Thermal Energy October 2012 Objectives To identify temperature

Thermal Energy. October 2012

Objectives. • To identify temperature as the amount of kinetic energy of atoms and molecules. • To identify different temperature scales and convert from one scale to another. • To describe heat as the transfer of energy between substances at different temperature. • To identify the three methods of heat transfer. • To apply the principle of energy conservation to calculate changes in potential, kinetic and internal energy. • To identify latent and specific heat of substances. • To calculate phase changes of different substances. • To describe thermal expansion.

Temperature In every day life, temperature is the measure of how hot or cold something is. Many properties of matter change with it. For example, most materials expand when heated.

Temperature Scales To measure temperature quantitatively we use numerical scales. The most common scale today is Celsius scale. In USA the Fahrenheit scale is also common. And the most important scale for scientific work is the absolute, or Kelvin, scale. The conversion between the two temperature scales Celsius and Fahrenheit can be written as:

Absolute Temperature Robert Boyle (1627 -1691) established a relation known as Boyle’s Law that states that the volume of a gas is inversely proportional to the absolute pressure applied to it when the temperature is kept constant. Temperature also affects the volume of a gas. Jacques Charles (17461823) found that when the pressure is kept not too high and constant, the volume of a gas increases with temperature at a nearly constant rate. The graph of volume vs. temperature is a straight line that if projected to low temperatures it crosses the axis at about -273 C. Such a graph can be drawn for any gas, and the straight line always projects back to -273 C at zero volume. At lower temperatures the gas would have a negative volume, which makes no sense. It could be argued that -273 C is the lowest temperature possible. This temperature is called the ABSOLUTE ZERO of temperature and is the basis of the Kelvin scale. T( K) = T( C) + 273. 15

Practice

Thermal Equilibrium If two objects at different temperatures are placed in thermal contact (meaning thermal energy can transfer from one to the other) the two objects will eventually reach the same temperature. They are then said to be in thermal equilibrium.

Kinetic Theory


Heat is defined as the transferred energy from one object to another because of a difference in temperature. The common unit for heat is the calorie (cal) and is defined as the amount of heat needed to raise the temperature of one gram of water by 1 Celsius degree. More often used than the calorie is the kilocalorie (kcal) equivalent to 1000 calories. It is also called a Calorie (With a capital C), and it is by this unit that the energy value of food is specified. In the British system of units the Btu (British Thermal Unit) is used and corresponds to 0. 252 kcal = 1055 J.

James Prescott Joule (1818 -1889) and others performed a number of experiments that were crucial for establishing the view that heat, like work, represents a transfer of energy. Joule determined that a given amount of work done was always equivalent to a particular amount of heat input. Quantitatively 4. 186 joules of work was found to be the equivalent to 1 calorie of heat. This is known as the mechanical equivalent of heat.

Working off the extra Calories Suppose you throw your caution to the wind and eat too much ice cream and cake on the order of 500 Calories. To compensate, you want to do an equivalent amount of work climbing stairs or a mountain. How much total height must you climb? Remember W = ΔPE = mgh NOTE: The human body does not transform energy with 100% efficiency it is more like 20% efficient. So you will actually have to climb 20% of the way (Which is still a lot)

Temperature, Heat and Internal Energy Using the kinetic theory, we can make a clear distinction between temperature, heat and internal energy (sometimes referred as thermal energy). Temperature (in kelvins) is a measure of the average kinetic energy of individual molecules. Internal Energy refers to the total energy of all the molecules in the object. Heat refers to a transfer of energy from one object to another because of a difference in temperature.

Experiment Material Needed: Two identical glasses. Hot Water. Cold Water. Liquid Food Coloring. Fill your glasses. One should have hot water in it, the other cold water. Pick a color of food coloring. Put three drops of food coloring in each glass. Share your observations

Specific Heat If heat flows into an object, the object temperature rises (assuming no phase change). But how much does the temperature rise? It depends. The amount of heat Q required to change the temperature of a given material is proportional to the mass and the change in temperature. Q = mcΔT Where c is a quantity characteristic of the material called its specific heat.

