Thermochemistry Chapter 16 Section 1 Thermochemistry Objectives Define

Thermochemistry

Chapter 16 Section 1 Thermochemistry Objectives • Define temperature and state the units in which it is measured. • Define heat and state its units. • Perform specific-heat calculations. • Solve problems involving enthalpies of reaction, enthalpies of formation, and enthalpies of combustion.

Section 1

Chapter 16 Section 1 Thermochemistry • Chemical reactions usually either absorb or release energy as heat. • Thermochemistry is the study of the transfers of energy as heat that accompany chemical reactions and physical changes.

Chapter 16 Section 1 Thermochemistry Heat and Temperature • Amount of Energy = Joules (J) or calories (cal) • Heat is the energy transferred between matter • Energy always moves from matter at a higher temperature to matter at a lower temperature.

Chapter 16 Section 1 Thermochemistry Specific Heat • The amount of energy required to raise the temperature of one gram by one Celsius degree (1°C) or one kelvin (1 K). • J/(g • °C) or J/(g • K).
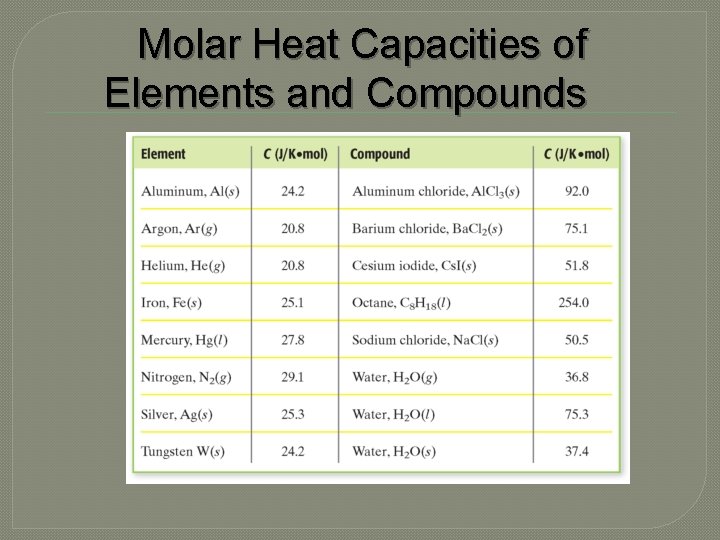
Molar Heat Capacities of Elements and Compounds

Chapter 16 Section 1 Thermochemistry Specific Heat To find the quantity of energy gained or lost with a change of temperature. q = energy absorbed or released Cp = Specific Heat m= mass ∆ T= change in temperature = Tfinal - Tinitial

How do we know energy is released or absorbed? �Endothermic • Energy is Absorbed • Reactant • q = positive number (+) �Exothermic • Energy is released • Product • q = negative number (-)

Chapter 16 Section 1 Thermochemistry Sample Problem A A 4. 0 g sample of glass was heated from 274 K to 314 K, a temperature increase of 40. K, and was found to have absorbed 32 J of energy as heat. a. What is the specific heat of this type of glass? b. How much energy will the same glass sample Let’s information gain. Organize when it the is heated from 314 K to 344 K? q = 32 J Cp = X m= 4. 0 g ∆ T= Tfinal – Tinitial = 314 K – 274 K = 40 K

