Thermochemistry pt 2 Calorimetry H can be found

Thermochemistry pt 2

Calorimetry ΔH can be found experimentally or calculated from known enthalpy changes Measure heat flow with a calorimeter Heat capacity (J/K) – amount of heat needed to raise the temperature by one degree Specific Heat capacity (J/g·K) – amount of heat needed to raise the temperature of 1 g of a substance by one degree

Specific Heat Specific heat can be found by measuring the temperature change (ΔT) a known mass (m) undergoes when it loses or gains a specific amount of heat (q) Specific heat (c) = q / (m x ΔT) What is the specific heat of water if 209 J of heat is needed to increase the temperature of 50. 0 g of water by 1. 00 K? Why does it not matter whether we work in Celsius or Kelvin?

Concept Practice How much heat is needed to warm 250 g of water from 22°C to near its boiling point, 98°C? The specific heat of water is 4. 18 J/g·K. What is the molar heat capacity of water? Answer = 7. 9 x 104 J, 75. 2 J/mol·K

Concept Practice Large beds of rocks are used in some solarheated homes to store heat. Assuming the specific heat of the rocks is 0. 82 J/g·K, how much heat has been absorbed if the temperature of 50. 0 kg of rocks increases by 12. 0°C. What temperature change occurs if the rocks emit 450 k. J of heat? Answer = 4. 9 x 105 J, 11 K=11°C

Constant Pressure Calorimetry Assume that calorimeter prevents the loss or gain of heat qr x n completely transferred to the solution -qr x n=qs o l n = mcΔT = -qr x n

Concept Practice When a student mixes 50 m. L of 1. 0 M HCl and 50 m. L of 1. 0 M Na. OH in a coffee-cup calorimeter the temperature of the resultant solution increases from 21. 0°C to 27. 5°C. Calculate the enthalpy change for the reaction assuming the total volume of the solution is 100 m. L, that its density is 1. 0 g/m. L and the its specific heat capacity is 4. 18 J/g·°C. -54 k. J/mol

Concept Practice When 50. 0 m. L of 0. 100 M Ag. NO 3 and 50. 0 m. L of 0. 100 M HCl are mixed in a constant pressure calorimeter, the temperature of the mixture increases from 22. 30°C to 23. 11°C. The temperature increase is caused by the following reaction: Ag. NO 3(aq) + HCl(aq) → Ag. Cl(s) + HNO 3(aq) Calculate the heat of the reaction assuming the combined solution has a mass of 100. 0 g and a specific heat of 4. 18 J/g·°C. Answer = -68 k. J/mol

Constant Volume Calorimetry Combustion reactions are studied using a bomb calorimeter To measure heat of combustion we need the heat capacity of the calorimeter (Cc a l) qr x n = -Cc a l x ΔT Remember at constant volume q = ΔE

Concept Practice Methylhydrazine (CH 6 N 2) is commonly used as a liquid rocket fuel. The combustion of methylhydrazine with oxygen produces nitrogen, carbon dioxide and liquid water. When 4. 00 g of the fuel is combusted in a bomb calorimeter, the temperature of the calorimeter increases from 25. 00°C to 39. 50°C. In a separate experiment, the heat capacity of the calorimeter is found to be 7. 794 k. J/°C. What is the heat of reaction for the combustion of a mole of the rocket fuel in this calorimeter? Answer = -1. 30 x 103 k. J/mol

Concept Practice A 0. 5865 sample of lactic acid (HC 3 H 5 O 3) is burned in a calorimeter whose heat capacity is 4. 812 k. J/°C. The temperature increases from 23. 10°C to 24. 95°C. Calculate the heat of combustion of a) lactic acid per gram and b) per mole.

Hess's Law = If a reaction is carried out in a series of steps, ΔH for the reaction will equal the sum of the enthalpy changes for the individual steps. Combustion of methane CH 4(g) + 2 O 2(g) → CO 2(g) + 2 H 2 O(g) → 2 H 2 O(l) ΔH = -802 k. J ΔH = -88 k. J CH 4(g) + 2 O 2(g) + 2 H 2 O(g) → CO 2(g) + 2 H 2 O(l) ΔH = -890 k. J

Hess's Law

Hess's Law The number of steps does not matter ΔH for any process can be measured as long as we know the ΔH for each of the individual steps This allows us to find ΔH for reactions that are difficult to measure. The combustion of carbon to form carbon monoxide The conversion of graphite to diamond

