

Enthalpies of Formation Using Enthalpies of Formation, Calculate Enthalpies of Reaction • For a reaction (Method #2) • Note: n & m are stoichiometric coefficients. • Calculate heat of reaction for the combustion of methane gas giving carbon dioxide and water. CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g)

CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g) ∆Hof (k. J/mol): Try this

One more time!!!! C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) + ? ? ?
![Hess’s Law Second Method Hreaction reactants Hf [products] ø ø - Hf [reactants] Methane Hess’s Law Second Method Hreaction reactants Hf [products] ø ø - Hf [reactants] Methane](http://slidetodoc.com/presentation_image_h2/870da92f7e54884425ce0586b71fdeb2/image-36.jpg)
Hess’s Law Second Method Hreaction reactants Hf [products] ø ø - Hf [reactants] Methane and oxygen destroyed products elements CO 2 and water formed ø ø Hreaction = Hf [products] - Hf [reactants]

Standard Enthalpy Changes of Reactions Standard Enthalpy Change of Combustion e. g. C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) H 1 = -2220 k. J 2 C 3 H 8(g) + 10 O 2(g) 6 CO 2(g) + 8 H 2 O(l) H 2 = ? H 2 = -4440 k. J It is more convenient to report enthalpy changes per mole of the main reactant reacted/product formed. ANY IDEAS WHAT IT WOULD BE FOR THE FORMATION OF 18. 02 g OF WATER?

Standard Enthalpy Changes of Reactions Standard Enthalpy Change of Combustion ø The standard enthalpy change of combustion ( Hc) of a substance is the enthalpy change when one mole of the substance burns completely under standard conditions. e. g. C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) ø Hc = -2220 k. J mol-1 1 mole The standard enthalpy change of combustion of propane is -2220 k. J mol-1

rd 3 Method For Heats of Rx Really this is NOT another law, it is still Hess’s Law BUT It looks different, yet we get to the same place , the same answer. There is an old Chinese proverb which says: There are many ways to the top of a mountain, but the view from the top is always the same.

Develop an analogy for soccer and scoring a goal.
Develop an analogy for soccer and scoring a goal.

Hess’s Law Flip Method https: //www. youtube. com/watch? v=9 oa. Nm. Eq. Y-ss
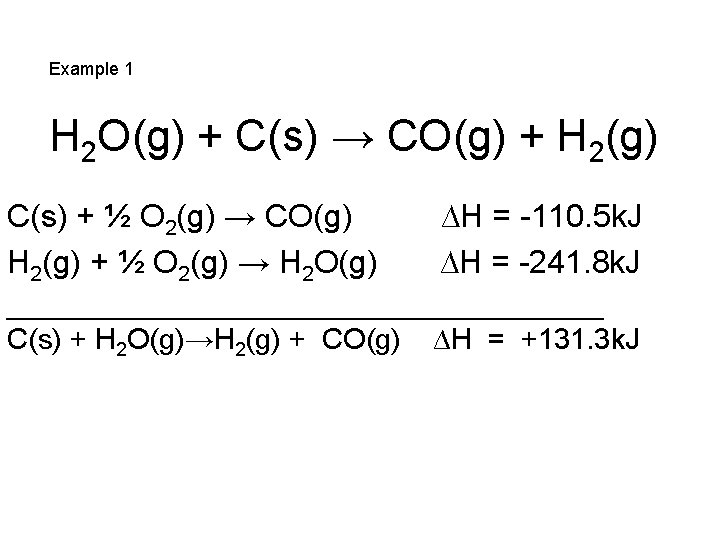
Example 1 H 2 O(g) + C(s) → CO(g) + H 2(g) C(s) + ½ O 2(g) → CO(g) H 2(g) + ½ O 2(g) → H 2 O(g) ∆H = -110. 5 k. J ∆H = -241. 8 k. J ___________________ C(s) + H 2 O(g)→H 2(g) + CO(g) ∆H = +131. 3 k. J