q = Cp x m x ∆ T 32 J = Cp (4. 0 g)(40 K) 0. 2 J/g. K= Cp

b. How much energy will the same glass sample gain when it is heated from 314 K to 344 K? Let’s Organize the information q=X Cp = 0. 2 J/g K m= 4. 0 g ∆ T= Tfinal – Tinitial = 344 K – 314 K= 30 K
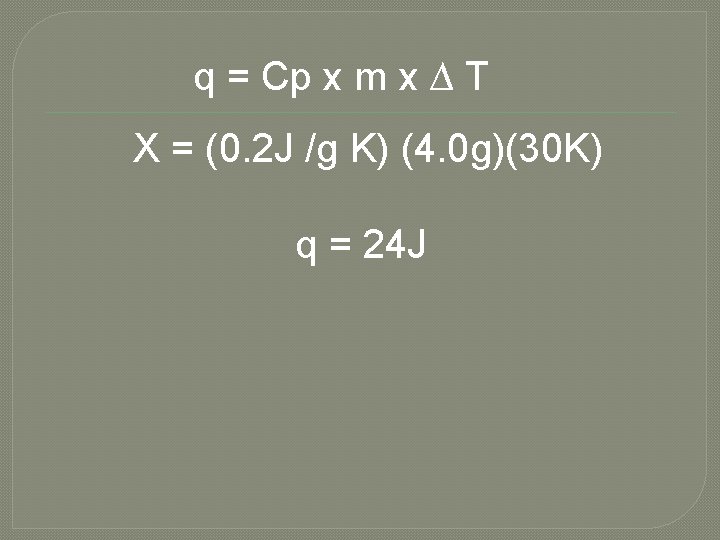
q = Cp x m x ∆ T X = (0. 2 J /g K) (4. 0 g)(30 K) q = 24 J

Chapter 16 Section 1 Thermochemistry Enthalpy of Reaction • The energy absorbed as heat during a chemical reaction at constant pressure is represented by ∆H.

Chapter 16 Section 1 Thermochemistry Enthalpy of Reaction, continued Enthalpy change is always the difference between the enthalpies of products and reactants. ∆H = Hproducts – Hreactants

Chapter 16 Section 1 Thermochemistry 2 H 2(g) + O 2(g) 2 H 2 O(g) + 483. 6 k. J The expression above is an example of a thermochemical equation • an equation that includes the quantity of energy released or absorbed as heat during the reaction as written.

Exothermic Reaction • Releases Energy • The energy of the products is less than the energy of the reactants. example: 2 H 2(g) + O 2(g) 2 H 2 O(g) + 483. 6 k. J
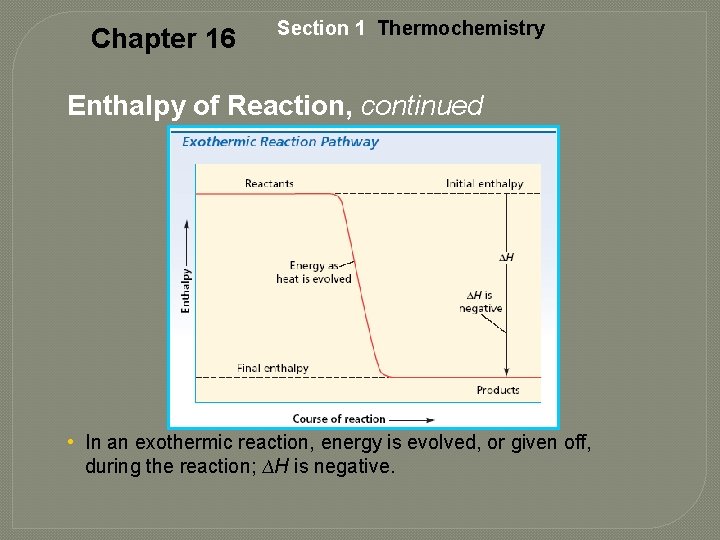
Chapter 16 Section 1 Thermochemistry Enthalpy of Reaction, continued • In an exothermic reaction, energy is evolved, or given off, during the reaction; ∆H is negative.