Example How much heat input is needed to raise the temperature of an empty 20 kg vat made of iron from 10 C to 90 C? What if the vat is filled with 20 kg of water?

Calorimetry When different parts of an isolated system are at different temperatures, heat will flow from the part at higher temperature to the part at lower temperature, that is within the system. If the system is isolated no energy is transferred into or out of it. So the conservation of energy again plays an important role. The heat lost by one part of the system is equal to the heat gained by the other part. Heat lost = Heat gained Energy out of one part = energy into another part.

Example If 0. 20 kg of tea at 95 C is poured into a 150 g glass cup initially at 25 C what will be the common final temperature T of the tea and cup when equilibrium is reached, assuming no heat flows to the surroundings. Solution: 86 C

Practice * An automobile cooling system holds 16 kg of water. How much heat does it absorb if its temperature rises from 20 C to 90 C? * A 35 g glass thermometer reads 21. 6 C before it is placed in 135 g of water. When the water and thermometer come to equilibrium, thermometer reads 39. 2 C. What was the original temperature of the water? Solution: 40. 1 C

Collaborative Work : HEAT TRANSFER • The students are assigned in groups of three persons. The sorting method will be by student ID number as per the assistance list. • Each team will assign one member to be the captain of the team. • A second member will be the spoke person and will present the results. • The third member will be the closer and responsible for presenting the group conclusions on the activity. • The teams will have 25 minutes to complete the research. • Then in 10 minutes the teams will present the work to the evaluators. • And after that 5 minutes to present your conclusions.

Heat Transfer Types The Students must construct a collage of images containing the three (3) types of Heat Transfer 1. Conduction 2. Convection 3. Radiation Clearly define in three sections of the collage these different types.

Evaluation CATEGORY Distinguished (4) Proficient (3) Apprentice (2) Novice (1) Physic Concepts Show complete Show substantial 30% understanding of the physics concepts used. Clearly Demonstrates deep substantial understanding of the lesson overall. the overall lesson. Show partial understanding of the physics concepts used. Demonstrates partial understanding of the overall lesson. Show complete lack or very limited understanding of the underlying concepts needed OR are not provided. Demonstrates complete lack of understanding of the overall lesson. Real World Models Real world models 50% indicate strong evidence of creativity and imagination. Real world models show partial evidence of creativity and imagination. Models are messy, difficult to understand, OR missing. Real world models lack creativity OR are not provided. Real world models shows substantial evidence of creativity and imagination.

Evaluation Continued CATEGORY Verbal and nonverbal communication 20% Distinguished (4) Proficient (3) Apprentice (2) Novice (1) Accomplishes all of Accomplishes most Accomplishes just Accomplishes none the following: Good of the following: three or two of the or just one of the tone and voice Good tone and following: Good volume, fluid voice volume, fluid tone and voice speaking, adequate speaking, body volume, fluid body stance, good use of speaking, body use of space and stance, good use of resources. space and . resources.
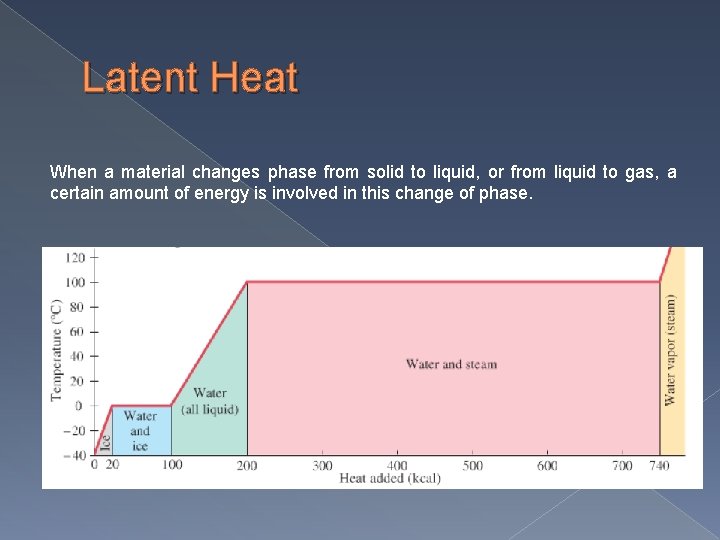
Latent Heat When a material changes phase from solid to liquid, or from liquid to gas, a certain amount of energy is involved in this change of phase.
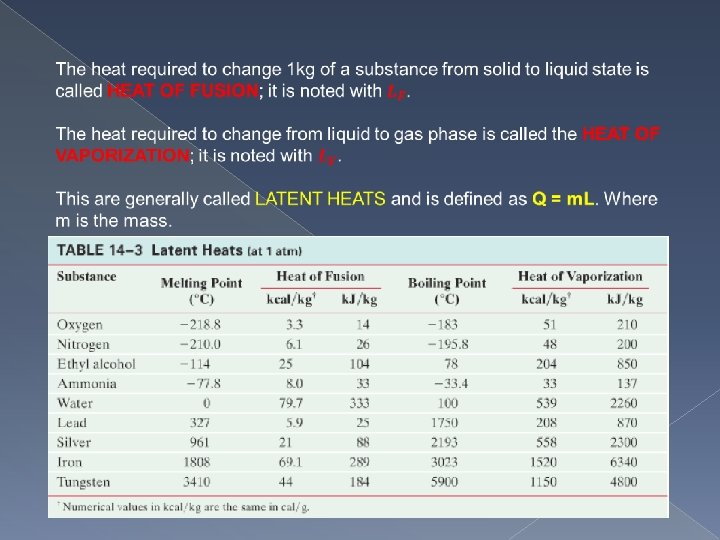

