Thermochemistry The study of energy changes that occur







- Slides: 8

Thermochemistry The study of energy changes that occur during chemical ____ : Ø at constant volume DU = q. V no ____ Ø at constant pressure DH = q. P only ______ work For practical reasons most measurements are made at constant _____, so thermochemistry mostly deals with DH. If DH > 0 the reaction is ____. If DH < 0 the reaction is ____. For comparison purposes we need to refer DH to the same ____ and ____. To define a standard reaction enthalpy each component of the reaction must be in its ________ – the most stable form at 1 bar pressure and (usually) 25°C. 1 bar = 105 Pa © Paul Percival 1 atm = 1. 01325 bar Modified by Jed Macosko 9/30/2020

Reaction Enthalpy 1 Hess’s Law The standard enthalpy change in any reaction can be expressed as the ____ of the standard enthalpy changes, at the same temperature, of a ____ of reactions into which the overall reaction can be formally divided. Combine chemical equations as if ______ equations, e. g. Standard Reaction Enthalpy reaction enthalpy at ____ … and at ____ T or some other T © Paul Percival Modified by Jed Macosko 9/30/2020

Reaction Enthalpy 2 Standard (molar) enthalpy of ____ Heat of formation of a substance from its elements, all substances being in their standard state. By definition, for all ____ Enthalpy of ____ DH° for total oxidation of a substance e. g. C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O Dc. H° = -2808 k. J mol-1 Enthalpy of ____ DH° when an unsaturated organic compound becomes fully saturated e. g. C 6 H 6 + 3 H 2 C 6 H 12 DH° = -246 k. J mol-1 Enthalpy of ______ Bond dissociation enthalpy DH° for the dissociation of a molecule into its constituent gaseous atoms e. g. C 2 H 6 (g) 2 C(g) + 6 H(g) DH° = 2883 k. J mol-1 Bond ____ single bond enthalpy An average value taken from a series of compounds and often combined for a ____ estimate e. g. DH°(C 2 H 6) = DH°(C-C) + 6 DH°(C-H) © Paul Percival Modified by Jed Macosko 9/30/2020

Temperature Dependence of DH° The temperature dependence of reaction enthalpies can be expressed in terms of the T dependence of the enthalpies of the reaction _____ : This general phenomenon is known as Kirchoff’s Law. e. g. assuming that the Cp values are ______ independent. © Paul Percival Modified by Jed Macosko 9/30/2020

Reactions at Constant Volume For ____ and liquids For ideal gases The relationship between ______ and DU is particularly important when relating thermochemical enthalpies (DH) to molecular properties (Umolecular), e. g. for a single bond energy DU = DH – RT as seen in the case of O 2(g) 2 O(g). In practice, ____ is usually so much smaller than DH that it is often ignored. © Paul Percival Modified by Jed Macosko 9/30/2020

Enthalpies of Ions in Solution Enthalpy of ____ DH° for solution of a substance in a stated amount of solvent Enthalpy of ____ DH° for dilution of a solution to a lower concentration Enthalpy of solution to ______ dilution infinite amount of solvent for an The enthalpy of formation for a species in ____ can be found by combining with the of the ____ species: for individual ions in solution can only be found if one is arbitrarily fixed. By convention this is ____. The ____ state for a substance in solution (not just ions) is a concentration of 1 mole solute in 1 kg solution (1 molal). © Paul Percival Modified by Jed Macosko 9/30/2020

Enthalpy of Formation of an Ionic Solid Consider individual steps in the formation of Na. Cl. 1. 2. 3. 4. Na. Cl(aq) 5. Na(s) + ½Cl 2(g) Na. Cl(aq) Step 5 could be creation of solid Na. Cl instead of solution 5'. leading us to the enthalpy of formation of solid Na. Cl: Na(s) + ½Cl 2(g) Na. Cl(s) © Paul Percival Modified by Jed Macosko 9/30/2020

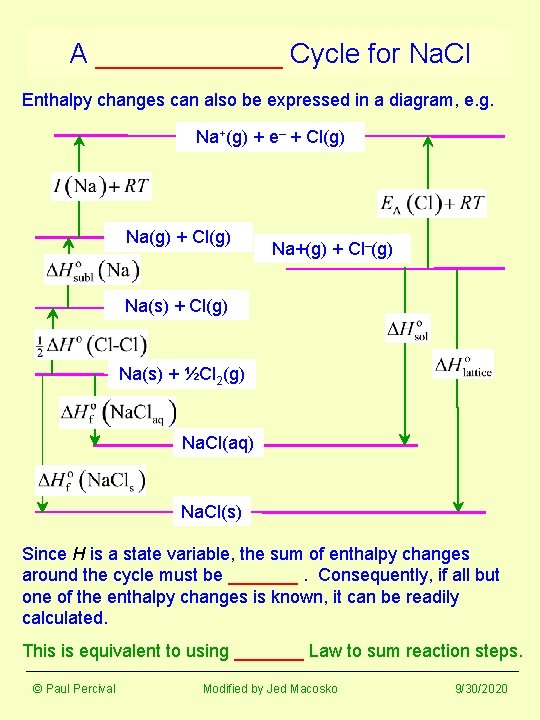
A ______ Cycle for Na. Cl Enthalpy changes can also be expressed in a diagram, e. g. Na+(g) + e– + Cl(g) Na(g) + Cl(g) Na+(g) + Cl–(g) Na(s) + Cl(g) Na(s) + ½Cl 2(g) Na. Cl(aq) Na. Cl(s) Since H is a state variable, the sum of enthalpy changes around the cycle must be _______. Consequently, if all but one of the enthalpy changes is known, it can be readily calculated. This is equivalent to using _______ Law to sum reaction steps. © Paul Percival Modified by Jed Macosko 9/30/2020