Chapter 16 Section 1 Thermochemistry Endothermic Reaction • Absorbed energy • The products have a larger enthalpy than the reactants, and the reaction absorbs energy. • example: 2 H 2 O(g) + 483. 6 k. J 2 H 2(g) + O 2(g)
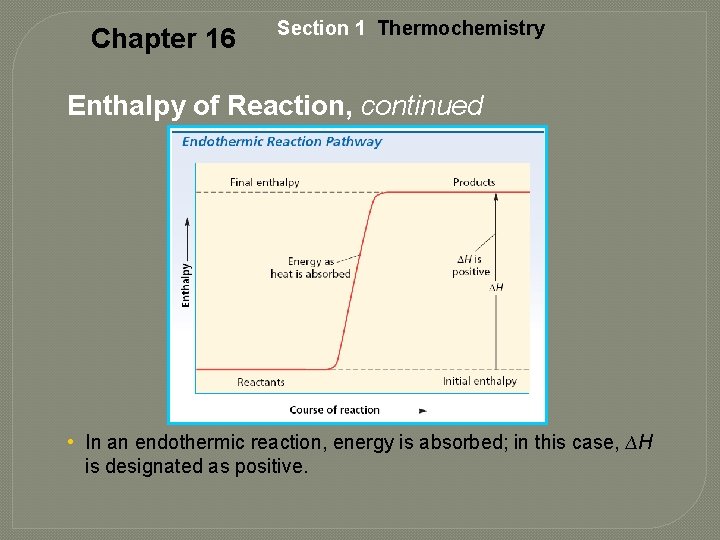
Chapter 16 Section 1 Thermochemistry Enthalpy of Reaction, continued • In an endothermic reaction, energy is absorbed; in this case, ∆H is designated as positive.

Chapter 16 Section 1 Thermochemistry Enthalpy of Combustion • A combustion calorimeter, shown below, is a common instrument used to determine enthalpies of combustion.

Chapter 16 Section 1 Thermochemistry Calculating Enthalpies of Reaction • The basis for calculating enthalpies of reaction is known as Hess’s law:

Chapter 16 Section 1 Thermochemistry Calculating Enthalpies of Reaction, continued To demonstrate how to apply Hess’s law, we will work through the calculation of the enthalpy of formation for the formation of methane gas, CH 4, from its elements, hydrogen gas and solid carbon: C(s) + 2 H 2(g) CH 4(g)

Chapter 16 Section 1 Thermochemistry Calculating Enthalpies of Reaction, continued • The component reactions in this case are the combustion reactions of carbon, hydrogen, and methane: C(s) + O 2(g) CO 2(g) H 2(g) + O 2(g) H 2 O(l) CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l)

Chapter 16 Section 1 Thermochemistry Calculating Enthalpies of Reaction, continued • We are now ready to add the three equations together using Hess’s law to give the enthalpy of formation for methane and the balanced equation. C(s) + O 2(g) CO 2(g) 2 H 2(g) + O 2(g) 2 H 2 O(l) CO 2(g) + 2 H 2 O(l) CH 4(g) + 2 O 2(g) C(s) + 2 H 2(g) CH 4(g)

Chapter 16 Section 1 Thermochemistry Standard Enthalpies of Formation • Using Hess’s law, any enthalpy of reaction may be calculated using enthalpies of formation for all the substances in the reaction of interest, without knowing anything else about how the reaction occurs. ∆H 0 = ∑[n( of products) ] – ∑[n( of reactants)]

Sample Problem A �Use the standard enthalpies of formation to determine ∆H 0 rxn for the following reaction. 4 NH 3 (g) + 5 O 2 (g) 4 NO (g) + 6 H 2 O (g)

Step 1. Balance the equation 4 NH 3 (g) + 5 O 2 (g) 4 NO (g) + 6 H 2 O (g)

Step 2: Find each for each reactant and product Reactant or Product ∆H 0 f NH 3(g) -45. 9 O 2(g) 0. 0 NO(g) +91. 3 H 2 O(g) -241. 8 4 NH 3 (g) + 5 O 2 (g) 4 NO (g) + 6 H 2 O (g) 4(-45. 9) -183. 6 5(0) 0 4(91. 3) 365. 2 6(-241. 8) -1450. 8
![Step 3: plug in and solve ∆H 0 = ∑[n(∆H 0 of products) ] Step 3: plug in and solve ∆H 0 = ∑[n(∆H 0 of products) ]](http://slidetodoc.com/presentation_image/1fd26aaca208b68745e0b0d1ade8c3ae/image-30.jpg)
Step 3: plug in and solve ∆H 0 = ∑[n(∆H 0 of products) ] – ∑[n ∆H 0 of reactants)] 4 NH 3 (g) + 5 O 2 (g) 4 NO (g) + 6 H 2 O (g) -183. 6 0 365. 2 -1450. 8 ∆H 0 = [365. 2 + (-1450. 8)] - [-183. 6 + 0] ∆H 0 = -1085. 6 – (-183. 6) ∆H 0 = -902

