Thermochemistry 1 Thermochemistry p Thermodynamics is the science




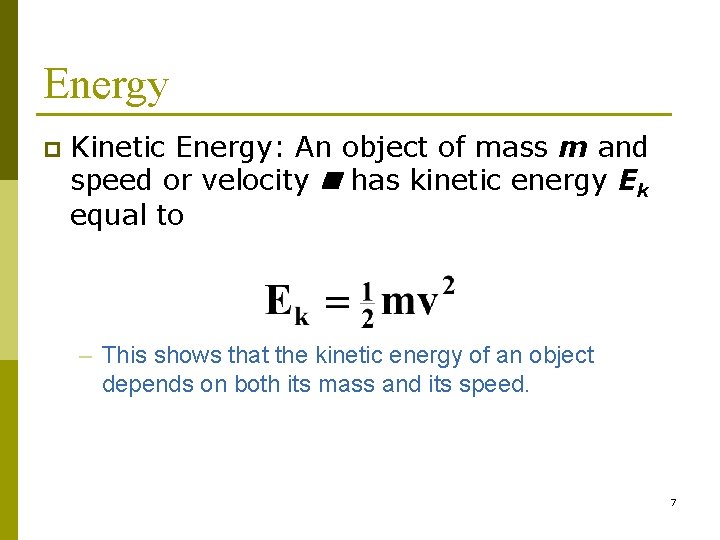

















































- Slides: 58

Thermochemistry 1

Thermochemistry p Thermodynamics is the science of the relationship between heat and other forms of energy. • Thermochemistry is the study of the quantity of heat absorbed or evolved by chemical reactions. 2

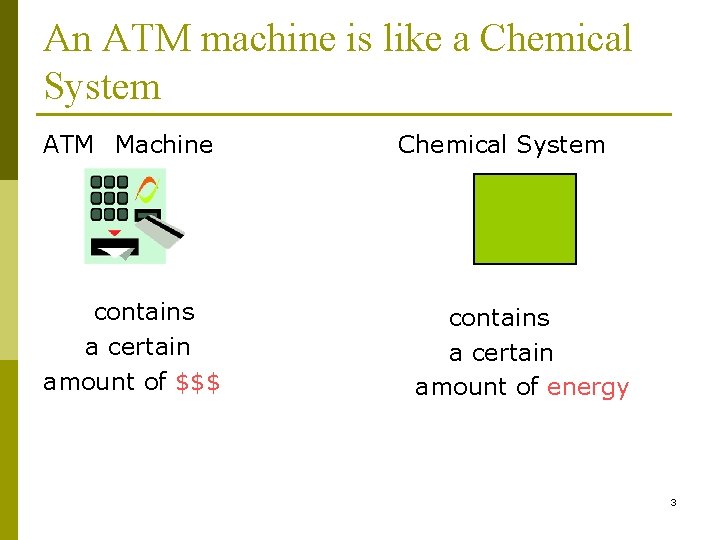
An ATM machine is like a Chemical System ATM Machine contains a certain amount of $$$ Chemical System contains a certain amount of energy 3

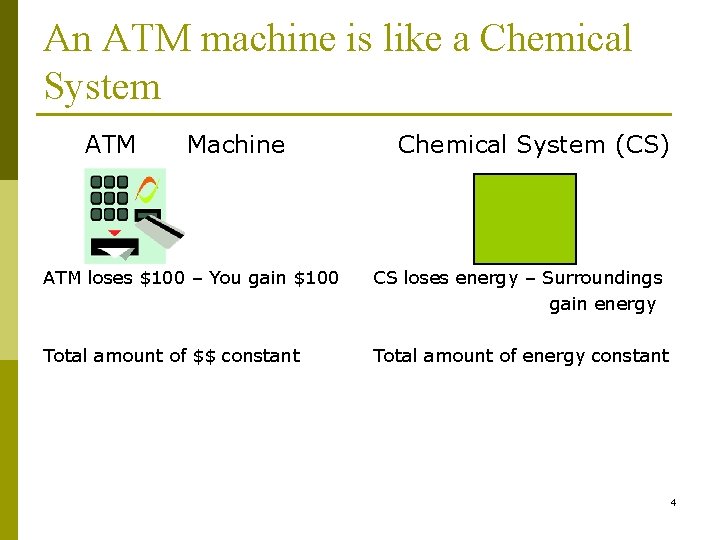
An ATM machine is like a Chemical System ATM Machine Chemical System (CS) ATM loses $100 – You gain $100 CS loses energy – Surroundings gain energy Total amount of $$ constant Total amount of energy constant 4