Concept Practice C(s) + O 2(g) → CO 2(g) ΔH = -393. 5 k. J CO(g) + ½ O 2(g) → CO 2(g) ΔH = -283. 0 k. J Use the above reactions calculate the enthalpy of combustion of C to CO. C(s) + ½ O 2(g) → CO(g) Answer = -110. 5 k. J

Concept Practice Carbon occurs in two forms, graphite and diamond. The enthalpy of combustion of graphite is -395. k. J/mol and that of diamond is 395. 4 k. J/mol. Calculate the ΔH for the conversion of graphite to diamond. Answer = 1. 9 k. J

Concept Practice Calculate ΔH for the reaction 2 C(s) + H 2(g) → C 2 H 2(g) using the following pieces of data: C 2 H 2(g) + 5/2 O 2(g) → 2 CO 2(g) + H 2 O(l) ΔH = -1299. 6 k. J C(s) + O 2(g) → CO 2(g) ΔH = -393. 5 k. J H 2(g) + ½ O 2(g) → H 2 O(l) ΔH = -285. 8 k. J Answer = 226. 8 k. J

Enthalpy Various ΔH measured according to the type of process. ΔHv a p = vaporization ΔHf u s = fusion ΔHf = formation Enthalpy changes depend on temperature, pressure and state of reactants and products To compare enthalpies we define a standard state

Standard States A substance is at standard state when it is in its pure form at one atmosphere and the temperature of interest. Standard enthalpy (ΔH°) is when all reactants and products are in their standard states. Standard enthalpy of formation (ΔHf°) is the change in enthalpy that forms one mol of the compound from its elements. 2 C(graphite) + 3 H 2(g) + ½O 2(g) → C 2 H 5 OH(l) ΔHf°= -277. 7 k. J ΔHf° of elements in their standard state is zero.

Concept Practice For which of the following reactions at 25°C would the enthalpy change represent a standard enthalpy of formation? For those that are not, what changes should be made in the reaction conditions? 2 Na(s) + ½O 2(g) → Na 2 O(s) 2 K(l) + Cl 2(g) → 2 KCl(s) C 6 H 12 O 6(s) → 6 C(diamond) + 6 H 2(g) + 3 O 2(g)

Calculating ° ΔH from rxn ° ΔHf ΔH°rxn = Σ nΔHf° (products) - Σ nΔHf° (reactants) Calculate the standard enthalpy change for the combustion of 1 mol of benzene to carbon dioxide and liquid water. Answer = -3267 k. J
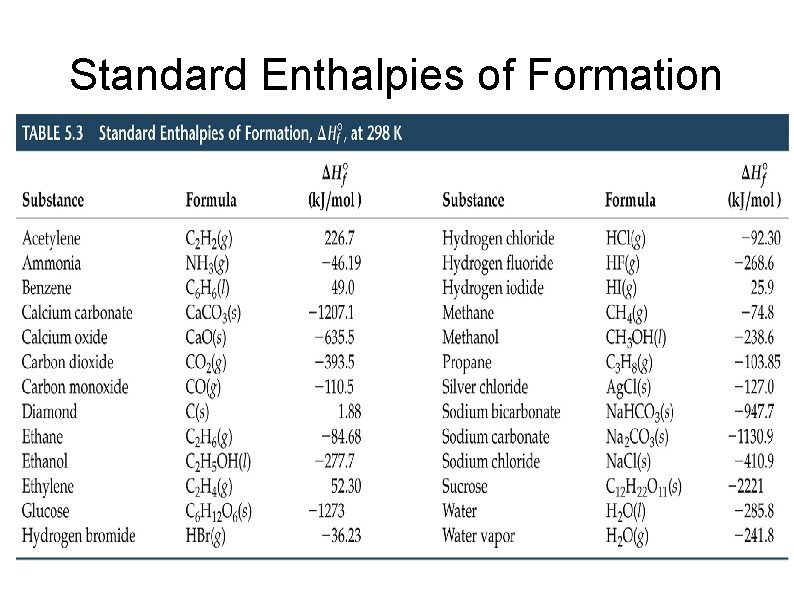
Standard Enthalpies of Formation

Concept Practice Calculate the enthalpy change for the combustion of 1 mol of ethanol (C 2 H 5 OH) The standard enthalpy change for the reaction Ca. CO 3(s) → Ca. O(s) + CO 2(g) is 178. 1 k. J. Calculate the standard enthalpy of formation of calcium carbonate. Given the following standard enthalpy of

Homework 44, 47, 49, 52, 54, 57, 59, 61, 68, 70, 75, 108, 111,
- Slides: 24