Sample Problem B �The thermite reaction in which powdered aluminum reacts with iron oxide, is highly exothermic. Use the standard enthalpies of formation to find ∆H 0 rxn for thermite reaction Al(s) + Fe 2 O 3(s) Al 2 O 3(s) + Fe(s) Reactant or Product ∆H 0 f Al(s) 0. 0 Al 2 O 3(s) -1675. 7 Fe(s) 0. 0 Fe 2 O 3(s) -824. 2
![Sample Problem B ∆H 0 = ∑[n(∆H 0 of products) ] – ∑[n ∆H Sample Problem B ∆H 0 = ∑[n(∆H 0 of products) ] – ∑[n ∆H](http://slidetodoc.com/presentation_image/1fd26aaca208b68745e0b0d1ade8c3ae/image-32.jpg)
Sample Problem B ∆H 0 = ∑[n(∆H 0 of products) ] – ∑[n ∆H 0 of reactants)] 2 Al(s) + Fe 2 O 3(s) Al 2 O 3(s) + 2 Fe(s) 0 -824. 2 -1675. 7 0. 0 ∆H 0 = [-1675. 7 + 0] - [-824. 2 + 0] ∆H 0 = -1675. 7 – (-824. 2) ∆H 0 = -851. 5

Section 2

Chapter 16 Section 2 Driving Force of Reactions Objectives • Explain the relationship between enthalpy change and the tendency of a reaction to occur. • Explain the relationship between entropy change and the tendency of a reaction to occur. • Discuss the concept of free energy, and explain how the value of this quantity is calculated and interpreted. • Describe the use of free energy change to determine the tendency of a reaction to occur.

Enthalpy and Reaction Tendency • Majority of chemical reactions in nature are exothermic. • In nature a reaction proceeds in a direction that leads to a lower energy state. • Some endothermic reactions do occur spontaneously.

Chapter 16 Entropy and Reaction Tendency • Melting is naturally occurring endothermic process. • An ice cube melts spontaneously at room temperature • energy is transferred from the warm air to the ice.

Chapter 16 Entropy and Reaction Tendency, continued • In nature reactions proceed in the direction that increases the randomness of a system. • This tendency toward randomness is called entropy.

Chapter 16 Entropy and Reaction Tendency, continued • The decomposition of ammonium nitrate: 2 NH 4 NO 3(s) 2 N 2(g) + 4 H 2 O(l) + O 2(g) • On the left side are 2 mol of solid ammonium nitrate. • The right-hand side of the equation shows 3 mol of gaseous molecules plus 4 mol of a liquid. • Particles on the right-hand side of the equation is more random than the arrangement on the left side and hence is less ordered.
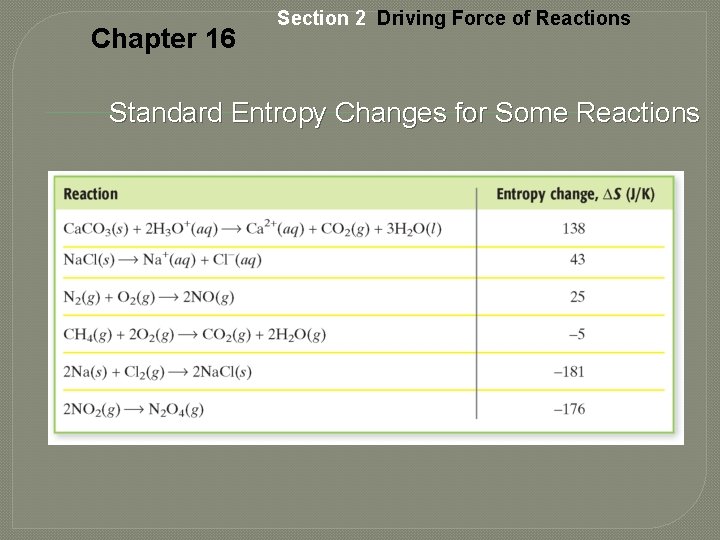
Chapter 16 Section 2 Driving Force of Reactions Standard Entropy Changes for Some Reactions

