Thermochemistry Thermochemistry All changes in matter both physical

Thermochemistry

Thermochemistry • All changes in matter, both physical and chemical, involve changes in energy • Thermochemistry is the study of energy changes is physical and chemical processes

Energy • All forms of energy can be classified as either: – Kinetic Energy - energy of motion – Potential Energy - stored energy • SI unit = joule (J) = kg·m 2/s 2 • 1 J is approximately the amount of energy required to lift an apple 1 m (very small!) so often expressed as k. J (1000 J) • 1 calorie = 4. 184 J
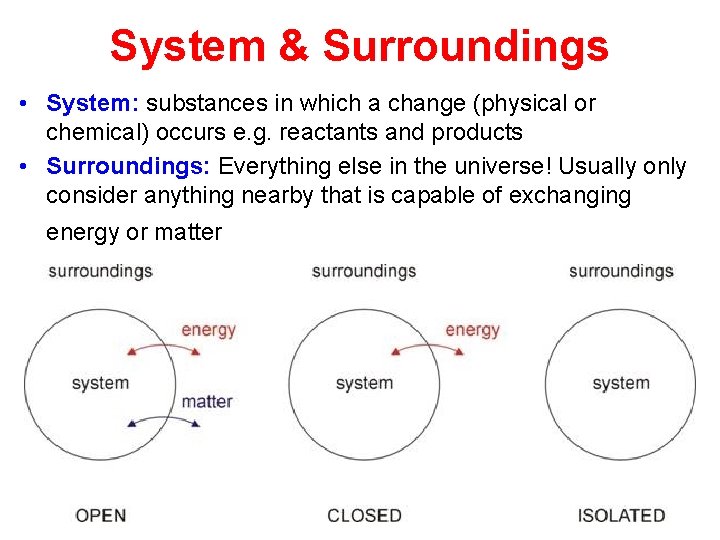
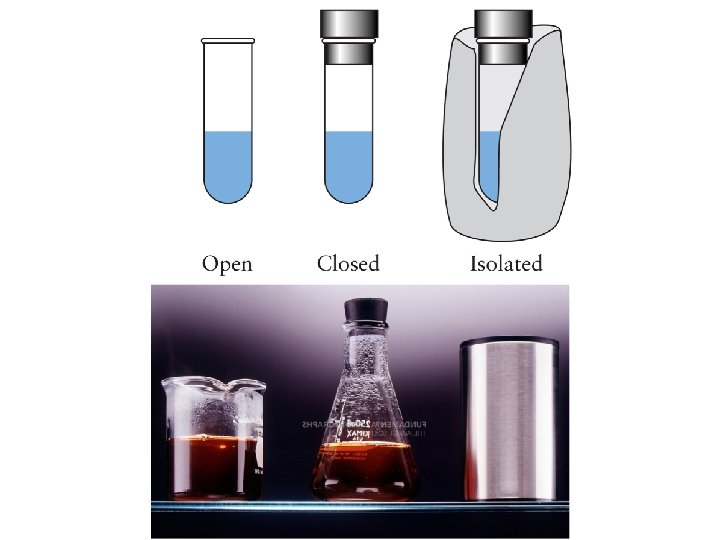
System & Surroundings • System: substances in which a change (physical or chemical) occurs e. g. reactants and products • Surroundings: Everything else in the universe! Usually only consider anything nearby that is capable of exchanging energy or matter
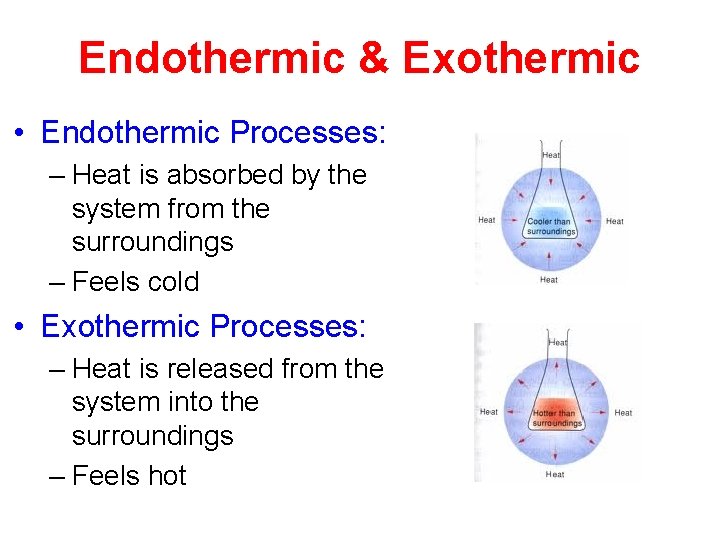
Endothermic & Exothermic • Endothermic Processes: – Heat is absorbed by the system from the surroundings – Feels cold • Exothermic Processes: – Heat is released from the system into the surroundings – Feels hot

Energy, Temperature & Heat Thermal Energy: The sum of all kinetic energies of all the particles in a system. Cannot be measured. Temperature: A measure of the average kinetic energy of all the particles in a system. Can be measured. Heat: Kinetic energy transferred between substances. I. e. an object cannot possess heat.

Heat Capacity: Amount of heat required to raise the temperature of a substance of any mass by 1˚C. C = J/˚C Specific Heat Capacity: Amount of heat required to raise the temperature 1 g of a substance by 1˚C. c = J/g • ˚C • You are provided with a glass of milk and a swimming pool of milk. – Which will have the higher heat capacity? – Which will have the higher specific heat capacity? Molar Heat Capacity: Amount of heat required to raise the temperature 1 mole of a substance by 1˚C. c = J/mol • ˚C


Factors that Affect Heat • Mass - the greater the number of particles, the more heat required • Type of Substance - each substance has a different specific heat capacity • Temperature Change - the greater the temperature change the more heat required Q = mc∆T

Calculating Heat When a 1. 25 kg sample of water was heated in a kettle, its temperature increased from 16. 4˚C to 98. 9˚C. How much heat did the water absorb? Q = mc∆T Q=? m = 1. 25 kg c = 4. 18 J/g • ˚C ∆T = T 2 - T 1 = 98. 9˚C - 16. 4 ˚C = 82. 5˚C

Practice • • p. 300 #1 -5 p. 302 #8 -13 p. 305 #1 -6 Specific Heat Capacity Worksheet
- Slides: 12