Energy p Energy is defined as the capacity to move matter. Energy can be in many forms: – Radiant Energy -Electromagnetic radiation. – Thermal Energy - Associated with random motion of a molecule or atom. – Chemical Energy - Energy stored within the structural limits of a molecule or atom. 5

Energy p There are three broad concepts of energy: – Kinetic Energy is the energy associated with an object by virtue of its motion. – Potential Energy is the energy an object has by virtue of its position in a field of force. – Internal Energy is the sum of the kinetic and potential energies of the particles making up a substance. We will look at each of these in detail. 6
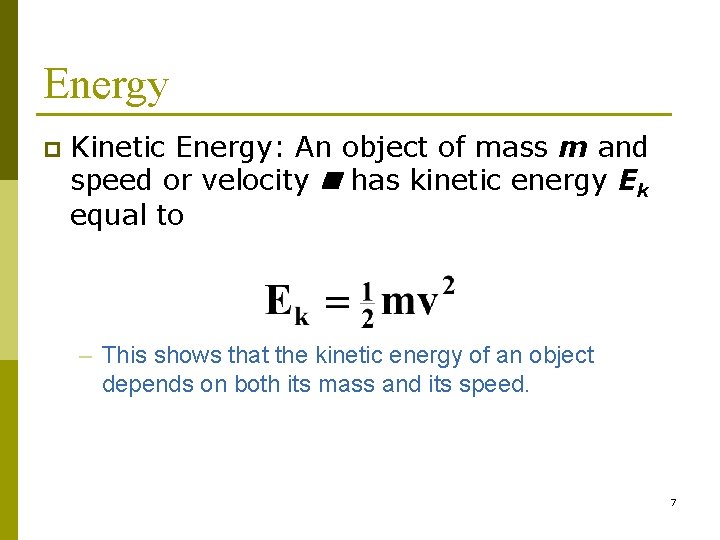
Energy p Kinetic Energy: An object of mass m and speed or velocity has kinetic energy Ek equal to – This shows that the kinetic energy of an object depends on both its mass and its speed. 7

A Problem to Consider p Consider the kinetic energy of a person whose mass is 130 lb (59. 0 kg) traveling in a car at 60 mph (26. 8 m/s). – The SI unit of energy, kg. m 2/s 2, is given the name Joule. 8

Energy p Potential Energy: This energy depends on the “position” (such as height) in a “field of force” (such as gravity). • For example, water of a given mass m at the top of a dam is at a relatively high “position” h in the “gravitational field” g of the earth. 9

A Problem to Consider p Consider the potential energy of 1000 lb of water (453. 6 kg) at the top of a 300 foot dam (91. 44 m). 10

Energy p Internal Energy is the energy of the particles making up a substance. • The total energy of a system is the sum of its kinetic energy, potential energy, and internal energy, U. 11

Energy p The Law of Conservation of Energy: Energy may be converted from one form to another, but the total quantities of energy remain constant. 12

Heat of Reaction p In chemical reactions, heat is often transferred from the “system” to its “surroundings, ” or vice versa. • The substance or mixture of substances under study in which a change occurs is called thermodynamic system (or simply system. ) • The surroundings are everything in the vicinity of thermodynamic system. 13

Heat of Reaction p Heat is defined as the energy that flows into or out of a system because of a difference in temperature between the system and its surroundings. • Heat flows from a region of higher temperature to one of lower temperature; once the temperatures become equal, heat flow stops. (See Animation: Kinetic Molecular Theory/Heat Transfer) 14

Heat of Reaction p Heat is denoted by the symbol q. n n The sign of q is positive if heat is absorbed by the system. The sign of q is negative if heat is evolved by the system. – Heat of Reaction is the value of q required to return a system to the given temperature at the completion of the reaction. 15

