Thermochemistry 1 Thermochemistry The branch of physical chemistry

Thermochemistry 1

Thermochemistry The branch of physical chemistry that deals with the heat changes accompanying various physical and chemical transformations. Heat is either evolved (exothermic) or absorbed (endothermic) in a chemical or physical change. 2

Thermochemistry System: A part of the universe which is under observation is called the system OR The part of the world in which we have interest. e. g. reaction vessel, calorimeter, and so on. Surroundings: The remaining portion of the universe which is not a part of the system is called the surroundings. e. g. rooms, buildings around etc. 3

Conti…. . Closed system: A type of system in which mass can not pass through the boundary but energy can move (enter or exit) freely. Example; Bomb calorimeter in which energy can move freely but no matter can enter or exit. Isolated system: Nothing can enter or leave. The system that has neither mass nor energy exchange with its surroundings. A substance contained in the ideal thermos flask is an example of the isolated system. 4
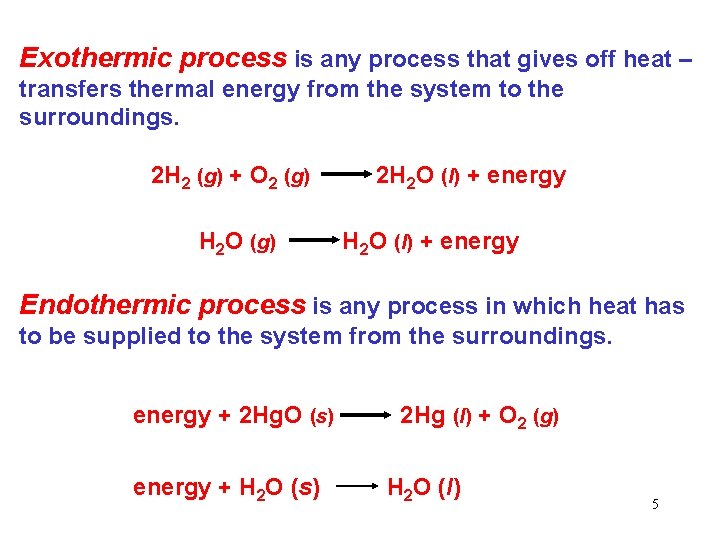
Exothermic process is any process that gives off heat – transfers thermal energy from the system to the surroundings. 2 H 2 (g) + O 2 (g) H 2 O (g) 2 H 2 O (l) + energy Endothermic process is any process in which heat has to be supplied to the system from the surroundings. energy + 2 Hg. O (s) energy + H 2 O (s) 2 Hg (l) + O 2 (g) H 2 O (l) 5
Energy Changes in Chemical Reactions Heat is the transfer of thermal energy between two bodies that are at different temperatures. Temperature is a measure of the degree of coldness or hotness of a body. 900 C 400 C 6
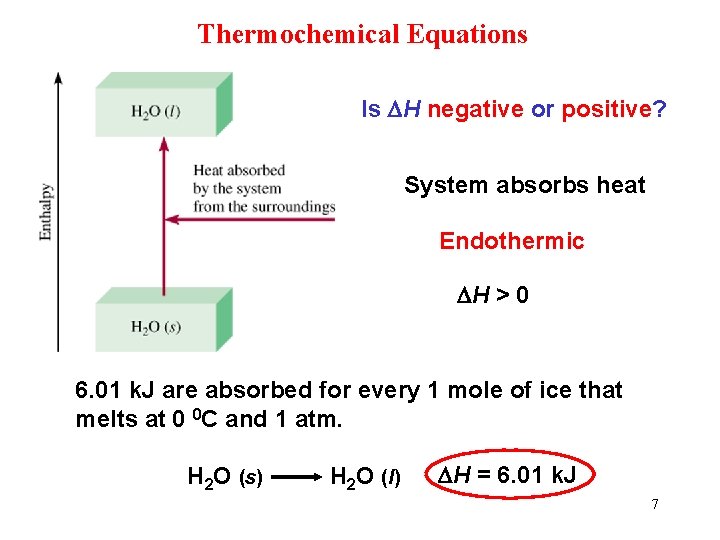
Thermochemical Equations Is DH negative or positive? System absorbs heat Endothermic DH > 0 6. 01 k. J are absorbed for every 1 mole of ice that melts at 0 0 C and 1 atm. H 2 O (s) H 2 O (l) DH = 6. 01 k. J 7
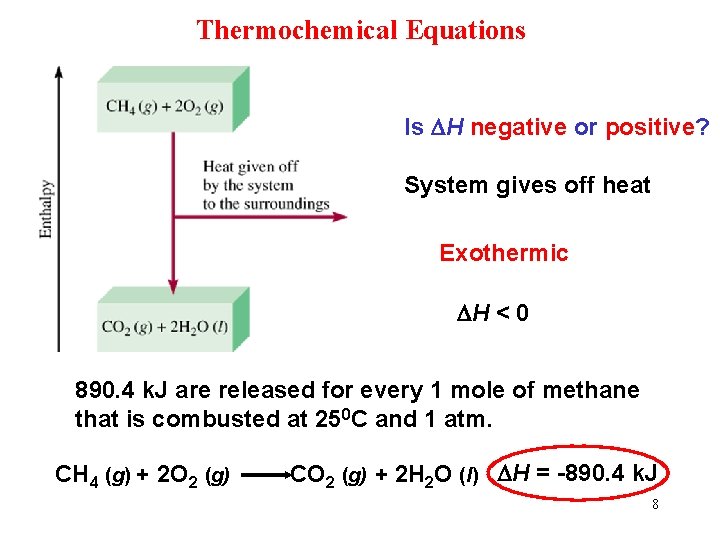
Thermochemical Equations Is DH negative or positive? System gives off heat Exothermic DH < 0 890. 4 k. J are released for every 1 mole of methane that is combusted at 250 C and 1 atm. CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (l) DH = -890. 4 k. J 8