Example 2 4 C(s) + 5 H 2(g) → C 4 H 10(g) Use these equations to calculate the molar enthalpy change to make butane gas. C 4 H 10(g) + 6½O 2(g) → 4 CO 2(g) + 5 H 2 O(g) ∆H= -2657. 4 k. J/mol C(s) + O 2(g) → CO 2(g) ∆H= -393. 5 k. J/mol H 2(g) + ½O 2(g) → H 2 O(g) ∆H= -241. 8 k. J/mol

Example 2 4 C(s) + 5 H 2(g) → C 4 H 10(g) 5 H 2 O(g) + 4 CO 2(g) → 6 ½ O 2(g) + C 4 H 10(g) ∆H= +2657. 4 k. J/mol 4 C(s) + 4 O 2(g) → 4 CO 2(g) ∆H= 4(-393. 5 k. J/mol) 5 H 2(g) + 2½O 2(g) → 5 H 2 O(g) ∆H= 5(-241. 8 k. J/mol) ___________________________ 4 C(s) + 5 H 2(g) → C 4 H 10(g) ∆H = -125. 6 k. J/mol

Hess’s Law Start Finish A State Function: Path independent. Both lines accomplished the same result, they went from start to finish. Net result = same. #(min University of Berkley) https: //www. youtube. com/watch? v=Ktx. KNXYhpy 4 46

Determine the heat of reaction for the reaction: 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) Using the following sets of reactions: N 2(g) + O 2(g) 2 NO(g) H = 180. 6 k. J N 2(g) + 3 H 2(g) 2 NH 3(g) H = -91. 8 k. J 2 H 2(g) + O 2(g) 2 H 2 O(g) H = -483. 7 k. J Hint: The three reactions must be algebraically manipulated to sum up to the desired reaction. and. . the H values must be treated accordingly. 47

Goal: 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) Using the following sets of reactions: N 2(g) + O 2(g) 2 NO(g) H = 180. 6 k. J N 2(g) + 3 H 2(g) 2 NH 3(g) H = -91. 8 k. J 2 H 2(g) + H = -483. 7 k. J NH 3: O 2 : NO: H 2 O: O 2(g) 2 H 2 O(g) Reverse and x 4 2 NH 3 2 N 2 + 6 H 2 H = +183. 6 k. J Found in more than one place, SKIP IT (its hard). x 2 x 3 2 N 2 + 2 O 2 4 NO 6 H 2 + 3 O 2 6 H 2 O H = 361. 2 k. J H = -1451. 1 k. J 48
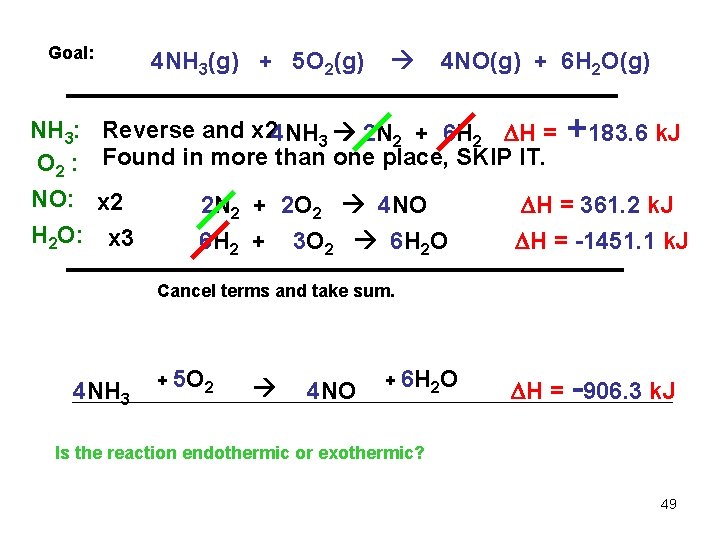
Goal: NH 3: O 2 : NO: H 2 O: 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) Reverse and x 24 NH 3 2 N 2 + 6 H 2 H = Found in more than one place, SKIP IT. x 2 x 3 2 N 2 + 2 O 2 4 NO 6 H 2 + 3 O 2 6 H 2 O +183. 6 k. J H = 361. 2 k. J H = -1451. 1 k. J Cancel terms and take sum. 4 NH 3 + 5 O 2 4 NO + 6 H 2 O H = -906. 3 k. J Is the reaction endothermic or exothermic? 49