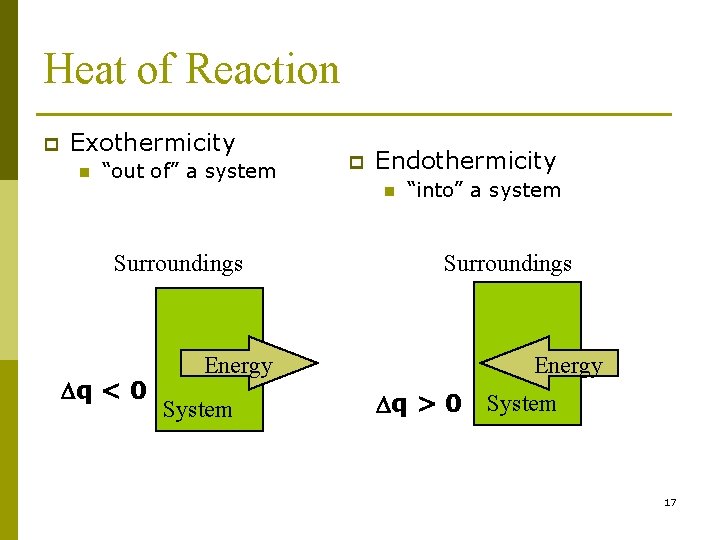
Heat of Reaction An exothermic process is a chemical reaction or physical change in which heat is evolved (q is negative). p An endothermic process is a chemical reaction or physical change in which heat is absorbed (q is positive). p 16

Heat of Reaction p Exothermicity n “out of” a system p Endothermicity n Surroundings Dq < 0 “into” a system Surroundings Energy System Energy Dq > 0 System 17

Enthalpy and Enthalpy Change p The heat absorbed or evolved by a reaction depends on the conditions under which it occurs. • Usually, a reaction takes place in an open vessel, and therefore at the constant pressure of the atmosphere. • The heat of this type of reaction is denoted qp, the heat at constant pressure. 18

Enthalpy and Enthalpy Change p Enthalpy, denoted H, is an extensive property of a substance that can be used to obtain the heat absorbed or evolved in a chemical reaction. – An extensive property is one that depends on the quantity of substance. – Enthalpy is a state function, a property of a system that depends only on its present state and is independent of any previous history of the system. 19

Enthalpy and Enthalpy Change p The change in enthalpy for a reaction at a given temperature and pressure (called the enthalpy of reaction) is obtained by subtracting the enthalpy of the reactants from the enthalpy of the products. 20

Enthalpy and Enthalpy Change p The change in enthalpy is equal to the heat of reaction at constant pressure. This represents the entire change in internal energy (DU) minus any expansion “work” done by the system. 21

Enthalpy and Enthalpy Change p Enthalpy and Internal Energy – The internal energy of a system, U, is precisely defined as the heat at constant pressure plus the work done by the system: (See Animation: Work vs. Energy Flow) – In chemical systems, work is defined as a change in volume at a given pressure, that is: 22

Enthalpy and Enthalpy Change p Since the heat at constant pressure, qp, represents DH, then – So ΔH is essentially the heat obtained or absorbed by a reaction in an open vessel where the work portion of ΔU is unmeasured. 23

Thermochemical Equations p A thermochemical equation is the chemical equation for a reaction (including phase labels) in which the equation is given a molar interpretation, and the enthalpy of reaction for these molar amounts is written directly after the equation. 24

Thermochemical Equations p In a thermochemical equation it is important to note phase labels because the enthalpy change, DH, depends on the phase of the substances. 25

Thermochemical Equations p The following are two important rules for manipulating thermochemical equations: – When a thermochemical equation is multiplied by any factor, the value of ΔH for the new equation is obtained by multiplying the ΔH in the original equation by that same factor. – When a chemical equation is reversed, the value of ΔH is reversed in sign. 26

Applying Stoichiometry and Heats of Reactions p Consider the reaction of methane, CH 4, burning in the presence of oxygen at constant pressure. Given the following equation, how much heat could be obtained by the combustion of 10. 0 grams CH 4? 27