Example: Making Ice How much energy does a freezer have to remove from 1. 5 kg of water at 20 C to make ice at -12 C. Approach: We need to calculate the energy to reduce the water from 20 to 0 degrees, then to change it to ice, and then to reduce the ice from 0 to -12 degrees. Solution: 660 k. J.

Practice How much heat is needed to melt 16. 50 kg of silver that is initially at 20 C? What mass of steam at 100 C must be added to 1 kg of ice at 0 C to yield liquid water at 20 C?

Evaporation The latent heat to change a liquid to a gas is needed not only at the boiling point. Water can change from liquid to gas even at room temperature. This process is called evaporation. The value of the heat of vaporization increases slightly with a decrease in temperature. At 20 C for example it is 2450 k. J/kg compared to 2260 k. J/kg at 100 C. When water evaporates, the remaining liquid cools because the energy required to evaporate comes from the water itself, (Internal Energy) and therefore the temperature must drop. Evaporation of water from the skin is one of the most important methods the body uses to control temperature.

Heat Transfer: Conduction When a silver spoon is placed in a hot bowl of soup, the end that you hold becomes hot as well, even though it is not directly in contact with the source of heat. We say the heat has been conducted from the hot end to the cold end. Heat conduction is carried via molecular collisions. Some materials are good heat conductors, while others are bad conductors and therefore are good insulators.

Heat Transfer: Convection Although liquids and gases are not generally not very good conductors of heat, they can transfer heat quite rapidly by convection. Convection is the process whereby heat flows by the mass movement of molecules from one place to another. A forced air furnace, in which air is heated and then blown by a fan into a room is an example of forced convection. Natural convection occurs as well, one familiar example is hot air rises.

Heat Transfer: Radiation Convection and Conduction require the presence of matter as a medium to carry heat. Radiation occurs without any medium at all. Radiation consists essentially of electromagnetic waves. The heat received from the sun is the most common example of radiation. The warmth we receive from fire is mainly radiant energy. Most of the air heated by a fire in a fireplace rises by convection up the chimney and does not reach us.

Collaborative Activity. Using the world wide web, research how a thermos bottles work. Specify how each type of heat transfer is prevented to keep temperature. In groups of two, write a small report with your findings

Video: Cooling a Six Pack Watch the video and then answer the questions that follow. http: //www. youtube. com/watch? v=N 354 f. Chhj. V 0

Questions: 1. What is theory behind the myth of burying a six pack in the sand, pouring gasoline over and set it on fire is supposed to cool the six pack ? 2. Why does Adam proposes checking various traditional cooling methods? 3. How is that the fire extinguisher cools the six pack? 4. Why do you think both Adam and Jamie devices use spiral forms? 5. In the end which way do you think is the most time-cost efficient method to cool a six pack?