Thermochemical Equations • The stoichiometric coefficients always refer to the number of moles of a substance. H 2 O (s) • H 2 O (l) DH = 6. 01 k. J If you reverse a reaction, the sign of DH changes. H 2 O (l) H 2 O (s) DH = -6. 01 k. J HOWEVER; • If you multiply both sides of the equation by a factor n, then DH must change by the same factor n. 2 H 2 O (s) 2 H 2 O (l) DH = 2 x 6. 01 = 12. 0 k. J 9

ENTHALPY OF REACTION (HEAT OF REACTION) It is defined as the amount of heat evolved or absorbed when molar quantities of reactants shown by the balanced equation react completely at a given temperature. C (s) + O 2 (g) CO 2 (g) ∆H = - 94. 03 k cal OR ∆H = - 393. 5 KJ 10

STANDARD ENTHALPY CHANGES (∆Ho) v. Enthalpy of a reaction depends upon temperature and therefore for comparison of enthalpies of reactions of different substances a standard state is chosen. v A convenient state for a substance is most stable state at 1 atmosphere and at specific temperature usually at 298 k. ∆Ho 298 = Ho of Products ─ Ho of Reactants 11

By convention enthalpy of every element in its standard State is assigned as zero value. For example Ho C (graphite) and Ho O 2 (g) = 0 VARIOUS TYPES OF ENTHALPY CHANGES a) b) c) d) Enthalpy of Formation Enthalpy of Combustion or Heat of Combustion Enthalpy of Neutralization or Heat of Neutralization Enthalpy of Solution Heat of Solution 12

a) Enthalpy of Formation Enthalpy change (∆H) involved in the formation of one mole of compound from its elements. If elements are at their standard state then enthalpy of formation is called standard enthalpy of formation (∆Hof) or standard heat of formation. Standard enthalpy of formation of CH 4 from elements at 298 K is ─ 74. 81 k. J/mol. C (graphite) + 2 H 2 (g) CH 4 (g) ∆Hof , 298 = ─ 74. 81 k. J/mol 13

b) Enthalpy of Combustion or Heat of Combustion Enthalpy change involved in the complete combustion of 1 mol of substance. If the substances are all in their respective standard states, then denoted by ∆Hoc. CH 4(g)+ 2 O 2(g) CO 2(g)+ 2 H 2 O(l) ∆Hoc= ─ 890. 40 k. J/mol 14

c) Enthalpy of Neutralization or Heat of Neutralization The change in enthalpy when 1 gram equivalent of an acid is neutralized by 1 gram equivalent of a base in dilute solution. HCl + Na. OH HNO 3 + Na. OH H+ + OH- Na. Cl + H 2 O ∆Ho = ─ 57. 32 k. J Na. NO 3 + H 2 O ∆Ho = ─ 57. 32 k. J 15

d) Enthalpy of Solution or Heat of Solution Enthalpy change associated when 1 mole of the solute is dissolved in a specific amount of solvent. HCl (g) + 25 H 2 O(l) HCl. 25 H 2 O ∆Ho = ─ 69. 5 k. J HCl (g) + 40 H 2 O(l) HCl. 40 H 2 O ∆Ho = ─ 73. 18 k. J 16

Thermodynamics The study of energy and its interconversions is called thermodynamics. 17

Thermodynamics • First law of thermodynamics: “the internal energy of an isolated system is constant”. Mathematical statement of first law of thermodynamics: • ∆u = q +w, w is the work done on a system and q is the energy transferred as heat to the system. 18