Measuring Heats of Reaction p To See how heats of reactions are measured, we must look at the heat required to raise the temperature of a substance, because a thermochemical measurement is based on the relationship between heat and temperature change. • The heat required to raise the temperature of a substance is its heat capacity. 28

Measuring Heats of Reaction p Heat Capacity and Specific Heat – The heat capacity C, of a sample of substance is the quantity of heat required to raise the temperature of the sample of substance one degree Celsius. – Changing the temperature of the sample requires heat equal to: 29

A Problem to Consider p Suppose a piece of iron requires 6. 70 J of heat to raise its temperature by one degree Celsius. The quantity of heat required to raise the temperature of the piece of iron from 25. 0 o. C to 35. 0 o. C is: 30

Measuring Heats of Reaction p Heat capacities are also compared for one gram amounts of substances. The specific heat capacity (or “specific heat”) is the heat required to raise the temperature of one gram of a substance by one degree Celsius. – To find the heat required you must multiply the specific heat, s, of the substance times its mass in grams, m, and the temperature change, DT. 31

A Problem to Consider p Calculate the heat absorbed when the temperature of 15. 0 grams of water is raised from 20. 0 o. C to 50. 0 o. C. (The specific heat of water is 4. 184 J/g. o. C. ) 32

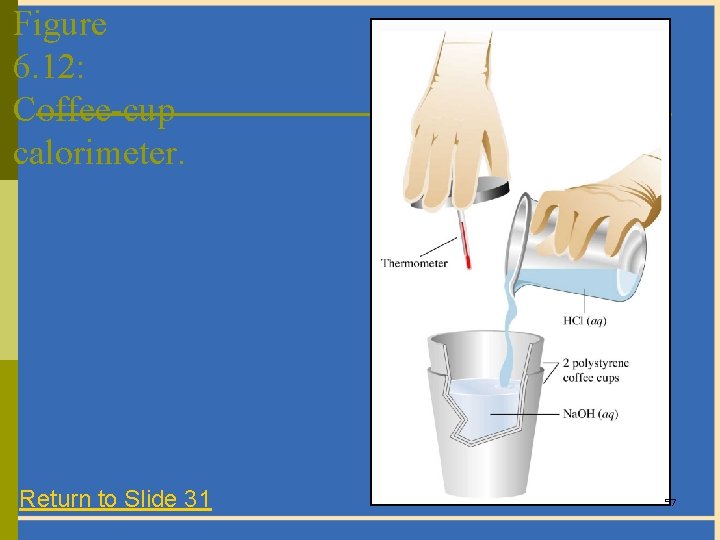
Heats of Reaction: Calorimetry p A calorimeter is a device used to measure the heat absorbed or evolved during a physical or chemical change. (See Figure 6. 12) – The heat absorbed by the calorimeter and its contents is the negative of the heat of reaction. 33

A Problem to Consider p When 23. 6 grams of calcium chloride, Ca. Cl 2, was dissolved in water in a calorimeter, the temperature rose from 25. 0 o. C to 38. 7 o. C. If the heat capacity of the solution and the calorimeter is 1258 J/o. C, what is the enthalpy change per mole of calcium chloride? 34

Heats of Reaction: Calorimetry • First, let us calculate the heat absorbed by the calorimeter. • Now we must calculate the heat per mole of calcium chloride. 35

Heats of Reaction: Calorimetry p Calcium chloride has a molecular mass of 111. 1 g, so • Now we can calculate the heat per mole of calcium chloride. 36

Hess’s Law p Hess’s law of heat summation states that for a chemical equation that can be written as the sum of two or more steps, the enthalpy change for the overall equation is the sum of the enthalpy changes for the individual steps. (See Animation: Hess’s Law) 37

Hess’s Law p For example, suppose you are given the following data: • Could you use these data to obtain the enthalpy change for the following reaction? 38

Hess’s Law p If we multiply the first equation by 2 and reverse the second equation, they will sum together to become third. 39