Determine the heat of reaction for the reaction: C 2 H 4(g) + H 2(g) C 2 H 6(g) Use the following reactions: C 2 H 4(g) + 3 O 2(g) 2 CO 2(g) + 2 H 2 O(l) H = -1401 k. J C 2 H 6(g) + 7/2 O 2(g) 2 CO 2(g) + 3 H 2 O(l) H = -1550 k. J H 2(g) + 1/2 O 2(g) H 2 O(l) H = -286 k. J Consult your neighbor if necessary. 50

Determine the heat of reaction for the reaction: Goal: C 2 H 4(g) + H 2(g) C 2 H 6(g) H = ? Use the following reactions: C 2 H 4(g) + 3 O 2(g) 2 CO 2(g) + 2 H 2 O(l) H = -1401 k. J C 2 H 6(g) + 7/2 O 2(g) 2 CO 2(g) + 3 H 2 O(l) H = -1550 k. J H 2(g) + 1/2 O 2(g) H 2 O(l) H = -286 k. J C 2 H 4(g) : use 1 as is C 2 H 4(g) + 3 O 2(g) 2 CO 2(g) + 2 H 2 O(l) H = -1401 k. J H 2(g) : # 3 as is H 2(g) + 1/2 O 2(g) H 2 O(l) H = -286 k. J C 2 H 6(g) : rev #2 2 CO 2(g) + 3 H 2 O(l) C 2 H 6(g) + 7/2 O 2(g) H = +1550 k. J C 2 H 4(g) + H 2(g) C 2 H 6(g) H = -137 k. J 51

Summary: enthalpy is a state function and is path independent. 52
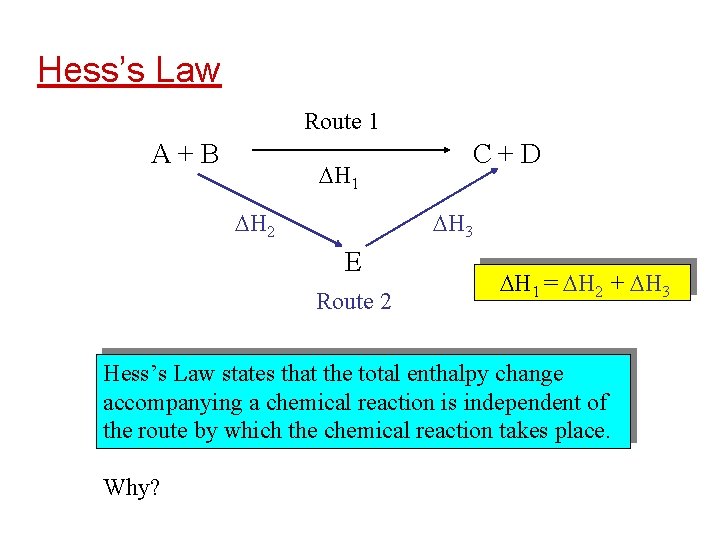
Hess’s Law Route 1 A+B H 1 H 2 C+D H 3 E Route 2 H 1 = H 2 + H 3 Hess’s Law states that the total enthalpy change accompanying a chemical reaction is independent of the route by which the chemical reaction takes place. Why?

Importance of Mathematical Hess’s Law The enthalpy change of some chemical reactions cannot be determined directly because: • the reactions cannot be performed in the laboratory • the reaction rates are too slow • the reactions may involve the formation of side products But the enthalpy change of such reactions can be determined indirectly by applying Hess’s Law.
![Enthalpy Change of Formation of CO(g) ø ø Given: Hf [CO 2(g)] = -393. Enthalpy Change of Formation of CO(g) ø ø Given: Hf [CO 2(g)] = -393.](http://slidetodoc.com/presentation_image_h2/870da92f7e54884425ce0586b71fdeb2/image-55.jpg)
Enthalpy Change of Formation of CO(g) ø ø Given: Hf [CO 2(g)] = -393. 5 k. J mol-1; Hc [CO(g)] = -283. 0 k. J mol-1 ø Hf [CO(g)] C(graphite) + ½O 2(g) CO(g) + ½O 2(g) H 1 CO 2(g) H 2 ø Hf [CO(g)] + H 2 = H 1 ø Hf [CO(g)] = H 1 - H 2 = -393. 5 - (-283. 0 = )-110. 5 k. J mol-1

