Thermochemical Reactions Enthalpy Remember Exothermic reactions q H

Thermochemical Reactions

Enthalpy • Remember: • Exothermic reactions: -q = -ΔH = negative • Endothermic reactions: q = ΔH = positive • If energy is a product, the reaction is exothermic and ΔH is negative! • If energy is a reactant, the reaction is endothermic and ΔH is positive!

Thermochemical Equations • Problems involving enthalpy changes are similar to stoichiometry problems! • We have to relate moles to energy here! • Ca. O + H 2 O Ca(OH)2 + 65. 2 k. J • 1 mole Ca(OH)2 = -65. 2 k. J energy • 1 mol Ca(OH)2 or -65. 2 k. J 1 mol Ca(OH)2

Thermochemical Equations • Calculate the amount of heat produced when 3. 4 mol of Ca(OH)2 is formed. • Ca. O + H 2 O Ca(OH)2 + 65. 2 k. J • Given • 3. 4 mol Ca(OH)2 Find ΔH, k. J • -65. 2 k. J 1 mol Ca(OH)2 ΔH = 3. 4 mol Ca(OH)2 -65. 2 k. J = -221. 68 k. J 1 mol Ca(OH)2

Thermochemical Equations • Calculate the amount of energy needed to react 36. 75 g of C with sulfur. • C + 2 S CS 2 ΔH = 89. 3 k. J Given 36. 75 g C Find ΔH, k. J • 1 mol C 89. 3 k. J ΔH = 36. 75 g C 1 mol C 12. 01 g C 89. 3 k. J = 273. 25 k. J 1 mol C
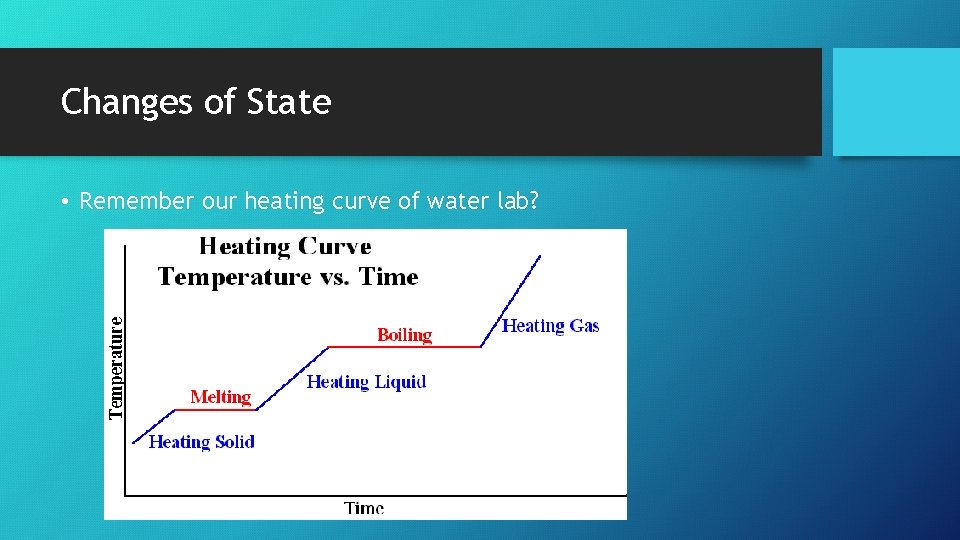
Changes of State • Remember our heating curve of water lab?

Changes of State • All solids absorb heat as they melt to become liquids. • The gain of heat causes a change of state instead of an increase in temperature. • Temperature remains constant.

Changes of State • The heat absorbed by one mole of a solid substance as it melts to a liquid at constant temperature is the molar heat of fusion (ΔHfus). • The quantity of heat absorbed by a melting solid is exactly the same as the quantity of heat released when the liquid solidifies. • ΔHfus = -ΔHsolid • The heat absorbed by one mole of a liquid substance as it vaporizes to a gas at constant temperature is the molar heat of vaporization (ΔHvap). • The quantity of heat absorbed by a vaporizing liquid is exactly the same as the quantity of heat released when the gas condenses. • ΔHvap = -ΔHcond

Changes of State
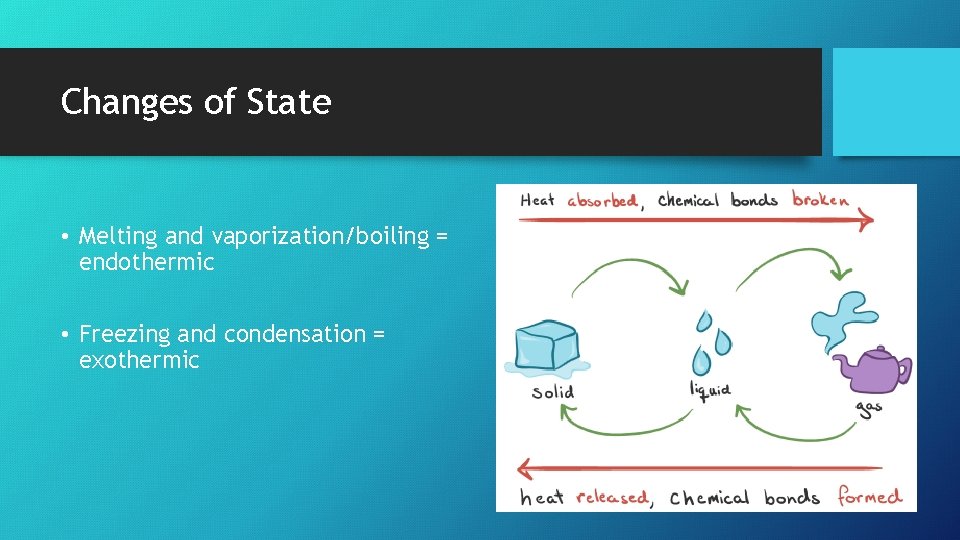
Changes of State • Melting and vaporization/boiling = endothermic • Freezing and condensation = exothermic

Heat of Solution • During the formation of a solution, heat is either released or absorbed. • The enthalpy change caused by one mole of a substance is the molar heat of solution • ΔHsoln • Ca. Cl 2 (s) Ca 2+ (aq) + 2 Cl- (aq) ΔHsoln = -82. 8 k. J/mol

Heat of Solution • Calculate the amount of heat released when 3. 5 mol of Ca. Cl 2 is dissolved in water. • Ca. Cl 2 (s) Ca 2+ (aq) + 2 Cl- (aq) ΔHsoln = -82. 8 k. J/mol • Given 3. 5 mol Ca. Cl 2 ΔHsoln = -82. 8 k. J/mol ΔH = 3. 5 mol Ca. Cl 2 Find ΔH, k. J -82. 8 k. J = -289. 8 k. J 1 mol Ca. Cl 2
- Slides: 12