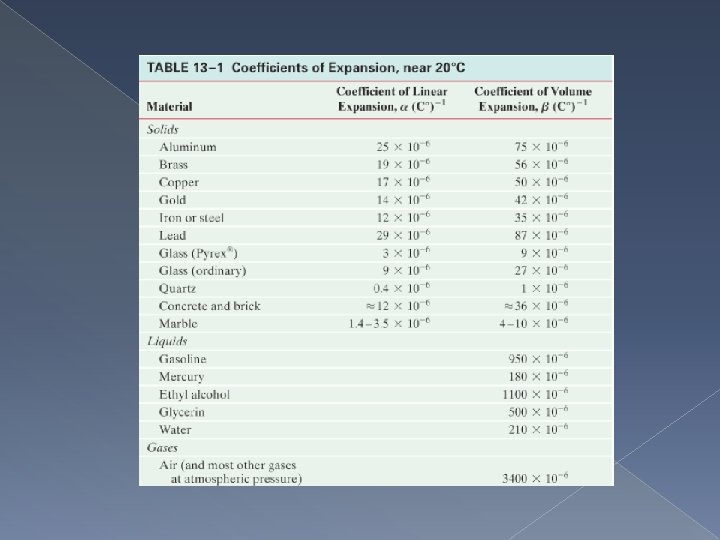
Thermal Expansion Most substances expand when heated and contract when cooled. However the amount of expansion or contraction varies, depending on the material.

Linear Expansion

Volume Expansion

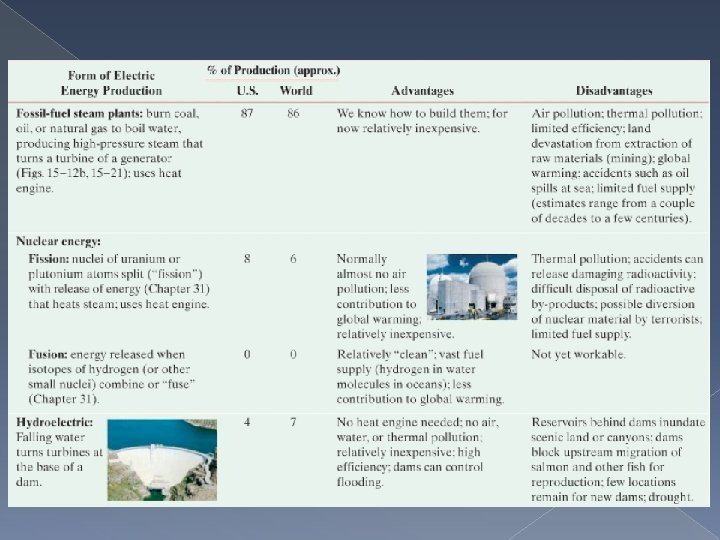
Thermal Pollution and Global Warming Much of the energy we use in every day life makes use of a heat engine. Over 90% of the electric energy produced in the U. S. is generated at fossil-fuel plants, where oil, coal or gas is burned to move a steam engine.

Thermal Pollution is the heat produced by every heat engine and is absorbed by the environment such as rivers or lakes, or by the air using large cooling towers. In the case of cooling towers, the output heat raises the temperature of the atmosphere, which affects weather.

Air Pollution Chemicals released in the burning of fossil fuels gives rise to smog and other problems. One big problem is the buildup of Carbon Dioxide in Earth’s atmosphere. Carbon Dioxide absorbs infrared radiation that the Earth naturally emits, causing Global Warming, a serious problem that can be addressed by limiting the burning of fossil fuels. Thermal pollution however is unavoidable. Engineers can try to design and build more efficient engines. But nature imposes a limit to efficiency defined in the second law of thermodynamics that says 100% efficiency is not possible to achieve. What we can do is use less energy and conserve our fuel resources.


References • Giancoli , Douglas C. Physics Sixth Edition. USA Pearson 2005 • Serway, Raymond A. Essentials of College Physics. USA Thomson 2007
- Slides: 44