Calorimetry Fuse school ( calorimeters and heats of combustion ) https: //www. youtube. com/watch? v=if. Ttdv. F 98 T 8

10. 2 Introduction to Thermodynamics is the study of the interconversion of heat and other kinds of energy. In thermodynamics, there are three types of systems: An open system can exchange mass and energy with the surroundings. A closed system allows the transfer of energy but not mass. An isolated system does not exchange either mass or energy with its surroundings.
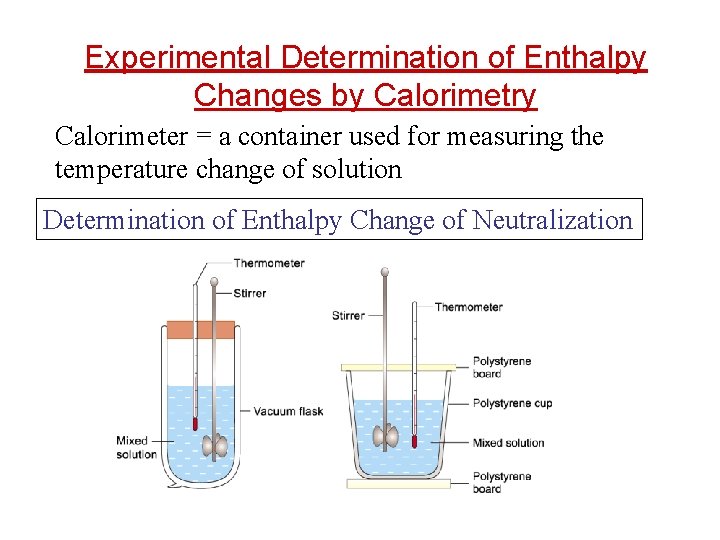
Experimental Determination of Enthalpy Changes by Calorimetry Calorimeter = a container used for measuring the temperature change of solution Determination of Enthalpy Change of Neutralization
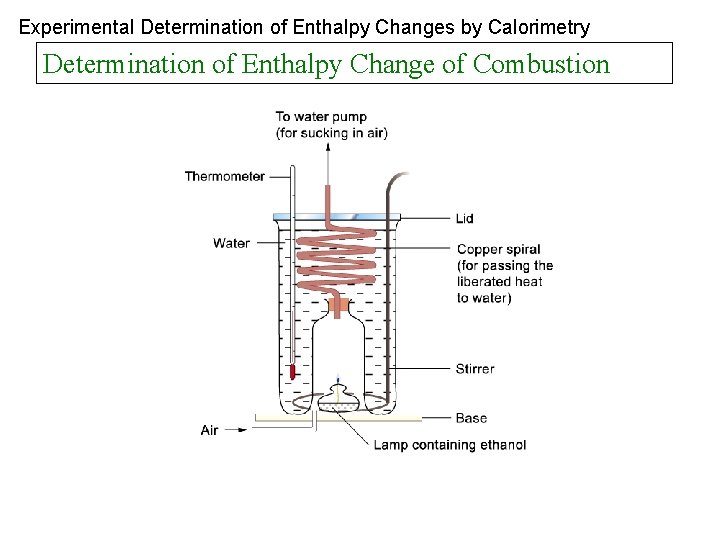
Experimental Determination of Enthalpy Changes by Calorimetry Determination of Enthalpy Change of Combustion
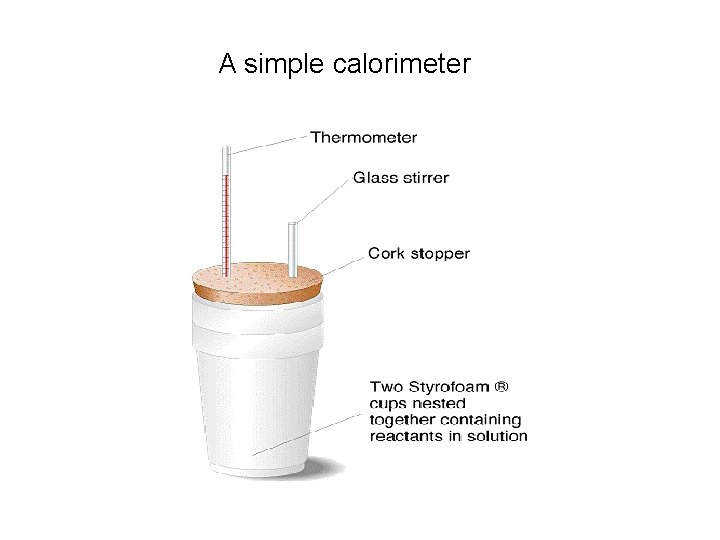
A simple calorimeter