Standard Enthalpies of Formation p The term standard state refers to the standard thermodynamic conditions chosen for substances when listing or comparing thermodynamic data: 1 atmosphere pressure and the specified temperature (usually 25 o. C). – The enthalpy change for a reaction in which reactants are in their standard states is denoted ΔHo (“delta H zero” or “delta H naught”). 40

Standard Enthalpies of Formation p The standard enthalpy of formation of a substance, denoted DHfo, is the enthalpy change for the formation of one mole of a substance in its standard state from its component elements in their standard state. – Note that the standard enthalpy of formation for a pure element in its standard state is zero. 41

Standard Enthalpies of Formation p The law of summation of heats of formation states that the enthalpy of a reaction is equal to the total formation energy of the products minus that of the reactants. - S is the mathematical symbol meaning “the sum of”, and m and n are the coefficients of the substances in the chemical equation. 42

A Problem to Consider p Large quantities of ammonia are used to prepare nitric acid according to the following equation: – What is the standard enthalpy change for this reaction? Use Table 6. 2 for data. 43

A Problem to Consider p You record the values of DHfo under the formulas in the equation, multiplying them by the coefficients in the equation. • You can calculate DHo by subtracting the values for the reactants from the values for the products. 44

A Problem to Consider p Using the summation law: – Be careful of arithmetic signs as they are a likely source of mistakes. 45

Fuels p A fuel is any substance that is burned to provide heat or other forms of energy. • In this section we will look at: – Foods as fuels – Fossil fuels – Coal gasification and liquefaction 46

Fuels p Food fills three needs of the body: – It supplies substances for the growth and repair of tissue. – It supplies substances for the synthesis of compounds used in the regulation of body processes. – It supplies energy. About 80% of the energy we need is for heat. The rest is used for muscular action and other body processes 47

Fuels p A typical carbohydrate food, glucose (C 6 H 12 O 6) undergoes combustion according to the following equation. – One gram of glucose yields 15. 6 k. J (3. 73 kcal) when burned. 48

Fuels p A representative fat is glyceryl trimyristate, C 45 H 86 O 6. The equation for its combustion is: – One gram of fat yields 38. 5 k. J (9. 20 kcal) when burned. Note that fat contains more than twice the fuel per gram than carbohydrates contain. 49

Fuels p Fossil fuels account for nearly 90% of the energy usage in the United States. – Anthracite, or hard coal, the oldest variety of coal, contains about 80% carbon. – Bituminous coal, a younger variety of coal, contains 45% to 65% carbon. – Fuel values of coal are measured in BTUs (British Thermal Units). – A typical value for coal is 13, 200 BTU/lb. – 1 BTU = 1054 k. J 50

Fuels p Natural gas and petroleum account for nearly three-quarters of the fossil fuels consumed per year. – Purified natural gas is primarily methane, CH 4, but also contains small quantities of ethane, C 2 H 6, propane, C 3 H 8, and butane, C 4 H 10. – We would expect the fuel value of natural gas to be close to that for the combustion of methane. 51

Fuels p Petroleum is a very complicated mixture of compounds. – Gasoline, obtained from petroleum, contains many different hydrocarbons, one of which is octane, C 8 H 18. – This value of DHo is equivalent to 44. 4 k. J/gram. 52

Fuels p With supplies of petroleum estimated to be 80% depleted by the year 2030, the gasification of coal has become a possible alternative. – First, coal is converted to carbon monoxide using steam. – The carbon monoxide can then be used to produce a variety of other fuels, such as methane. 53

Operational Skills p p p p Calculating kinetic energy. Writing thermochemical equations. Manipulating thermochemical equations. Calculating the heat of reaction from the stoichiometry. Relating heat and specific heat. Calculating DH from calorimetric data. Applying Hess’s law. Calculating the enthalpy of reaction from standard enthalpies of formation. 54

Animation: Kinetic Molecular Theory/Heat Transfer (Click here to open Quick. Time animation) Return to Slide 12 55

Animation: Work vs. Energy Flow (Click here to open Quick. Time animation) Return to Slide 20 56

Figure 6. 12: Coffee-cup calorimeter. Return to Slide 31 57

Animation: Hess’s Law (Click here to open Quick. Time animation) Return to Slide 35 58