Conti… Thermodynamics if the system does work on the surrounding (energy flows out of the system), w is negative. if the surroundings do work on the system (energy flows into the system), w is positive. problem: calculate ∆u for a system undergoing endothermic process in which 15. 6 k. J of heat flows and where 1. 4 k. J work done on the system. Using equation; ∆u = q + w= 15. 6 + 1. 4 =17. 0 k. J 19

Thermodynamics A gas expanding against pressure p, w is a negative quantity as required, since work flows out of the system. when a gas is compressed, ∆v is a negative quantity, which makes w a positive quantity (work flows into the system). Problem: calculate the work associated with the expansion of a gas from 46 L to 64 L at a constant external pressure of 15 atm. solution: w = - p ∆v = -15 x(64 -46) =-270 L. atm note that since the gas expands, it does work on its surrounding. 20

Hess’s Law • Hess’s law of heat summation: It states that the total enthalpy change for a reaction is the same whether the reaction takes place in a single step or in several steps. OR • It states that for a chemical equation that can be written as the sum of two or more steps, the enthalpy change for the overall equation is the sum of the enthalpy changes for the individual steps. 21

Hess’s Law Consider the formation of carbon dioxide (CO 2). Direct Method C (s) + O 2 (g) CO 2 (g) ∆H = - 393. 5 KJ Indirect Method C (s) + ½ O 2 CO (g) CO 2 (g) ∆H = - 108. 98 KJ ∆H = - 284. 52 KJ __________________ Adding= C (s) + O 2 (g) CO 2 (g) ∆H = - 393. 50 KJ 22

Lavoisier and Laplace law This law states that the quantity of heat required for the decomposition of a compound into its components equals the heat evolved during the formation of the same quantity of the compound from the same components. Example: reversibility of thermo chemical equation showing the formation and decomposition reactions. C (s) + ½ O 2 (g) CO (g) ∆H = - 108. 98 KJ Decomposition CO (g) C (s) + ½ O 2 (g) ∆H = + 108. 98 23 KJ

Calorimetry (Measuring Heats of Reactions) Calorimetry, Latin words calor meaning heat, and the Greek metry meaning to measure, is the science of measuring the amount of heat. All calorimetric techniques are therefore based on the measurement of heat that may be generated (exothermic process), consumed (endothermic process) by a sample. 24

Calorimeter A calorimeter is a device used to measure heat of reaction. To measure heats of reactions, enclose reactants in a calorimeter, initiate the reaction, and measure the temperature difference before and after the reaction. The temperature difference enables us to evaluate the heat released in the reaction. The heat capacity is the amount of heat required to raise the temperature of the entire calorimeter by 1 K. 25

The Bomb Calorimeter For combustion reactions, all reactants are enclosed in an explosive-proof steel container, called the bomb whose volume does not change during a reaction. The bomb is then submerged in water or other liquid that absorbs the heat of reaction. The application of bomb calorimeter is called the bomb calorimetery. 26

The Bomb Calorimeter 27
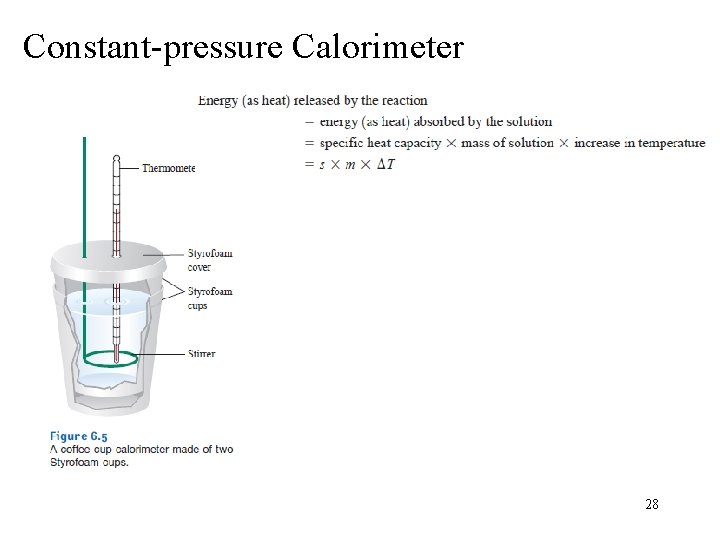
Constant-pressure Calorimeter 28

Example 29

Heat capacity Vs. Specific Heat 30

31

Solution 32

33

34

Solution 35

Recommended Exercises • Pages 275 -278 Problems #: 13, 15, 16, 17, 30, 31, 32, 33, 39, 41, 43, 49, 51, 67, 72, 73. 36

THE END 37
- Slides: 37