Specific heat capacity is the amount of heat needed to raise the temperature of 1 g of substance by 1 K Specific heat capacity of water = 4. 18 KJ kg-1 K-1 or 4. 18 J g -1 K 1 Be careful with the units it could also be quoted as KJ g-1 K-1 Ensure you use the correct units in your calculation! To measure the heat released in a process we arrange for the heat to be transferred to a substance (usually water) then measure the temperature rise.

Specific Heat and Heat Capacity The heat associated with a temperature change may be calculated: q = mcΔT q = CΔT m is the mass. (c) is the specific heat. ΔT is the change in temperature (ΔT = Tfinal – Tinitial). C is the heat capacity.

Calculate the heat to take 255 g of water from 25. 2 C to 90. 5 C. Strategy Use q = mcΔT to calculate q. ΔT = 90. 5°C – 25. 2°C = 65. 3°C. c = 4. 184 J/g∙°C, m = 255 g, Solution q = 4. 184 J/g. C × 255 g × 65. 3°C = 6. 97× 104 J or 69. 7 k. J SPECIFIC HEAT PROBLEM It takes 487. 5 J to heat 25 grams of copper from 25 °C to 75 °C. What is the specific heat in Joules/g·°C? q = CΔT 487. 5 J = (25 g) c (75 °C - 25 °C) 487. 5 J = (25 g) c (50 °C) Answer: The specific heat of copper is 0. 39 J/g·°C.

Cal Problems worked 4 min https: //www. youtube. com/watc h? v=Hs 5 x 0 -IU 2 F 4

Qsystem = - Qsurroundings Example Problem A 12. 9 gram sample of an unknown metal at 26. 5°C is placed in a Styrofoam cup containing 50. 0 g of water at 88. 6°C. The water cools down and the metal warms up until thermal equilibrium is achieved at 87. 1°C. Determine the specific heat capacity of the unknown metal. The specific heat capacity of water is 4. 18 J/g/°C. ANSWER: This problem is like two problems in one. At the center of the problem-solving strategy is the recognition that the quantity of heat lost by the water (Qwater) = (Qmetal). Part 1: Determine the Heat Lost by the Water Given: m = 50. 0 g C = 4. 18 J/g/°C Tinitial = 88. 6°C Tfinal = 87. 1°C ΔT = -1. 5°C Solve for Qwater= m • C • ΔT = (50. 0 g) • (4. 18 J/g/°C) • (-1. 5°C) Qwater = -313. 5 J (unrounded) (The - sign indicates that heat is lost by the water) Part 2: Determine the value of Cmetal Given: Qmetal = 313. 5 J (use a + sign since the metal is gaining heat) Rearrange Qmetal = mmetal • Cmetal • ΔTmetal to obtain Cmetal = Qmetal / (mmetal • ΔTmetal) = (313. 5 J)/[(12. 9 g) • (60. 6°C)] Cmetal = 0. 40103 J/g/°C