Chapter 16 Section 2 Driving Force of Reactions Increasing Entropy (Increasing Randomness) S<L< G

Chapter 16 Section 2 Driving Force of Reactions Entropy and Reaction Tendency, continued • Entropy = ∆S • ∆S = positive = increase in entropy • ∆S = negative = decrease in entropy

So how do ∆H and ∆S predict a reaction? ?

Chapter 16 Section 2 Driving Force of Reactions Free Energy • Processes in nature are driven in two directions: toward least enthalpy and toward largest entropy. • This combined enthalpy-entropy function is called the free energy, G, of the system; it is also called Gibbs free energy.

Chapter 16 Section 2 Driving Force of Reactions Free Energy, continued ∆G 0 = ∆H 0 – T∆S 0

Chapter 16 Section 2 Driving Force of Reactions Free Energy, continued ∆G 0 = ∆H 0 – T∆S 0 • The expression for free energy change is for substances in their standard states. • The product T∆S and the quantities ∆G and ∆H have the same units, usually k. J/mol. If ∆G < 0, the reaction is spontaneous.
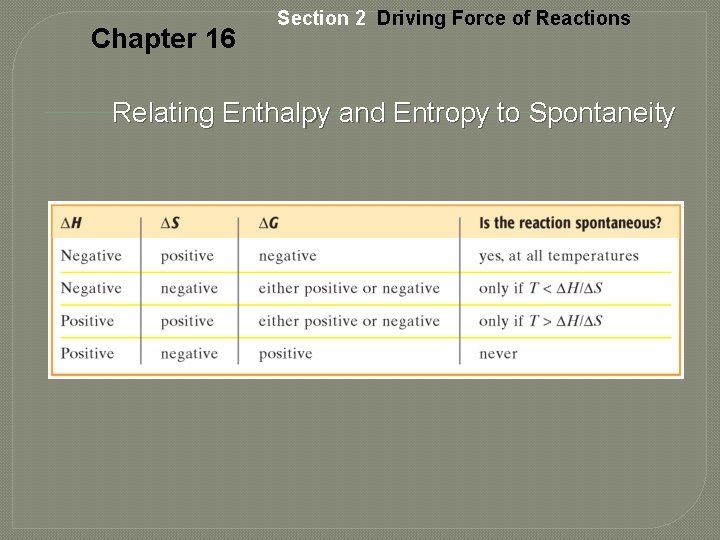
Chapter 16 Section 2 Driving Force of Reactions Relating Enthalpy and Entropy to Spontaneity

Chapter 16 Free Energy, continued Sample Problem D the reaction NH 4 Cl(s) NH 3(g) + HCl(g), at 298. 15 K, ∆H 0 = 176 k. J/mol and ∆S 0 = 0. 285 k. J/(mol • K). Calculate ∆G 0, and tell whether this reaction is spontaneous in the forward direction. � For

∆G 0 = ∆H 0 – T∆S 0 the reaction NH 4 Cl(s) NH 3(g) + HCl(g), at 298. 15 K, ∆H 0 = 176 k. J/mol and ∆S 0 = 0. 285 k. J/(mol • K). Calculate ∆G 0, and tell whether this reaction is spontaneous in the forward direction. � For What do we know? ∆H 0 = 176 k. J/mol T = 298. 15 K ∆S 0 = 0. 285 k. J/(mol • K)

∆G 0 = ∆H 0 – T∆S 0 What do we know? ∆H 0 = 176 k. J/mol T = 298. 15 K ∆S 0 = 0. 285 k. J/(mol • K) ∆G 0 = 176 - (298. 15 x 0. 285) ∆G = 91. 0 ∆G = positive = not spontaneous
- Slides: 49