A metal pellet with a mass of 100. 0 g, originally at 88. 4°C, is dropped into 125 g of water at 25. 1°C. The final temperature is 31. 3 o. C. Calculate the heat capacity C of the pellet. Strategy Water constitutes the surroundings; the pellet is the system. Use qsurr = mcΔT to determine the heat absorbed by the water; then use q = CΔT to determine the heat capacity of the metal pellet, no mass needed as C is J/C mwater = 125 g, cwater = 4. 184 J/g∙C, and ΔTwater = 6. 2°C. 4. 184 J × 125 g × 6. 2°C = 3242. 6 J gained by water g∙°C The heat absorbed by the water must be released by the pellet: qwater = -qpellet (3242. 6 J lost by pellet) and ΔTpellet = -57. 1°C. From q = CΔT we have -3242. 6 J = Cpellet × (-57. 1°C) Thus, Cpellet = 56. 8 J/°C

Excess powdered zinc was added to 100 ml of 0. 2 mol/L copper (II) sulphate solution. A temperature rise of 10 o. C was recorded. Find the MOLAR enthalpy change for the reaction. H = m x c x T H = 100 g x 4. 18 KJ kg-1 K-1 x 10 KJ 1000 = 4. 180 KJ This is for the no of moles of Cu. SO 4 used in the experiment No of moles of Cu. SO 4 = 0. 2 x 100 = 0. 02 moles 1000 H = 4. 180 = 209 KJ mol-1 0. 02 The reaction is exothermic so we need to put in a negative sign Hr = - 209 KJ mol-1 of Cu. SO 4

Combustion To find the heat of combustion of a substance a known mass of the substance is burned, the heat released transferred to water and the enthalpy change found as before In an experiment to find the heat of combustion of ethanol the following results were obtained Initial mass of lamp + ethanol = 65. 20 g Final mass of lamp = 64. 28 g Final temperature of water Initial temperature of water = = 47. 1 o. C 28. 5 o. C Mass of the water = 300 g What are the products of complete combustion of ethanol? What mass of ethanol was burnt? How many moles is this? What quantity of heat was transferred to the water? Find Hc of ethanol Identify any sources of error Is ethanol a good fuel?

C 2 H 5 OH + 3 O 2 2 CO 2 + 3 H 2 O q=mc∆t H = 300 x 4. 18 KJ kg-1 K-1 x 18. 6 K = 23. 3 KJ 1000 H lost = h gained (so 23. 3 KJ gained by calorimeter was lost in burning) Mass of ethanol used = 0. 92 g = 0. 92 g = 0. 020 mol 46 g/mol H=mc∆t / n Hc = - 23. 3 = -1160 KJ/mol 0. 020

Errors Heat lost to surroundings (air, can thermometer) Errors in measuring temperature change (unavoidable error in reading thermometer) Errors in measuring masses (unavoidable error in reading balance) The enthalpy change of combustion is high ethanol is a good fuel

Chemistry in Action: Fuel Values of Foods and Other Substances C 6 H 12 O 6 (s) + 6 O 2 (g) 6 CO 2 (g) + 6 H 2 O (l) H = -2820 k. J/mol Per gram Sugar = 2999 KJ / 180 g = 15. 7 KJ/gram 1 cal = 4. 184 J 1 Cal = 1000 cal = 4184 J =4. 184 KJ If 1. 0 gram sugar is = 15. 7 KJ Or 15. 7 KJ / 4. 184 KJ/Cal = 3. 75 Cal So sugar burned is approx 4 Cal/g Here 26 g x 4 Cal/g = 104 Cal from sugar

Foods and Fuels Cheetos burned v Cereal https: //www. youtube. com/watch? v=UDgea. AMd. YIY Foods • 1 nutritional Calorie, 1 Cal = 1000 cal = 1 kcal. • Energy in us comes from carbohydrates and fats TED X (3 min, Calories) https: //www. youtube. com/watch? v=VEQa. H 4 Lru. Uo • Intestines: carbohydrates converted into glucose: C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O, H = -2816 k. J • Fats break down as follows: 2 C 57 H 110 O 6 + 163 O 2 114 CO 2 + 110 H 2 O, H = -75, 520 k. J Fats contain more energy; are not water soluble, so are good for energy storage.

The END
- Slides: 73